Scalable Pd-Catalyzed Synthesis of 2-Pyridinecarboxamide Diaryl Ketones for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to complex heterocyclic scaffolds, particularly those serving as critical precursors for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN102603624A, which discloses a novel preparation method for 2-pyridinecarboxamide diaryl ketone compounds. This technology represents a paradigm shift in how chemists approach the construction of nitrogen-containing heterocycles, moving away from laborious multi-step sequences toward direct, catalytic functionalization. The core innovation lies in the utilization of a palladium-catalyzed system that enables the direct acylation of pyridine-2-carboxyl arylamide compounds with aryl iodides. This approach not only streamlines the synthetic pathway but also enhances the overall atom economy of the process. For R&D directors and process chemists, understanding the nuances of this transformation is vital, as it offers a robust platform for generating diverse libraries of bioactive molecules. The versatility of this method allows for the introduction of various substituents, thereby facilitating the rapid optimization of lead compounds in drug discovery programs.
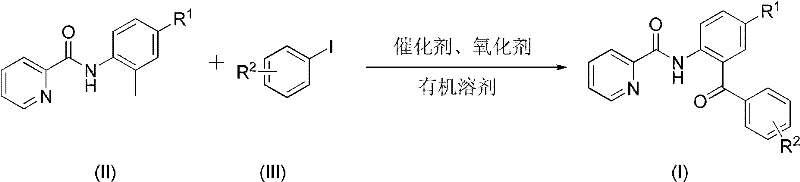
Furthermore, the economic implications of adopting this technology cannot be overstated for procurement and supply chain stakeholders. By simplifying the synthetic route, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing. The reliance on commercially available starting materials, such as substituted aryl iodides and picolinic acid derivatives, ensures a stable supply chain with minimal risk of raw material shortages. The process operates under relatively mild thermal conditions compared to traditional Friedel-Crafts acylations, which often require harsh Lewis acids and generate significant amounts of corrosive waste. Consequently, this method aligns perfectly with modern green chemistry principles, reducing the environmental footprint of chemical production. As a reliable pharmaceutical intermediate supplier, leveraging such advanced methodologies allows us to offer clients higher quality materials at competitive price points, ensuring continuity of supply for critical drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of diaryl ketone structures, especially those containing sensitive nitrogen heterocycles like pyridine, has been fraught with challenges. Classical approaches often involve Friedel-Crafts acylation, which suffers from poor regioselectivity and requires stoichiometric amounts of hazardous Lewis acids such as aluminum chloride. These conditions are incompatible with many functional groups, necessitating additional protection and deprotection steps that drastically increase the step count and reduce overall yield. Moreover, the workup for these reactions is notoriously difficult, generating large volumes of acidic waste that require expensive disposal procedures. Another common route involves the use of organometallic reagents like Grignard or organolithium species, which demand strictly anhydrous conditions and cryogenic temperatures, posing significant safety risks and operational costs on a large scale. The inability to tolerate diverse functional groups limits the structural diversity accessible through these conventional methods, hindering the exploration of chemical space in medicinal chemistry.
The Novel Approach
In stark contrast, the method disclosed in the patent utilizes a transition metal-catalyzed C-H activation strategy that fundamentally changes the disconnection logic. By employing a palladium catalyst in conjunction with a silver oxidant, the reaction directly couples the C-H bond of the aryl ring with the aryl iodide, bypassing the need for pre-functionalized organometallic nucleophiles. This direct approach significantly shortens the synthetic timeline and improves the overall efficiency of the process. The reaction conditions are remarkably tolerant of various functional groups, including halogens, esters, and ethers, as evidenced by the successful synthesis of derivatives I-1 through I-11. The use of common organic solvents like xylene or mesitylene further simplifies the operational protocol, making it amenable to standard reactor setups found in most chemical manufacturing facilities. This novel approach not only solves the selectivity issues inherent in electrophilic aromatic substitution but also provides a modular platform for synthesizing complex architectures with high precision and reliability.
Mechanistic Insights into Pd-Catalyzed C-H Acylation
The mechanistic pathway of this transformation is a sophisticated dance of coordination chemistry and redox processes centered around the palladium catalyst. The cycle likely initiates with the coordination of the Pd(II) species to the nitrogen atom of the pyridine or the amide oxygen, acting as a directing group to facilitate proximal C-H bond activation. This concerted metalation-deprotonation (CMD) step generates a cyclopalladated intermediate, which is the key species determining the regioselectivity of the reaction. Subsequently, the aryl iodide undergoes oxidative addition to the Pd(II) center, formally oxidizing it to a Pd(IV) species or proceeding through a Pd(II)/Pd(0) cycle depending on the specific mechanistic manifold. The presence of the silver oxidant is crucial here, as it serves to re-oxidize any Pd(0) formed back to the active Pd(II) state, thereby sustaining the catalytic turnover. This redox neutrality regarding the palladium is essential for maintaining high catalytic efficiency and minimizing the loading of the expensive metal.
Following the oxidative addition, the reaction proceeds through a migratory insertion or reductive elimination step to form the new carbon-carbon bond, releasing the desired diaryl ketone product. The choice of oxidant, such as silver acetate or silver carbonate, plays a dual role: it acts as a terminal oxidant to close the catalytic cycle and may also assist in the removal of iodide ions from the coordination sphere, preventing catalyst poisoning. The solvent choice, typically high-boiling aromatics like xylene, ensures that the reaction temperature can be maintained between 100°C and 150°C, providing the necessary thermal energy to overcome the activation barrier of the C-H cleavage step. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters, such as ligand environment and additive concentration, to maximize yield and minimize the formation of side products like homocoupling byproducts. This deep mechanistic understanding is what separates a viable commercial process from a mere laboratory curiosity.
How to Synthesize 2-Pyridinecarboxamide Diaryl Ketone Efficiently
The practical execution of this synthesis is designed for robustness and scalability, making it an ideal candidate for technology transfer from the lab to the plant. The procedure begins with the precise weighing of the pyridine-2-carboxyl arylamide substrate and the corresponding aryl iodide, ensuring the molar ratios are optimized to drive the reaction to completion while minimizing excess reagent waste. These solids are combined with the palladium catalyst, such as Pd(OAc)2 or PdCl2, and the silver oxidant in a reaction vessel equipped with efficient stirring and heating capabilities. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Combine pyridine-2-carboxyl arylamide, aryl iodide, palladium catalyst, and silver oxidant in an organic solvent such as xylene.
- Heat the reaction mixture to a temperature range of 100°C to 150°C and maintain stirring for 5 to 24 hours until completion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target ketone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike traditional methods that might require custom-synthesized organometallic reagents with short shelf lives, this process relies on stable, commodity-grade aryl iodides and picolinic acid derivatives that are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and price volatility, ensuring a steady flow of materials for continuous manufacturing. Furthermore, the operational simplicity reduces the burden on facility infrastructure, as there is no need for specialized cryogenic equipment or inert gas manifolds beyond standard nitrogen blanketing, which translates to lower capital expenditure and operating costs.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the avoidance of expensive, moisture-sensitive reagents lead to a significant decrease in the overall cost of goods sold (COGS). By removing the need for protection groups and harsh Lewis acids, the process reduces the consumption of auxiliary chemicals and the associated waste disposal fees. The high atom economy of the direct C-H coupling means that a larger proportion of the starting mass ends up in the final product, maximizing resource utilization. Additionally, the use of recyclable solvents like xylene further contributes to cost efficiency, allowing for solvent recovery and reuse in subsequent batches. These cumulative factors result in a leaner, more cost-effective manufacturing process that enhances profit margins without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in pharmaceutical supply chains. The tolerance for functional group diversity means that the same platform technology can be used to produce a wide range of analogues, providing flexibility to respond to changing market demands or clinical trial requirements. Since the reaction does not rely on exotic catalysts or unstable intermediates, the risk of batch failure due to reagent degradation is minimized. This reliability allows for better production planning and inventory management, reducing the need for safety stock and freeing up working capital. Ultimately, a more predictable manufacturing process leads to shorter lead times and improved customer satisfaction.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to tonnage is straightforward due to the absence of exothermic hazards associated with organometallic additions. The reaction can be safely conducted in standard stainless steel reactors, facilitating a smooth transition from pilot plant to commercial production. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical emissions. The use of silver salts, while requiring recovery protocols, is manageable within modern waste treatment frameworks, and the overall E-factor of the process is superior to classical alternatives. This commitment to sustainable manufacturing not only reduces environmental impact but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method utilizes direct C-H activation, eliminating the need for pre-functionalized substrates and reducing the number of synthetic steps, which significantly lowers production costs and waste generation.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process uses robust conditions with common solvents like xylene and stable catalysts, making it highly suitable for commercial scale-up from kilogram to tonnage production.
Q: What is the purity profile of the resulting intermediates?
A: The method yields high-purity products, typically ranging from 70% to 78% isolated yield after standard column chromatography, with well-defined impurity profiles suitable for downstream drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridinecarboxamide Diaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development pipelines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-pyridinecarboxamide diaryl ketone meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that accelerate your path to clinical trials and market approval.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs. Let us be your partner in turning innovative chemistry into commercial reality.
