Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
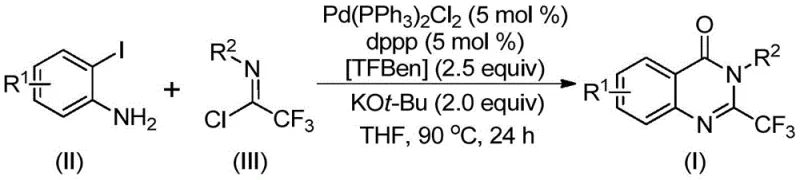
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs which are critical for enhancing metabolic stability and bioavailability in drug candidates. Patent CN112125856A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing significant bottlenecks in current synthetic routes. This technology leverages a transition metal palladium-catalyzed carbonylative tandem reaction, utilizing readily available o-iodoanilines and trifluoroacetimidoyl chlorides as starting materials. A key innovation lies in the substitution of hazardous gaseous carbon monoxide with a solid surrogate, 1,3,5-tricarboxylate phenol ester (TFBen), which dramatically improves operational safety and scalability. For R&D directors and process chemists, this represents a viable pathway to access complex scaffolds found in anticonvulsants, antitumor agents, and hypnotic medications with high efficiency and purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl quinazolinones has been fraught with challenges that hinder commercial viability and process safety. Traditional protocols often rely on the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently require harsh reaction conditions and result in moderate to low yields. Other methods involve the use of unstable trifluoroacetamides or isatoic anhydrides, necessitating pre-activation steps that increase material costs and waste generation. Furthermore, many existing routes depend on the direct use of carbon monoxide gas, a highly toxic and flammable reagent that demands specialized high-pressure equipment and rigorous safety protocols, making scale-up prohibitively expensive and risky for many manufacturing facilities. These limitations collectively restrict the substrate scope and impede the rapid development of new drug candidates requiring this specific pharmacophore.
The Novel Approach
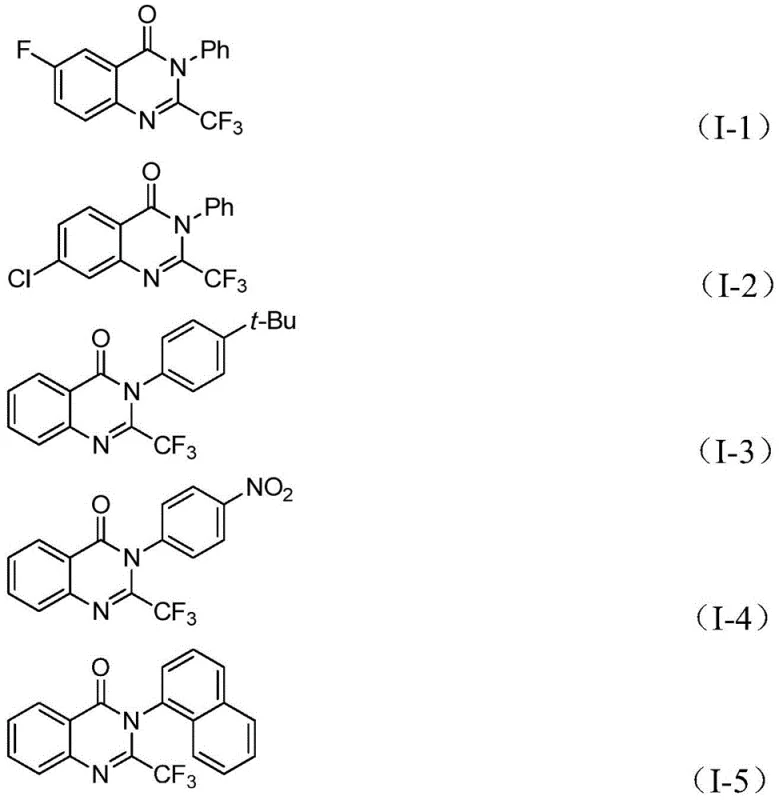
The methodology disclosed in the patent offers a transformative solution by employing a palladium-catalyzed carbonylation strategy that operates under significantly milder conditions. By utilizing trifluoroacetimidoyl chloride and o-iodoaniline derivatives, the process achieves high atom economy and broad functional group tolerance. The replacement of gaseous CO with the solid surrogate TFBen allows the reaction to proceed in standard laboratory glassware or reactors without the need for high-pressure autoclaves, thereby reducing capital expenditure and safety risks. This approach not only simplifies the operational workflow but also enhances the overall yield, with specific embodiments demonstrating conversion rates as high as 99%. The ability to tolerate diverse substituents such as halogens, alkyl groups, and nitro groups on the aromatic rings ensures that this method is versatile enough for the synthesis of a wide array of pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism involves a sophisticated sequence of organometallic transformations initiated by the base-promoted coupling of the starting materials. Initially, potassium tert-butoxide facilitates an intermolecular carbon-nitrogen bond coupling between the o-iodoaniline and the trifluoroacetimidoyl chloride to generate a trifluoroacetamidine intermediate in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium species. Under the thermal conditions of 90°C, the solid CO surrogate TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This step is crucial as it introduces the carbonyl functionality required for the quinazolinone core without handling gas cylinders.
Following the CO insertion, the base promotes the formation of a palladium-nitrogen bond, leading to the construction of a seven-membered palladacycle intermediate. The final step involves a reductive elimination that releases the desired 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst for the next cycle. This mechanistic pathway is highly efficient because it minimizes side reactions often associated with harsh acidic or basic conditions found in older methods. The mild nature of the catalytic cycle ensures that sensitive functional groups on the substrate remain intact, resulting in a cleaner crude reaction profile and simplifying downstream purification processes, which is a critical factor for maintaining high purity standards in API manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a standardized procedure that balances reaction efficiency with operational simplicity, making it ideal for both laboratory optimization and pilot plant production. The process utilizes a specific ratio of catalyst to ligand to ensure maximum turnover numbers while minimizing precious metal usage. Detailed below is the strategic framework for executing this transformation, focusing on the critical parameters that drive high yield and reproducibility. For a comprehensive, step-by-step technical guide including exact quantities and workup procedures, please refer to the standardized synthesis instructions provided in our technical documentation.
- Combine palladium catalyst (Pd(PPh3)2Cl2), ligand (dppp), base (KOt-Bu), solid CO source (TFBen), trifluoroacetimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and stir for 16 to 30 hours to allow the carbonylation and cyclization to proceed.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers substantial strategic advantages over legacy technologies. The shift away from hazardous gaseous reagents and expensive activating agents translates directly into reduced operational overhead and lower insurance costs associated with chemical storage and handling. By utilizing commercially available and inexpensive starting materials like o-iodoanilines and trifluoroacetimidoyl chlorides, manufacturers can secure a more stable and cost-effective supply chain, mitigating the risks associated with specialty reagent shortages. Furthermore, the simplified reaction setup reduces the complexity of the manufacturing infrastructure required, allowing for faster technology transfer between sites and more flexible production scheduling.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the need for specialized high-pressure reactors and extensive safety monitoring systems, leading to significant capital and operational expenditure savings. Additionally, the use of a solid CO surrogate and widely available palladium catalysts optimizes reagent costs, while the high yields observed across various substrates minimize raw material waste and maximize output per batch. This efficiency drives down the cost per kilogram of the final intermediate, providing a competitive edge in the pricing of complex pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents rather than pressurized gases or highly moisture-sensitive anhydrides ensures a more robust supply chain. Starting materials such as substituted o-iodoanilines are commodity chemicals with multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment maintenance or safety shutdowns, ensuring consistent delivery timelines for downstream API synthesis.
- Scalability and Environmental Compliance: The use of a solid carbon monoxide source significantly lowers the environmental footprint of the process by reducing the risk of fugitive emissions and simplifying waste treatment protocols. The reaction generates fewer byproducts compared to traditional condensation methods, facilitating easier purification and reducing the volume of solvent waste. This aligns with modern green chemistry principles and regulatory requirements, making the scale-up from grams to tons more straightforward and environmentally compliant without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method, focusing on safety, substrate compatibility, and process parameters. These insights are derived directly from the experimental data and embodiments described in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances is essential for R&D teams planning to integrate this technology into their existing pipelines for heterocycle synthesis.
Q: How does this method improve safety compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, eliminating the need for handling toxic and flammable carbon monoxide gas directly.
Q: What is the substrate scope for this synthesis?
A: The method demonstrates excellent compatibility with various substituents on both the o-iodoaniline and the imidoyl chloride, including halogens, alkyl groups, and nitro groups.
Q: What are the typical reaction conditions?
A: The reaction typically proceeds in THF at 90°C for 16 to 30 hours using a palladium catalyst system with potassium tert-butoxide as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, helping you optimize your budget without compromising on quality. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain resilience and product performance.
