Revolutionizing Alpha-Amidoketone Production: A Catalyst-Free Thermal Approach for Commercial Scale-Up
Introduction to Advanced Alpha-Amidoketone Synthesis
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical industry, particularly regarding purity profiles and process efficiency. A significant breakthrough in this domain is documented in Chinese Patent CN111960961B, which discloses a novel synthetic method for alpha-amidoketone compounds. These nitrogen-containing carbonyl compounds are pivotal intermediates in the construction of various natural products, medicines, and organic functional materials due to their unique reactivity and biological activity. The core innovation lies in the direct thermal reaction between alkenyl azide compounds and carboxylic acid compounds, bypassing the need for expensive and potentially toxic transition metal catalysts. This approach not only simplifies the operational workflow but also aligns perfectly with the principles of green chemistry by enhancing atom economy and reducing waste generation.
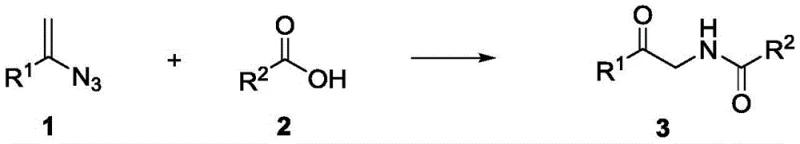
For R&D directors and process chemists, the implications of this technology are profound. Traditional routes often suffer from limitations such as harsh reaction conditions, narrow substrate tolerance, or the necessity for complex catalytic systems that require stringent removal steps to meet ICH Q3D guidelines on elemental impurities. By contrast, the method outlined in CN111960961B utilizes simple heating in common organic solvents to drive the transformation. This thermal activation strategy ensures that the resulting alpha-amidoketones are free from metal residues, thereby significantly lowering the burden on downstream purification processes. Furthermore, the broad applicability of this method allows for the modification of complex drug molecules, opening new avenues for medicinal chemistry campaigns aimed at optimizing pharmacokinetic properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amidoketones has relied heavily on transition metal catalysis or multi-step sequences involving unstable intermediates. Conventional protocols frequently employ rhodium, copper, or ruthenium complexes to facilitate nitrene transfer or C-H insertion reactions. While effective in laboratory settings, these methods pose substantial challenges when translated to commercial manufacturing. The primary concern is the residual metal content, which necessitates additional purification stages such as scavenging or recrystallization, inevitably leading to yield losses and increased production costs. Moreover, many traditional methods exhibit poor atom economy, generating stoichiometric amounts of byproducts that complicate waste management. The reliance on sensitive catalysts also introduces variability in reaction performance, making scale-up unpredictable and often requiring specialized equipment to handle air- or moisture-sensitive reagents.
The Novel Approach
The methodology presented in patent CN111960961B represents a paradigm shift by eliminating the catalyst entirely. The process involves merely mixing an alkenyl azide compound with a carboxylic acid compound in a solvent such as 1,2-dichloroethane (DCE) and heating the mixture to temperatures between 80°C and 120°C. This catalyst-free thermal pathway leverages the intrinsic reactivity of the azide group upon heating, likely generating a reactive nitrene species in situ that undergoes insertion or addition reactions with the carboxylic acid. The operational simplicity is unmatched; there is no need for inert atmosphere gloveboxes or rigorous drying of solvents beyond standard laboratory practices. Experimental data from the patent indicates that optimal yields, such as 76% for the model reaction producing compound 3a, are achieved at 110°C over 10 hours. This robustness makes the process highly attractive for industrial application where reliability and ease of execution are paramount.
Mechanistic Insights into Thermal Azide-Acid Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and predicting substrate compatibility. Although the patent does not explicitly detail a catalytic cycle, the reaction is postulated to proceed through a thermal decomposition mechanism. Upon heating, the alkenyl azide undergoes denitrogenation to form a reactive vinyl nitrene or azirine intermediate. This highly electrophilic species is then intercepted by the nucleophilic oxygen of the carboxylic acid or undergoes a rearrangement followed by nucleophilic attack. The absence of external catalysts suggests that the activation energy barrier is overcome solely by thermal input, which is facilitated by the choice of high-boiling solvents like DCE. This mechanism avoids the formation of metal-ligand complexes that can sometimes lead to side reactions or catalyst deactivation, ensuring a cleaner reaction profile.
From an impurity control perspective, this mechanism offers distinct advantages. Since no metal salts are introduced, the impurity spectrum is limited primarily to organic byproducts derived from substrate decomposition or incomplete conversion, which are generally easier to separate via standard chromatography or crystallization. The patent highlights the successful synthesis of a diverse array of derivatives, including those with electron-withdrawing and electron-donating groups on the aromatic rings, indicating that the electronic nature of the substrates does not drastically inhibit the thermal pathway. Furthermore, the tolerance for sterically hindered substrates, such as adamantane-carboxylic acid derivatives, suggests that the reactive intermediate is sufficiently small or flexible to access crowded reaction centers. This mechanistic robustness ensures consistent quality across different batches, a critical factor for supply chain stability.
How to Synthesize Alpha-Amidoketone Efficiently
To implement this synthesis effectively, one must adhere to the optimized conditions identified in the patent examples. The procedure typically involves charging a reaction vessel with the alkenyl azide and the carboxylic acid in a molar ratio of approximately 1.2:1 to 1.5:1, favoring the azide to drive the reaction to completion. The mixture is dissolved in 1,2-dichloroethane (DCE), which has been identified as the superior solvent compared to alternatives like methanol, toluene, or acetonitrile, providing yields up to 76% under optimal conditions. The reaction is then heated to 110°C and stirred for 10 hours. Post-reaction workup is straightforward, involving solvent removal followed by silica gel column chromatography. For a detailed, step-by-step standard operating procedure tailored to your specific facility capabilities, please refer to the guide below.
- Mix alkenyl azide compound and carboxylic acid compound in an organic solvent such as 1,2-dichloroethane.
- Heat the reaction mixture to a temperature between 80°C and 120°C, preferably 110°C, and stir for approximately 10 hours.
- Upon completion, cool the mixture, remove the solvent, and purify the resulting alpha-amidoketone via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of precious metal catalysts directly impacts the cost structure of the manufacturing process. Transition metals like rhodium or palladium are subject to volatile market prices and supply constraints; removing them from the bill of materials stabilizes costs and reduces exposure to geopolitical supply risks. Additionally, the simplified purification process means less solvent consumption and reduced waste disposal fees, contributing to a lower overall cost of goods sold (COGS). The use of commodity chemicals like DCE and simple heating equipment further enhances the economic viability, making this route suitable for both pilot-scale development and large-scale commercial production without requiring capital-intensive reactor modifications.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts. In traditional synthesis, the cost of the catalyst itself, combined with the expensive ligands often required, can constitute a substantial portion of the raw material budget. More importantly, the downstream processing costs associated with removing trace metals to meet pharmaceutical specifications are considerable. By adopting this thermal method, manufacturers eliminate the need for metal scavengers, specialized filtration media, and the associated analytical testing for residual metals. This streamlining of the purification train results in significant cost savings and improved throughput, allowing for more competitive pricing of the final alpha-amidoketone intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials. Alkenyl azides and carboxylic acids are commodity chemicals with established global supply chains, unlike specialized organometallic complexes which may have single-source suppliers. The robustness of the reaction conditions—tolerating temperatures up to 120°C and functioning in common solvents—means that production is less susceptible to disruptions caused by the unavailability of exotic reagents or sensitive equipment. This reliability ensures consistent delivery schedules for downstream customers, reducing the risk of production stoppages in their own API manufacturing lines. The ability to source raw materials from multiple vendors further mitigates supply risk.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this thermal method is inherently scalable due to its simplicity. The reaction does not generate hazardous gaseous byproducts in significant quantities beyond nitrogen gas, which is benign. The high atom economy mentioned in the patent implies that a greater proportion of the reactant mass is incorporated into the final product, minimizing waste generation. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. Reduced waste volumes lower the burden on wastewater treatment facilities and decrease the carbon footprint of the manufacturing process. Consequently, facilities can operate with greater environmental compliance and lower regulatory overhead, facilitating smoother audits and approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111960961B, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new drug development pipelines.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method described in CN111960961B operates entirely without metal catalysts, utilizing thermal activation instead. This eliminates the risk of heavy metal contamination in the final product.
Q: What is the substrate scope for the carboxylic acid component?
A: The method demonstrates excellent compatibility with a wide range of carboxylic acids, including complex drug molecules like ibuprofen, indomethacin, and dehydrocholic acid, allowing for late-stage functionalization.
Q: Can the resulting alpha-amidoketones be further derivatized?
A: Yes, the synthesized alpha-amidoketones serve as versatile intermediates that can be cyclized into oxazoles using sulfuric acid and acetic anhydride, or functionalized into alpha-phenoxy derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amidoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free thermal synthesis described in CN111960961B for the production of high-purity alpha-amidoketones. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative chemistry are realized at an industrial level. Our state-of-the-art facilities are equipped to handle thermal reactions safely and efficiently, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications. We are committed to delivering intermediates that facilitate your drug development success without the baggage of metal contamination.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalyst-free method for your target molecules. Please contact our technical procurement team today to request specific COA data for our alpha-amidoketone portfolio and to discuss route feasibility assessments tailored to your timeline. Let us help you optimize your supply chain and accelerate your path to market with reliable, high-quality chemical solutions.
