Advanced Copper-Catalyzed Synthesis of 3-Morpholinyl-4-Arylseleno Maleimides for Commercial Scale-Up
Introduction to Novel Maleimide Synthetic Technology
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN111978237A introduces a groundbreaking preparation method for 3-morpholinyl-4-arylseleno maleimide compounds, addressing critical bottlenecks in current synthetic routes. These 3,4-difunctionalized maleimides serve as privileged structures in medicinal chemistry, acting as core skeletons for marine natural alkaloids and potent antitumor agents such as Isogranulatimide and Himanimide A. The ability to access these motifs through a direct, multi-component coupling strategy represents a significant leap forward in process chemistry.
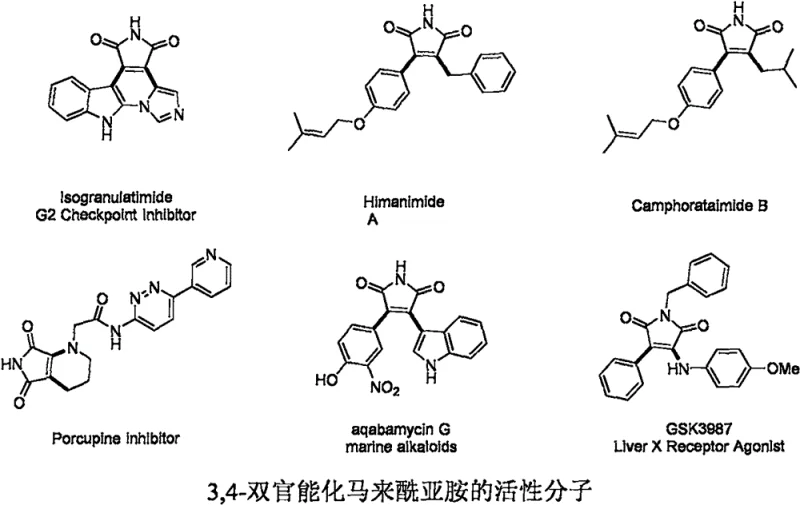
This technology leverages a transition metal copper catalyst system to drive a four-component tandem reaction involving arylboronic acids, elemental selenium powder, morpholine, and N-methylmaleimide. Unlike traditional approaches that rely on pre-functionalized reagents, this method utilizes cheap and readily available starting materials under oxidative conditions. For R&D directors and process chemists, this offers a streamlined pathway to generate diverse libraries of bioactive candidates, accelerating the drug discovery timeline while maintaining high standards of purity and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of arylselenyl groups into maleimide frameworks has been fraught with challenges regarding reagent availability and catalyst cost. Previous methodologies, such as those reported by the Baidya group, often relied on transition metal ruthenium catalysts to mediate the oxidoselenylation of maleimides with diaryl diselenides. While effective in academic settings, the requirement for precious metal ruthenium imposes a substantial financial burden on large-scale manufacturing operations. Furthermore, the necessity to pre-synthesize diaryl diselenides adds extra synthetic steps, increasing waste generation and extending lead times.
Additionally, the use of expensive noble metals complicates the purification process, as residual metal removal becomes a critical quality control hurdle for pharmaceutical intermediates. The reliance on specialized reagents also limits the structural diversity accessible to medicinal chemists, as the scope of available diselenides is narrower than that of arylboronic acids. These factors collectively hinder the efficient application of such reactions in the late-stage modification of drug molecules, thereby reducing the opportunities for discovering innovative therapeutic agents with optimized pharmacokinetic profiles.
The Novel Approach
The methodology disclosed in CN111978237A circumvents these obstacles by employing a cost-effective copper catalytic system that operates under an oxygen atmosphere. This novel approach enables the direct use of elemental selenium powder, a commodity chemical, rather than specialized diselenide reagents. By integrating arylboronic acids as the aryl source, the reaction taps into a vast catalog of commercially available building blocks, vastly expanding the chemical space accessible for derivatization. The synergistic action of the copper catalyst, silver salt, ligand, and base facilitates the simultaneous formation of carbon-nitrogen and carbon-selenium bonds in a single operational step.
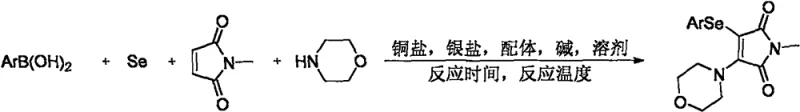
This four-component cross-coupling strategy not only simplifies the synthetic route but also enhances atom economy by minimizing auxiliary reagents. The reaction conditions are remarkably robust, utilizing standard organic solvents like DMF and moderate heating, which are easily scalable in industrial reactors. For procurement managers, this shift from precious metals to base metals like copper translates directly into reduced raw material costs and simplified supply chain logistics, making the production of high-value maleimide derivatives more economically viable.
Mechanistic Insights into Copper-Catalyzed Four-Component Cross-Coupling
The success of this transformation hinges on the precise orchestration of a multi-component catalytic cycle involving copper and silver species. The reaction initiates with the activation of elemental selenium by the copper catalyst, likely forming a reactive copper-selenium intermediate. Simultaneously, the arylboronic acid undergoes transmetallation or oxidative addition processes facilitated by the silver salt oxidant, which regenerates the active copper species and drives the catalytic turnover. The presence of molecular oxygen serves as the terminal oxidant, ensuring the continuous cycling of the metal centers without the accumulation of reduced byproducts that could stall the reaction.
The ligand, specifically 1,10-phenanthroline, plays a pivotal role in stabilizing the copper center and modulating its electronic properties to favor the desired cross-coupling pathway over competing homocoupling reactions. Experimental optimization revealed that substituting this ligand with phosphines like triphenylphosphine resulted in complete reaction failure, underscoring the unique steric and electronic environment provided by the phenanthroline scaffold. Similarly, the choice of base is critical; cesium carbonate was found to be superior to potassium or sodium carbonates, likely due to its higher solubility in organic media and stronger basicity which aids in the activation of the nucleophilic amine component.
Impurity control is inherently built into this mechanism through the high selectivity of the catalyst system. The specific combination of CuI, Ag2CO3, and Cs2CO3 suppresses side reactions such as the homocoupling of boronic acids or the oxidation of selenium to selenoxides. This selectivity ensures that the crude reaction mixture contains a high proportion of the target 3-morpholinyl-4-arylseleno maleimide, simplifying downstream purification. For quality assurance teams, this means fewer chromatographic steps are required to meet stringent purity specifications, reducing solvent consumption and processing time while maximizing overall yield.
How to Synthesize 3-Morpholinyl-4-Arylseleno Maleimide Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters identified in the patent to ensure reproducibility and high yield. The process involves charging a reaction vessel with the four key components—arylboronic acid, selenium powder, morpholine, and N-methylmaleimide—along with the catalytic system in anhydrous DMF. It is crucial to maintain an oxygen-rich environment throughout the reaction duration to sustain the oxidative catalytic cycle. The detailed standardized synthesis steps, including exact molar ratios and workup procedures, are outlined below for technical reference.
- Combine arylboronic acid, selenium powder, morpholine, and N-methylmaleimide in DMF solvent with CuI catalyst, Phen ligand, Ag2CO3, and Cs2CO3 base.
- Purge the reaction vessel with oxygen gas three times to ensure an oxidative environment conducive to the coupling reaction.
- Heat the mixture to 140°C and stir for 36 hours, followed by extraction, drying, and column chromatography purification to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that align with the strategic goals of cost reduction and supply chain resilience. By replacing expensive ruthenium catalysts and pre-synthesized diselenides with commodity copper salts and elemental selenium, the direct material costs are significantly lowered. This shift not only improves the gross margin on the final product but also mitigates the risk associated with the price volatility of precious metals. Furthermore, the use of widely available arylboronic acids ensures a stable and diversified supply base, reducing dependency on niche reagent suppliers.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for expensive metal scavenging resins and complex purification protocols typically required to meet residual metal limits in APIs. Additionally, the use of elemental selenium powder avoids the multi-step synthesis of diaryl diselenides, effectively cutting out an entire upstream production stage. This streamlining of the process flow leads to substantial savings in labor, energy, and equipment usage, making the manufacturing of these complex intermediates much more cost-efficient.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of common chemical feedstocks like arylboronic acids and morpholine, which are produced at massive scales globally. This abundance ensures consistent availability and short lead times, preventing production delays caused by raw material shortages. The robustness of the reaction conditions also allows for flexibility in sourcing, as the process is less sensitive to minor variations in reagent quality compared to more fragile noble metal-catalyzed systems.
- Scalability and Environmental Compliance: The reaction operates in standard polar aprotic solvents like DMF, which are well-understood in industrial settings and can be efficiently recovered and recycled. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying waste treatment and disposal procedures. This environmental compatibility facilitates easier regulatory approval for commercial scale-up, allowing manufacturers to increase production volumes from pilot plants to multi-ton facilities with confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on catalyst selection, substrate tolerance, and operational parameters. Understanding these nuances is essential for process engineers planning to adopt this technology for commercial production.
Q: Why is Cuprous Iodide (CuI) preferred over other copper salts in this synthesis?
A: Experimental data indicates that other copper salts such as copper acetate, cuprous chloride, and copper triflate result in no reaction. CuI, in synergy with the specific ligand and base system, is uniquely effective for activating the selenium powder and facilitating the cross-coupling.
Q: What are the advantages of using elemental selenium powder over diaryl diselenides?
A: Using elemental selenium powder eliminates the need for the pre-synthesis of expensive and potentially unstable diaryl diselenides. This direct utilization of cheap, stable selenium powder significantly simplifies the raw material supply chain and reduces overall production costs.
Q: Can this method tolerate various functional groups on the arylboronic acid?
A: Yes, the method demonstrates good tolerance for electron-donating groups like methyl and methoxy, yielding high conversion rates. However, strong electron-withdrawing groups like trifluoromethyl may result in lower yields, requiring further optimization for specific substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Morpholinyl-4-Arylseleno Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis in advancing drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-morpholinyl-4-arylseleno maleimide delivered meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore how this cost-effective methodology can be integrated into your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain objectives, ensuring a reliable partnership for your complex chemical synthesis needs.
