Scalable Visible Light Arylation of Tetrahydroquinoline Derivatives for Commercial Manufacturing
Scalable Visible Light Arylation of Tetrahydroquinoline Derivatives for Commercial Manufacturing
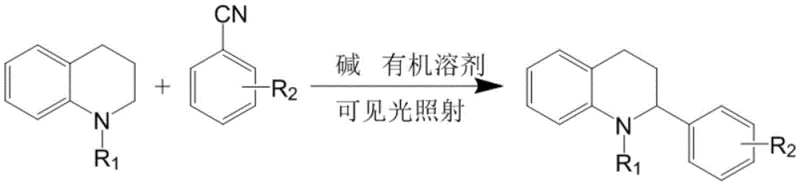
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in patent CN112812060A, which discloses a novel arylation method for tetrahydroquinoline derivatives. This technology represents a paradigm shift from traditional transition-metal catalyzed cross-couplings to a greener, visible-light-driven protocol. By leveraging the energy of visible light and simple inorganic bases, this method enables the direct functionalization of the tetrahydroquinoline core under exceptionally mild conditions. For R&D directors and process chemists, this offers a compelling alternative that simplifies purification workflows and reduces the environmental footprint of manufacturing. The ability to access these bioactive scaffolds without the burden of heavy metal residues is particularly valuable for the production of active pharmaceutical ingredients (APIs) where strict impurity limits are enforced.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of the tetrahydroquinoline skeleton, particularly at the C-2 or C-3 positions, has relied heavily on conventional synthetic strategies that often involve harsh reaction conditions and expensive reagents. Traditional approaches frequently utilize transition metal catalysts such as palladium, copper, or rhodium to facilitate C-C or C-N bond formation. While effective, these methods introduce significant downstream processing challenges, including the absolute necessity for rigorous metal scavenging steps to meet regulatory standards for residual metals in drug substances. Furthermore, many classical protocols require stoichiometric amounts of strong oxidants or operate at elevated temperatures, which can lead to thermal degradation of sensitive functional groups and the formation of complex impurity profiles. The reliance on noble metals also exposes the supply chain to volatility in raw material pricing and availability, creating bottlenecks for large-scale commercial production.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a metal-free, visible-light-mediated arylation strategy that circumvents these traditional hurdles. By employing N-substituted 1,2,3,4-tetrahydroquinoline and benzonitrile derivatives as starting materials, the reaction proceeds smoothly in the presence of an inorganic base and organic solvent under blue light irradiation. This approach eliminates the need for exogenous oxidants and transition metal catalysts entirely, resulting in a cleaner reaction matrix. The operational simplicity is profound; reactions can be conducted at room temperature, removing the energy costs associated with heating or cryogenic cooling. This not only enhances the safety profile of the manufacturing process but also significantly streamlines the workup procedure, as there are no metal complexes to remove. For a reliable pharmaceutical intermediate supplier, adopting such a streamlined protocol translates directly into improved process robustness and reduced time-to-market for new drug candidates.
Mechanistic Insights into Visible Light Mediated Radical Arylation
The mechanistic underpinning of this transformation likely involves a single-electron transfer (SET) process initiated by the interaction of the excited state species with the substrates under visible light irradiation. Although the patent does not explicitly detail a catalytic cycle involving a photocatalyst, the use of blue light suggests the generation of reactive radical intermediates from the benzonitrile derivative or the tetrahydroquinoline substrate itself, possibly facilitated by the base. This radical pathway allows for the direct coupling of the aromatic rings without the need for pre-functionalized halides or organometallic reagents typically required in cross-coupling chemistry. The absence of metal coordination complexes means that the reaction trajectory is governed primarily by electronic and steric factors of the organic substrates, leading to high regioselectivity. Understanding this mechanism is crucial for process optimization, as it highlights the importance of light intensity and wavelength control in maintaining consistent reaction kinetics across different batch sizes.
From an impurity control perspective, the metal-free nature of this reaction is a decisive advantage. In traditional metal-catalyzed processes, side reactions often involve the catalyst itself, leading to homocoupling byproducts or dehalogenated species that are difficult to separate. In this visible-light protocol, the primary byproducts are likely derived from unreacted starting materials or simple hydrolysis products, which are generally easier to remove via standard chromatographic or crystallization techniques. The use of inorganic bases like cesium carbonate or potassium phosphate further ensures that the reaction medium remains relatively benign, minimizing the risk of base-sensitive degradation. This clean impurity profile is essential for ensuring the high purity of the final tetrahydroquinoline arylation derivatives, making the process highly attractive for the synthesis of high-purity OLED material precursors or complex pharmaceutical intermediates where trace impurities can alter biological activity or material properties.
How to Synthesize Tetrahydroquinoline Arylation Derivatives Efficiently
Implementing this synthesis route requires careful attention to the ratio of reagents and the quality of the light source to ensure reproducibility. The patent specifies a mass ratio of N-substituted tetrahydroquinoline to benzonitrile derivative to base ranging from 2-4:1:2-4, with a preferred ratio of 2:1:2, indicating that an excess of the tetrahydroquinoline substrate drives the reaction to completion. The choice of solvent is also critical, with polar aprotic solvents like N,N-dimethylacetamide (DMAc) or dimethyl sulfoxide (DMSO) providing the optimal environment for the ionic and radical species involved. Operators must ensure that the reaction vessel allows for sufficient light penetration, as the photon flux is the driving force of the transformation. The detailed standardized synthesis steps see the guide below.
- Mix N-substituted 1,2,3,4-tetrahydroquinoline, benzonitrile derivative, and inorganic base in an organic solvent such as DMAc.
- Stir the reaction mixture at room temperature under irradiation of blue LED visible light for 15 to 40 hours.
- Perform workup via extraction with ethyl acetate, followed by rotary evaporation and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light arylation technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of precious metal catalysts represents a direct reduction in raw material costs, as palladium and other noble metals are subject to significant market fluctuations and high unit prices. Moreover, the removal of the metal scavenging step from the downstream processing workflow reduces the consumption of specialized resins and solvents, further driving down the cost of goods sold (COGS). The mild reaction conditions also imply lower energy consumption, as the process operates effectively at ambient temperature without the need for extensive heating or cooling infrastructure. These factors collectively contribute to a more resilient and cost-efficient supply chain for complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete avoidance of expensive transition metal catalysts and ligands. In traditional cross-coupling reactions, the cost of the catalyst system can be a major component of the overall budget, especially when high loadings are required. By switching to a base-promoted, light-driven system, manufacturers can eliminate this cost center entirely. Additionally, the simplified workup procedure reduces the labor and material costs associated with purification, as there is no need for specialized metal removal columns or multiple washes to reduce metal content to ppm levels. This streamlined process flow allows for faster batch turnover and higher overall equipment effectiveness.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as N-substituted tetrahydroquinolines and benzonitrile derivatives, are commodity chemicals that are readily available from multiple global suppliers. Unlike specialized organometallic reagents which may have long lead times or limited sources, these building blocks offer a stable and secure supply base. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but modern flow chemistry technologies and high-power LED arrays have made kilogram-to-ton scale photochemistry feasible. The absence of hazardous oxidants and heavy metals significantly simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The green chemistry metrics of this process are superior, with a lower E-factor due to reduced solvent and reagent usage. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for supplier selection in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light arylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into their existing manufacturing platforms.
Q: Does this arylation method require expensive transition metal catalysts?
A: No, the process described in patent CN112812060A is explicitly metal-free. It utilizes an inorganic base and visible light irradiation, eliminating the need for costly palladium or copper catalysts and the associated heavy metal removal steps.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds under remarkably mild conditions, specifically at room temperature. It requires an organic solvent like N,N-dimethylacetamide and irradiation with blue LED light, avoiding the need for extreme heating or cooling.
Q: Is this method suitable for generating diverse tetrahydroquinoline libraries?
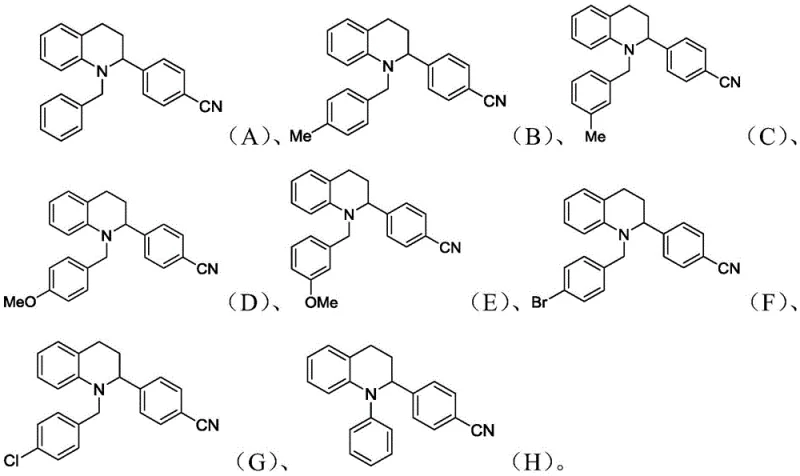
A: Yes, the method demonstrates broad substrate scope. It tolerates various substituents on both the tetrahydroquinoline nitrogen (such as benzyl, methylbenzyl, and phenyl groups) and the benzonitrile ring, allowing for the synthesis of structurally diverse derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photochemical synthesis in the production of high-value intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112812060A can be successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetrahydroquinoline derivative meets the highest quality standards required by the global pharmaceutical market.
We invite potential partners to engage with our technical team to explore how this green arylation method can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free protocol for your projects. We encourage you to contact our technical procurement team to obtain specific COA data for similar compounds and to discuss route feasibility assessments tailored to your unique molecular targets. Let us collaborate to bring safer, greener, and more cost-effective chemical solutions to the market.
