Advanced Synthesis of 2-Pyridinecarboxamide Diaryl Ketones for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex nitrogen-containing heterocycles, particularly those serving as pivotal scaffolds for drug discovery. Patent CN102603624A introduces a robust and highly versatile preparation method for 2-pyridinecarboxamide diaryl ketone compounds, addressing critical bottlenecks in the synthesis of these valuable organic intermediates. This technology leverages a palladium-catalyzed direct acylation strategy, enabling the construction of the diaryl ketone motif with high atom economy and operational simplicity. By utilizing readily available pyridine-2-carboxyl arylamide compounds and aryl iodides, this method bypasses the need for cumbersome pre-functionalization steps often required in classical organic synthesis. The resulting compounds are not only stable and easy to handle but also possess significant biological activity potential, making them attractive candidates for further medicinal chemistry optimization. Furthermore, the ability to hydrolyze these intermediates into 2-aminodiaryl ketones adds a layer of strategic value, positioning this synthesis as a key enabling technology for reliable pharmaceutical intermediate supplier networks aiming to diversify their portfolio with high-value scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to diaryl ketones and related aminoketone scaffolds often suffer from significant inefficiencies that hinder large-scale manufacturing. Classical Friedel-Crafts acylation, while conceptually simple, frequently requires harsh Lewis acidic conditions that are incompatible with sensitive functional groups often found in advanced drug candidates. Moreover, achieving regioselectivity in poly-substituted aromatic systems can be challenging, leading to complex mixtures of isomers that require energy-intensive separation processes. Alternative methods involving organometallic reagents, such as Grignard or organolithium species, demand strict anhydrous conditions and cryogenic temperatures, drastically increasing operational costs and safety risks in a production environment. These conventional approaches often lack the modularity required for rapid library synthesis, limiting the ability of R&D teams to explore structure-activity relationships efficiently. The reliance on multiple protection and deprotection steps further elongates the synthetic timeline, reducing overall throughput and increasing the environmental footprint due to excessive solvent and reagent consumption.
The Novel Approach
In stark contrast, the methodology disclosed in the patent offers a streamlined, transition-metal catalyzed solution that fundamentally reshapes the synthetic landscape for these targets. By employing a palladium catalyst in conjunction with a silver oxidant, the process facilitates a direct C-H activation and subsequent acylation at the ortho-position relative to the amide directing group. This transformation proceeds smoothly in high-boiling organic solvents like xylene or mesitylene at temperatures ranging from 100°C to 150°C, eliminating the need for extreme cryogenic conditions. The reaction demonstrates exceptional substrate tolerance, accommodating a wide array of electronic and steric environments on both the pyridine and aryl iodide components. As illustrated in the general reaction scheme below, the process converts simple starting materials directly into the target 2-pyridinecarboxamide diaryl ketones with impressive efficiency.
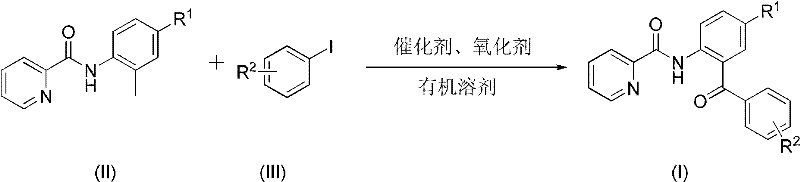
The versatility of this approach is further evidenced by the successful synthesis of diverse derivatives, including those bearing electron-withdrawing groups like cyano and trifluoromethyl, as well as electron-donating groups such as methoxy and methyl. This broad scope ensures that the method is applicable to cost reduction in pharmaceutical intermediate manufacturing across a wide spectrum of chemical space, allowing for the rapid generation of analogues without redesigning the core synthetic strategy.
Mechanistic Insights into Pd-Catalyzed Direct Acylation
The core of this transformative synthesis lies in the intricate interplay between the palladium catalyst and the silver oxidant, which drives the catalytic cycle forward through a series of well-defined organometallic steps. The reaction initiates with the coordination of the palladium(II) species to the nitrogen atom of the pyridine-2-carboxamide substrate, acting as a bidentate directing group that guides the metal center to the proximal ortho-C-H bond. This proximity effect facilitates the cleavage of the C-H bond via a concerted metalation-deprotonation (CMD) mechanism or electrophilic palladation, generating a stable five-membered palladacycle intermediate. This step is crucial as it determines the regioselectivity of the reaction, ensuring that acylation occurs exclusively at the desired position adjacent to the amide linkage. Following C-H activation, the aryl iodide substrate undergoes oxidative addition to the palladium center, formally increasing the oxidation state of the metal and incorporating the aryl moiety into the coordination sphere.
Subsequent migratory insertion or reductive elimination steps forge the new carbon-carbon bond between the aryl ring and the carbonyl carbon, releasing the final diaryl ketone product and regenerating the active palladium species. The silver oxidant plays a dual role in this cycle: it acts as a halide scavenger to facilitate the oxidative addition of the aryl iodide and serves as a terminal oxidant to re-oxidize any reduced palladium(0) species back to the active palladium(II) state, thereby sustaining the catalytic turnover. This mechanistic elegance allows for the use of catalytic amounts of palladium (typically 0.1 to 0.3 mmol relative to 1 mmol of substrate), significantly reducing the burden of heavy metal residues in the final product. The careful selection of ligands and additives, although not explicitly detailed as variable in every example, implies a robust system where the inherent directing power of the picolinamide group drives the reaction forward with high fidelity, minimizing the formation of undesired by-products and simplifying the purification process for high-purity OLED material or pharmaceutical precursor applications.
How to Synthesize 2-Pyridinecarboxamide Diaryl Ketones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the pyridine-2-carboxyl arylamide, the chosen aryl iodide, a divalent palladium catalyst such as Pd(OAc)2 or PdCl2, and a silver salt oxidant like AgOAc or Ag2CO3. The mixture is suspended in a suitable high-boiling solvent, typically xylene or mesitylene, to ensure homogeneous heating and efficient mass transfer. Detailed standardized synthesis steps see the guide below.
- Combine pyridine-2-carboxyl arylamide, aryl iodide, palladium catalyst, and silver oxidant in an organic solvent such as xylene or mesitylene.
- Heat the reaction mixture to a temperature between 100°C and 150°C and maintain stirring for 5 to 24 hours until completion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-pyridinecarboxamide diaryl ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the simplification of the supply chain for raw materials; since the method utilizes commodity chemicals like aryl iodides and picolinic acid derivatives, it reduces dependency on exotic or custom-synthesized building blocks that often carry long lead times and price volatility. This accessibility translates directly into enhanced supply chain reliability, ensuring that production schedules can be maintained without interruption due to material shortages. Furthermore, the operational simplicity of the reaction—requiring only heating and stirring followed by standard filtration and chromatography—lowers the barrier for technology transfer from R&D to manufacturing scales.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences inherently reduces the consumption of reagents, solvents, and labor hours associated with each synthetic transformation. By consolidating the construction of the diaryl ketone core into a single catalytic step, the overall process mass intensity (PMI) is significantly lowered, leading to substantial cost savings in waste disposal and raw material procurement. Additionally, the use of catalytic rather than stoichiometric amounts of expensive palladium minimizes the financial impact of precious metal usage, while the efficient recovery of silver by-products can further offset operational expenses.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups without the need for specialized handling, ensures consistent batch-to-batch quality. This reliability is critical for maintaining the continuity of supply for downstream customers who depend on these intermediates for their own API synthesis. The ability to source starting materials from multiple global vendors reduces the risk of single-source dependency, providing procurement teams with greater negotiating power and flexibility in managing vendor relationships.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in millimole scales with potential for expansion to kilogram and tonne levels using standard stainless steel reactors. The use of relatively benign solvents like xylene, which are widely accepted in industrial settings, simplifies regulatory compliance regarding volatile organic compound (VOC) emissions. Moreover, the simplified workup procedure reduces the volume of aqueous waste generated, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations without compromising productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and utility of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this route for potential adoption.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method utilizes direct C-H activation, eliminating the need for pre-functionalized substrates and reducing step count. It operates under relatively mild conditions (100-150°C) with commercially available catalysts like Pd(OAc)2, offering a streamlined pathway compared to multi-step traditional routes.
Q: Can this synthesis be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It uses robust solvents like xylene and mesitylene, and the workup involves simple filtration and standard column chromatography, making it suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the downstream application of these 2-pyridinecarboxamide compounds?
A: These compounds serve as versatile precursors. They can be easily hydrolyzed under basic conditions to yield 2-aminodiaryl ketone compounds, which are valuable building blocks in the synthesis of various active pharmaceutical ingredients (APIs).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridinecarboxamide Diaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing manufacturing costs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle complex catalytic reactions safely and effectively, guaranteeing the supply continuity that global pharmaceutical partners demand.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your supply chain.
