Advanced Photocatalytic Synthesis of Substituted Quinolines for Commercial Scale Manufacturing
Advanced Photocatalytic Synthesis of Substituted Quinolines for Commercial Scale Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways to construct complex heterocyclic scaffolds, which serve as the backbone for countless bioactive molecules. Patent CN113979937A introduces a groundbreaking methodology for preparing substituted aromatic heterocyclic compounds, specifically targeting the functionalization of quinoline derivatives through a novel photocatalytic system. This technology leverages a composite photocatalyst comprising 3,4,9,10-perylene dianhydride and graphitic carbon nitride (PTCDA/g-C3N4), which exhibits exceptional thermal and photostability compared to traditional organic dyes or metal-based catalysts. By utilizing this advanced catalytic system, the process successfully induces radical addition reactions under mild conditions, specifically in an air or oxygen atmosphere, thereby completely omitting the need for hazardous strong oxidants that have historically plagued this sector. This innovation not only addresses critical environmental concerns but also offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production facilities can operate with reduced maintenance costs and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of electron-deficient aromatic heterocycles, such as quinolines, has relied heavily on the Minisci reaction, a staple transformation in organic synthesis for introducing alkyl groups onto heteroaromatic rings. However, conventional implementations of the Minisci reaction typically necessitate the use of potent stoichiometric oxidants, such as ammonium persulfate ((NH4)2S2O8), to generate the requisite nucleophilic carbon radicals from precursor molecules. The reliance on such aggressive oxidizing agents introduces severe operational challenges, including significant corrosivity to reactor vessels and piping, which drastically shortens the lifespan of expensive manufacturing equipment and increases capital expenditure for replacements. Furthermore, the handling and disposal of large quantities of strong oxidants pose substantial safety risks to personnel and create complex waste streams that require rigorous and costly treatment protocols before discharge. These factors collectively contribute to inflated production costs and extended lead times, creating a bottleneck for the reliable pharmaceutical intermediate supplier who must balance efficiency with regulatory compliance and worker safety in a highly competitive global market.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113979937A presents a paradigm shift by employing a metal-free, heterogeneous photocatalytic system that operates effectively under ambient atmospheric conditions. The core of this innovation lies in the synergistic combination of 3,4,9,10-perylene dianhydride (PTCDA) with g-C3N4, creating a composite material that harnesses visible light energy to drive the formation of radical species without external chemical oxidants. This approach allows the reaction to proceed smoothly in the presence of simple air or oxygen, which acts as the terminal oxidant, thereby eliminating the corrosive impact associated with persulfates and other harsh reagents. The result is a significantly cleaner reaction profile that not only preserves the integrity of the manufacturing infrastructure but also simplifies the downstream purification process by reducing the load of inorganic salt byproducts. For procurement and supply chain leaders, this translates to cost reduction in fine chemical manufacturing through lowered equipment maintenance requirements and streamlined waste management, positioning this technology as a superior choice for modern, green chemistry initiatives.
Mechanistic Insights into PTCDA/g-C3N4 Photocatalytic Radical Addition
The mechanistic elegance of this process stems from the unique electronic properties of the quinoline ring system and the specific activation mode provided by the PTCDA/g-C3N4 composite catalyst. As illustrated in the general structure below, the aromatic heterocyclic compound features a quinoline core where the hydrogen atoms at the 2-position and 4-position are particularly susceptible to radical attack due to the electron-deficient nature of the pyridine ring nitrogen. 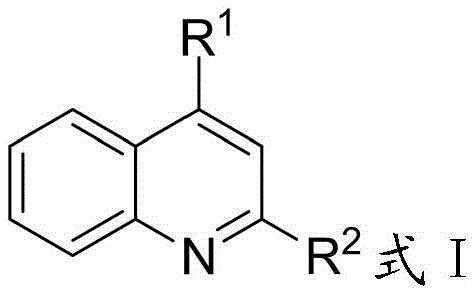 Under acidic conditions, typically facilitated by hydrochloric acid or trifluoroacetic acid, the nitrogen atom of the quinoline ring becomes protonated, further enhancing the electrophilicity of the ring and lowering the energy barrier for the subsequent radical addition step. The photocatalyst, upon irradiation with visible light in the range of 460-520 nm, generates electron-hole pairs that facilitate the single-electron oxidation of the pre-substitution compound, such as tetrahydrofuran or primary alcohols, to form the corresponding carbon-centered radicals. These radicals then add regioselectively to the protonated heterocycle, followed by rearomatization to yield the desired substituted product with high fidelity and minimal formation of regioisomeric impurities.
Under acidic conditions, typically facilitated by hydrochloric acid or trifluoroacetic acid, the nitrogen atom of the quinoline ring becomes protonated, further enhancing the electrophilicity of the ring and lowering the energy barrier for the subsequent radical addition step. The photocatalyst, upon irradiation with visible light in the range of 460-520 nm, generates electron-hole pairs that facilitate the single-electron oxidation of the pre-substitution compound, such as tetrahydrofuran or primary alcohols, to form the corresponding carbon-centered radicals. These radicals then add regioselectively to the protonated heterocycle, followed by rearomatization to yield the desired substituted product with high fidelity and minimal formation of regioisomeric impurities.
From an impurity control perspective, the omission of strong chemical oxidants plays a pivotal role in ensuring the purity of the final high-purity OLED material or pharmaceutical intermediate. Traditional oxidants often lead to over-oxidation side reactions or the degradation of sensitive functional groups present on the substrate, resulting in complex impurity profiles that are difficult to separate. In this photocatalytic system, the mild oxidative potential generated by the excited state of the catalyst is sufficiently selective to activate the specific C-H bonds adjacent to the oxygen atom in ethers or alcohols without attacking other vulnerable sites on the molecule. Furthermore, the use of a heterogeneous catalyst allows for easy separation via filtration, preventing metal contamination that is a common concern with homogeneous transition metal catalysts. This inherent selectivity and cleanliness of the reaction mechanism provide R&D directors with confidence in the reproducibility of the process and the consistency of the quality attributes, which are critical parameters for regulatory filings and long-term commercial viability.
How to Synthesize Substituted Quinoline Derivatives Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to existing laboratory and pilot plant infrastructure, requiring only standard photochemical reactors equipped with LED light sources. The process begins with the precise mixing of the aromatic heterocyclic starting material, the chosen pre-substitution compound (such as tetrahydrofuran for tetrahydrofuran-2-yl substitution), the optimized PTCDA/g-C3N4 photocatalyst, and a protonic acid in a suitable solvent like acetonitrile. Detailed standardized synthetic steps see the guide below.
- Mix the aromatic heterocyclic compound (Formula I), a pre-substitution compound such as tetrahydrofuran or primary alcohol, the PTCDA/g-C3N4 photocatalyst, and an acid like hydrochloric acid in a solvent such as acetonitrile.
- Subject the mixture to visible light irradiation (460-520 nm) in an air or oxygen atmosphere while maintaining the temperature between 25-35°C to induce radical addition.
- Quench the reaction using ammonium chloride, neutralize with sodium bicarbonate, extract with ethyl acetate, and purify via column chromatography to isolate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic and logistical aspects of chemical production, the adoption of this photocatalytic technology offers compelling advantages that directly impact the bottom line and operational resilience. By fundamentally altering the reaction conditions to exclude corrosive strong oxidants, the process dramatically extends the service life of reactors, valves, and containment systems, leading to substantial capital savings over the lifecycle of the production facility. Additionally, the ability to utilize ambient air or simple oxygen feeds as the oxidant source removes the dependency on specialized, hazardous chemical supplies, thereby simplifying inventory management and reducing the risks associated with the storage and transport of dangerous goods. This shift towards safer, milder reagents aligns perfectly with modern ESG (Environmental, Social, and Governance) goals, enhancing the corporate reputation of manufacturers who adopt this greener methodology while simultaneously mitigating regulatory risks associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of stoichiometric strong oxidants such as ammonium persulfate removes a significant cost center associated with both the purchase of reagents and the treatment of the resulting sulfate-rich waste streams. Without the need for extensive neutralization and heavy metal scavenging steps often required in traditional Minisci reactions, the downstream processing becomes markedly simpler, requiring fewer unit operations and less solvent consumption. This streamlining of the workflow results in higher overall throughput and reduced utility costs, as the reaction proceeds efficiently at near-room temperature (25-35°C) without the need for energy-intensive heating or cryogenic cooling. Consequently, the total cost of goods sold (COGS) is optimized, allowing for more competitive pricing strategies in the global marketplace for fine chemicals and intermediates.
- Enhanced Supply Chain Reliability: Relying on air or oxygen as the terminal oxidant decouples the production process from the volatility of the specialty chemical supply chain, ensuring that manufacturing can continue uninterrupted even during shortages of specific oxidizing agents. The robustness of the PTCDA/g-C3N4 catalyst, characterized by its high thermal and photostability, ensures consistent performance over extended periods, reducing the frequency of catalyst replenishment and minimizing batch-to-batch variability. This reliability is crucial for maintaining just-in-time delivery schedules and meeting the stringent demand forecasts of downstream pharmaceutical clients who require uninterrupted supply of critical intermediates. Furthermore, the use of commercially available and inexpensive starting materials like quinolines and common ethers enhances the overall security of the supply chain against raw material price fluctuations.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous oxidants make this process inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton commercial production without significant engineering hurdles. The reduced generation of corrosive and toxic byproducts simplifies compliance with increasingly stringent environmental regulations, lowering the burden on wastewater treatment facilities and reducing the risk of non-compliance penalties. The heterogeneous nature of the catalyst also facilitates its recovery and potential reuse, further minimizing the environmental footprint of the manufacturing process. These factors collectively position this technology as a future-proof solution for the commercial scale-up of complex polymer additives and pharmaceutical intermediates, ensuring long-term sustainability and operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method, derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the PTCDA/g-C3N4 system in terms of selectivity, yield, and operational safety.
Q: What is the primary advantage of using PTCDA/g-C3N4 over traditional oxidants?
A: The primary advantage is the elimination of strong oxidants like ammonium persulfate, which significantly reduces equipment corrosion and simplifies waste treatment protocols while maintaining high reaction yields.
Q: Can this method be applied to various quinoline substrates?
A: Yes, the method is versatile for aromatic heterocyclic compounds defined by Formula I where R1 is Me or Ar and R2 is H, or vice versa, allowing for diverse functionalization at the 2 or 4 positions.
Q: What are the optimal reaction conditions for scalability?
A: The reaction operates efficiently at mild temperatures of 25-35°C under 460-520 nm LED irradiation in an air atmosphere, making it highly suitable for energy-efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in reshaping the landscape of fine chemical synthesis, and we are uniquely positioned to bring these innovations to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of substituted aromatic heterocyclic compounds meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to quality and technical excellence makes us the ideal partner for companies seeking to leverage cutting-edge synthetic methodologies for their supply chains.
We invite you to engage with our technical procurement team to discuss how this novel photocatalytic route can be tailored to your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this oxidant-free protocol for your specific target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and our proven track record of delivering high-quality chemical solutions on time and within budget.
