Advanced Catalyst-Free Synthesis of Alpha-Amidoketones for Scalable Pharmaceutical Manufacturing
Introduction to Breakthrough Synthetic Technology
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, where efficiency and purity are paramount. A significant advancement in this field is documented in Chinese patent CN111960961A, which discloses a novel and highly efficient method for synthesizing alpha-amidoketone compounds. These nitrogen-containing carbonyl structures are not merely academic curiosities; they represent a critical class of intermediates found in numerous bioactive molecules, natural products, and functional materials. The core innovation lies in a streamlined tandem reaction between alkenyl azide compounds and carboxylic acid compounds. Unlike traditional methodologies that often rely on complex catalytic systems, this approach achieves high yields through a simple thermal process in an organic solvent. This technological leap offers a compelling value proposition for industry stakeholders seeking reliable pharmaceutical intermediate supplier partnerships that prioritize both chemical elegance and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alpha-amidoketone scaffold has been fraught with synthetic challenges that hinder large-scale adoption. Conventional literature methods frequently necessitate the use of transition metal catalysts, which introduce significant downstream processing burdens. For a procurement manager focused on cost reduction in API manufacturing, the requirement for precious metals translates directly into higher raw material costs and the need for expensive scavenging resins to meet strict residual metal specifications. Furthermore, many existing protocols suffer from poor atom economy, generating substantial chemical waste that complicates environmental compliance. The substrate scope in older methods is often narrow, failing to tolerate sensitive functional groups or sterically hindered structures, which limits their utility in late-stage functionalization of complex drug candidates. These inefficiencies create bottlenecks in the supply chain, extending lead times and increasing the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111960961A represents a paradigm shift towards green and sustainable chemistry. By eliminating the need for any external catalyst, the process simplifies the reaction setup to a mere mixture of reactants and solvent followed by heating. This catalyst-free nature ensures that the final product is free from metal contaminants, a critical quality attribute for pharmaceutical intermediates intended for human consumption. The reaction conditions are remarkably mild, typically operating between 80°C and 120°C, which reduces energy consumption compared to high-temperature pyrolysis methods. Moreover, the protocol demonstrates exceptional functional group tolerance, successfully accommodating a wide array of substituents including halogens, esters, and ketones without the need for protecting groups. This robustness allows chemists to utilize readily available starting materials, thereby enhancing supply chain reliability and reducing the dependency on specialized reagents.
Mechanistic Insights into Catalyst-Free Thermal Tandem Reaction
From a mechanistic perspective, this transformation is a fascinating example of a thermal tandem reaction that maximizes atom economy. The process initiates with the interaction between the alkenyl azide and the carboxylic acid under thermal activation. While the precise mechanistic pathway may involve the generation of reactive nitrene species or direct nucleophilic attack depending on the specific electronic properties of the substrates, the net result is the efficient formation of the carbon-nitrogen and carbon-carbon bonds required for the alpha-amidoketone structure. The absence of a catalyst suggests that the reaction is driven by the inherent reactivity of the azide functionality and the acidity of the carboxylic proton, facilitated by the polar environment of the solvent. This intrinsic reactivity minimizes the formation of side products associated with catalyst decomposition or ligand exchange, leading to cleaner reaction profiles. For R&D directors, this implies a simplified purification process, often requiring only standard silica gel chromatography to achieve high purity standards, thus accelerating the timeline from bench scale to pilot production.
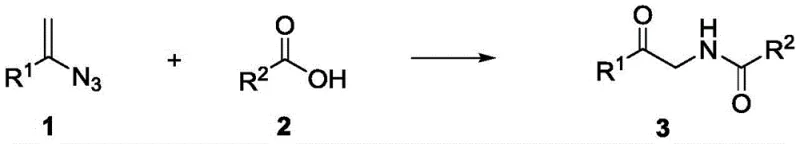
The versatility of this synthetic route is further evidenced by its compatibility with structurally complex molecules, making it an ideal tool for medicinal chemistry campaigns. The patent data highlights the successful incorporation of sophisticated carboxylic acid derivatives, including steroid backbones and non-steroidal anti-inflammatory drug (NSAID) scaffolds. This capability is crucial for the commercial scale-up of complex pharmaceutical intermediates, as it allows for the direct modification of advanced intermediates without degrading sensitive moieties. Additionally, the resulting alpha-amidoketones serve as powerful platforms for further diversification. As demonstrated in the patent, these intermediates can be seamlessly converted into oxazole derivatives through acid-catalyzed cyclization or functionalized at the alpha-position via oxidation. This modularity enhances the value of the initial synthesis, providing a single entry point to multiple distinct chemical libraries, which is a significant strategic advantage in drug discovery pipelines.
How to Synthesize Alpha-Amidoketone Efficiently
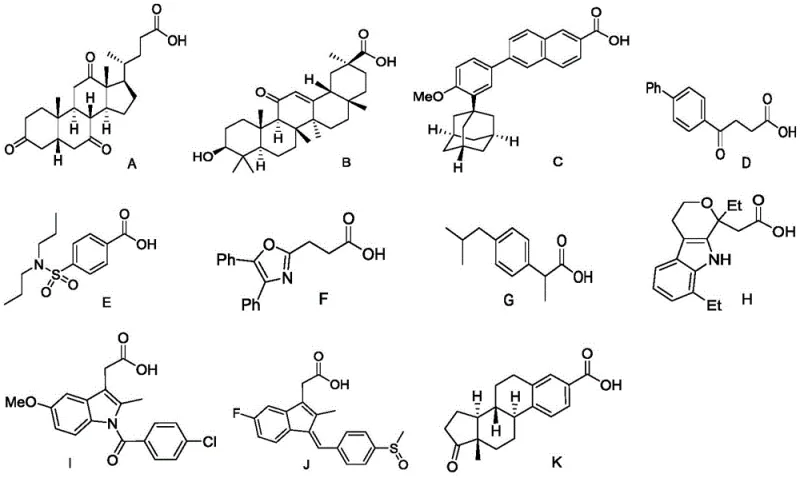
Implementing this synthesis in a laboratory or production setting requires adherence to specific optimized parameters to ensure maximum yield and reproducibility. The patent identifies 1,2-dichloroethane (DCE) as the preferred solvent, offering an optimal balance of solubility and boiling point to facilitate the reaction at 110°C. The stoichiometry is also critical, with a slight excess of the alkenyl azide (1.2 to 1.5 equivalents) relative to the carboxylic acid driving the reaction to completion. The process is operationally simple: the reagents are combined in the solvent, sealed in a reaction vessel, and heated with stirring for approximately 10 hours. Upon cooling, the solvent is removed under reduced pressure, and the crude residue is purified. This straightforward protocol minimizes the need for specialized equipment or rigorous exclusion of air and moisture, making it highly accessible for process chemistry teams aiming for rapid deployment.
- Mix alkenyl azide compound and carboxylic acid compound in an organic solvent such as 1,2-dichloroethane.
- Heat the reaction mixture to a temperature between 80°C and 120°C, preferably 110°C, and stir for approximately 10 hours.
- Upon completion, cool to room temperature, remove solvent, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of metal catalysts removes a significant cost center associated with both the purchase of expensive reagents and the subsequent purification steps required to remove metal traces. This simplification of the downstream processing workflow leads to substantial cost savings in manufacturing by reducing solvent usage, filtration media, and labor hours. Furthermore, the reliance on commodity chemicals like alkenyl azides and common carboxylic acids enhances supply chain resilience. These starting materials are widely available from multiple global vendors, mitigating the risk of single-source dependency and ensuring consistent availability even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain.
- Cost Reduction in Manufacturing: The catalyst-free nature of this process fundamentally alters the cost structure of producing alpha-amidoketones. By removing the need for transition metals, manufacturers avoid the high costs associated with catalyst procurement and the specialized waste treatment required for heavy metal disposal. The simplified workup procedure, which often avoids complex extraction or scavenging steps, significantly reduces the consumption of auxiliary materials and utilities. This lean manufacturing approach translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the marketplace. Additionally, the high atom economy ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste generation and associated disposal fees.
- Enhanced Supply Chain Reliability: The use of broadly available starting materials ensures that production schedules are not held hostage by the lead times of exotic reagents. Alkenyl azides and carboxylic acids are staple chemicals in the fine chemical industry, sourced from a mature and diversified supply base. This accessibility reduces the risk of production delays caused by raw material shortages. Moreover, the mild reaction conditions reduce the wear and tear on production equipment, lowering maintenance costs and extending the operational life of reactors. The ability to tolerate a wide range of functional groups means that the same process platform can be used to manufacture a diverse portfolio of intermediates, maximizing asset utilization and operational flexibility.
- Scalability and Environmental Compliance: The simplicity of the reaction setup—essentially a heat-and-stir protocol—makes this technology exceptionally amenable to scale-up. There are no complex dosing regimes or sensitive catalytic activation steps that often pose challenges when moving from gram to kilogram scales. The high yields and clean reaction profiles observed in the patent examples suggest that the process will maintain its efficiency at larger volumes. From an environmental perspective, the high atom economy and lack of toxic metal waste align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance reduces the regulatory burden on the manufacturer and enhances the sustainability profile of the final pharmaceutical product.
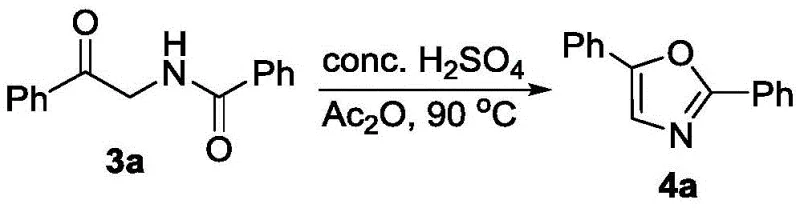
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the experimental data and claims presented in patent CN111960961A, providing a factual basis for evaluating the technology's fit within your specific production requirements. Understanding these nuances is essential for R&D teams planning to integrate this route into their existing workflows and for procurement specialists assessing the long-term viability of the supply chain.
Q: Does this synthesis method require expensive metal catalysts?
A: No, the method described in patent CN111960961A is completely catalyst-free, relying solely on thermal activation, which eliminates heavy metal contamination risks.
Q: What is the substrate scope for the carboxylic acid component?
A: The method exhibits exceptional breadth, tolerating complex drug molecules such as Adapalene, Ibuprofen, and Indomethacin, as well as various substituted benzoic acids and heterocycles.
Q: Can the resulting alpha-amidoketones be further derivatized?
A: Yes, the products serve as versatile intermediates that can be cyclized into oxazoles using sulfuric acid and acetic anhydride or functionalized to alpha-phenoxy derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amidoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in patent CN111960961A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this catalyst-free technology are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-amidoketone meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and consistency makes us a trusted ally in your drug development journey.
We invite you to leverage our capabilities to optimize your supply chain and reduce manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific target molecules, highlighting exactly how this new methodology can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your custom projects. Let us help you navigate the complexities of modern chemical synthesis with solutions that are efficient, scalable, and economically sound.
