Advanced Copper-Catalyzed Synthesis of 3-Morpholinyl-4-Arylseleno Maleimide Derivatives for Pharmaceutical Applications
Introduction to Next-Generation Maleimide Synthesis
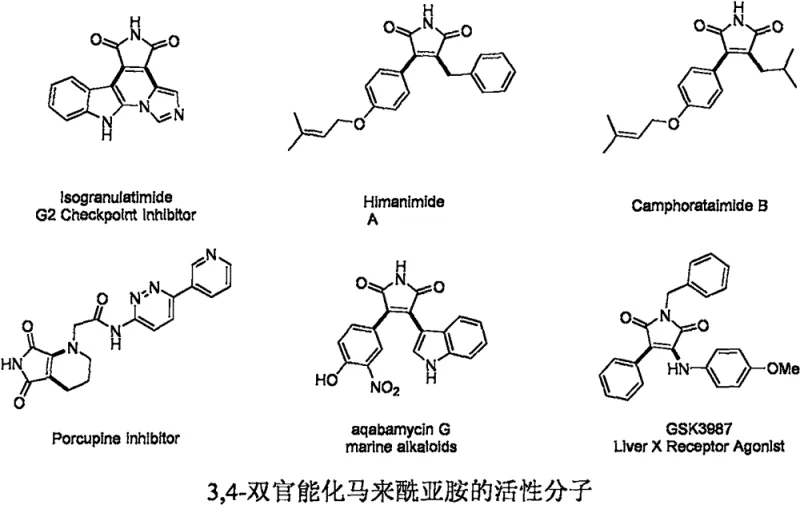
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient routes to complex heterocyclic scaffolds. Patent CN111978237B introduces a groundbreaking preparation method for 3-morpholinyl-4-arylseleno maleimide compounds, a class of molecules with profound significance in medicinal chemistry. These bifunctional maleimides serve as core scaffolds for a wide array of bioactive natural products and synthetic drug candidates, including potent G2 cell cycle checkpoint kinase inhibitors like Isogranulatimide and anti-breast cancer agents such as Camphoratamide B. The ability to construct these dense molecular architectures efficiently is paramount for accelerating drug discovery pipelines.
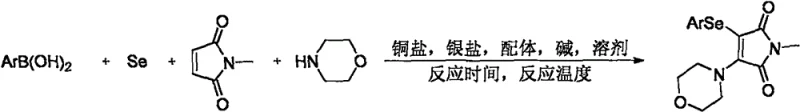
This patented technology addresses a critical gap in current synthetic methodologies by utilizing a transition metal copper-catalyzed four-component cross-coupling reaction. Unlike traditional multi-step sequences that often suffer from low atom economy and cumbersome purification processes, this novel approach leverages the synergistic catalysis of copper salts, silver additives, and specific ligands to achieve direct functionalization. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse chemical space for library synthesis and lead optimization without the burden of complex protecting group strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of arylseleno-substituted maleimides has relied heavily on less efficient strategies that hinder scalability and cost-effectiveness. Previous literature, such as reports involving transition metal ruthenium catalysis, necessitates the oxidative selenylation of maleimides using diaryl diselenides. This conventional pathway presents two major bottlenecks: firstly, the prerequisite synthesis of diaryl diselenides adds an extra synthetic step, increasing both time and material costs; secondly, the reliance on noble metal ruthenium catalysts introduces significant expense and potential heavy metal contamination issues that are difficult to mitigate in GMP environments. Furthermore, these older methods often lack the versatility required for late-stage functionalization of complex drug molecules, limiting their utility in modern medicinal chemistry campaigns.
The Novel Approach
The methodology disclosed in CN111978237B fundamentally disrupts this status quo by enabling a direct, one-pot assembly of the target scaffold from inexpensive and readily available starting materials. By employing elemental selenium powder directly as the selenylation reagent alongside arylboronic acids, the process bypasses the need for pre-functionalized diselenide intermediates entirely. This four-component tandem reaction operates under the co-concerted catalytic action of a copper catalyst, a silver salt, a ligand, and a base, facilitating the simultaneous formation of carbon-nitrogen and carbon-selenium bonds. This streamlined approach not only simplifies the operational workflow but also drastically improves the overall atom economy of the transformation.
For procurement managers and supply chain heads, this shift translates to a more robust and resilient supply chain. The reliance on commodity chemicals like selenium powder and arylboronic acids, rather than specialized, high-cost organoselenium reagents, ensures better price stability and availability. The reaction conditions, while requiring elevated temperatures, utilize standard laboratory equipment and solvents like DMF, making the transition from bench-scale discovery to commercial scale-up significantly smoother and less capital-intensive compared to processes requiring exotic catalysts or cryogenic conditions.
Mechanistic Insights into Copper-Catalyzed Four-Component Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to adapt this chemistry for specific analog synthesis. The reaction proceeds through a complex catalytic cycle where cuprous iodide acts as the primary transition metal center, activating the selenium species and facilitating the transmetallation with the arylboronic acid. The presence of silver carbonate is not merely incidental; it serves a dual role as both an oxidant to regenerate the active copper species and a promoter for the selenylation step. The ligand, specifically 1,10-phenanthroline, plays a pivotal role in stabilizing the copper center and modulating its electronic properties to favor the desired cross-coupling over homocoupling side reactions.
Impurity control is a key advantage of this specific catalytic system. Comparative data within the patent indicates that substituting the ligand with triphenylphosphine or changing the copper source to copper acetate results in a complete failure of the reaction. This high degree of specificity suggests that the catalytic pocket formed by the CuI-Phen complex is uniquely tuned to activate the relatively inert selenium powder and coordinate the morpholine nucleophile effectively. Consequently, the formation of byproducts such as homocoupled biaryls or over-selenylated species is minimized, leading to crude reaction profiles that are much cleaner and easier to purify via standard column chromatography.
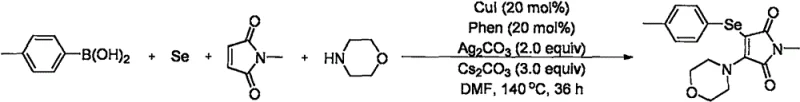
Furthermore, the use of cesium carbonate as the base is critical for maintaining the reaction pH and facilitating the deprotonation steps necessary for the catalytic cycle to turnover. The patent data highlights that weaker bases like sodium carbonate or potassium phosphate fail to drive the reaction to completion, underscoring the delicate balance of basicity required. For process chemists, this mechanistic clarity allows for rational troubleshooting and optimization, ensuring that the high yields reported in the examples—such as the 87% yield for the p-tolyl derivative—can be consistently reproduced on larger scales.
How to Synthesize 3-Morpholinyl-4-Arylseleno Maleimide Efficiently
To implement this synthesis in a laboratory or pilot plant setting, strict adherence to the optimized parameters is essential. The process involves charging a reaction vessel with the four key components: arylboronic acid, selenium powder, morpholine, and N-methylmaleimide, dissolved in anhydrous DMF. The atmosphere must be carefully controlled, typically by replacing the headspace with oxygen three times to ensure the necessary oxidative environment for the catalytic cycle. Following the addition of the catalytic cocktail (CuI, Phen, Ag2CO3, Cs2CO3), the mixture is heated to 140°C for a prolonged period, typically around 36 hours, to ensure full conversion.
- Combine arylboronic acid, selenium powder, morpholine, and N-methylmaleimide in an organic solvent such as DMF under an oxygen atmosphere.
- Add the catalytic system consisting of cuprous iodide, silver carbonate, 1,10-phenanthroline, and cesium carbonate to the reaction mixture.
- Heat the reaction mixture to 140°C for approximately 36 hours, then perform extraction and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that directly impact the bottom line and operational efficiency. The primary driver for cost reduction lies in the raw material selection; replacing expensive noble metal catalysts and pre-synthesized organoselenium reagents with base metal copper and elemental selenium powder results in substantial savings on bill of materials. Additionally, the elimination of intermediate isolation steps reduces solvent consumption and labor hours associated with multi-step processing, further driving down the cost of goods sold (COGS) for these valuable intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the strategic substitution of high-cost inputs with commodity chemicals. By utilizing copper iodide instead of ruthenium-based catalysts, manufacturers avoid the volatility associated with precious metal pricing. Moreover, the direct use of selenium powder removes the need for a separate synthesis step for diaryl diselenides, effectively cutting the production timeline and associated utility costs in half. This streamlined material flow ensures that the final API intermediate is produced with maximum economic efficiency.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials. Arylboronic acids and morpholine are bulk chemicals with established global supply chains, reducing the risk of shortages that often plague specialized reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, allowing for greater flexibility in vendor selection. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The scalability of this four-component coupling is supported by its operation in standard polar aprotic solvents like DMF, which are well-understood in large-scale chemical engineering. The simplified workup procedure, involving basic extraction and crystallization or chromatography, minimizes the generation of complex waste streams. While DMF recovery is necessary, the absence of heavy noble metal residues simplifies wastewater treatment protocols, aiding in compliance with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on critical process parameters and substrate compatibility.
Q: Why is cuprous iodide preferred over other copper salts in this synthesis?
A: According to the patent data, comparative experiments demonstrated that other copper salts like copper acetate or cuprous chloride resulted in no reaction. Cuprous iodide acts as the critical transition metal catalyst that enables the synergistic activation of the four components.
Q: What are the advantages of using selenium powder over diaryl diselenides?
A: Using elemental selenium powder eliminates the need for the pre-synthesis of diaryl diselenides, which simplifies the workflow and reduces raw material costs. It allows for a direct, one-step introduction of the arylseleno group.
Q: What represents the optimal reaction conditions for high yield?
A: The optimal conditions involve using DMF as the solvent, cesium carbonate as the base, and silver carbonate as the oxidant/additive at a temperature of 140°C for 36 hours under an oxygen atmosphere.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Morpholinyl-4-Arylseleno Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis for the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-morpholinyl-4-arylseleno maleimide delivered meets the highest industry standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain and reduce development risks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical landscape.
