Advanced Pyrimidine-Pyrazole Ruthenium Catalyst for Efficient Pharmaceutical Intermediate Synthesis
Advanced Pyrimidine-Pyrazole Ruthenium Catalyst for Efficient Pharmaceutical Intermediate Synthesis
The landscape of transition metal catalysis is undergoing a significant transformation driven by the demand for greener, more energy-efficient synthetic routes, particularly in the production of high-value pharmaceutical intermediates. Patent CN109912661B introduces a breakthrough pyrimidine-pyrazole metal ruthenium complex, chemically defined as RuC14H10Br2Cl2N8, which represents a substantial leap forward in visible-light photocatalysis. This novel complex leverages the unique redox properties of Ruthenium(II) coordinated with a nitrogen-rich pyrimidine-pyrazole ligand system to facilitate organic transformations under exceptionally mild conditions. Unlike traditional thermal catalysis that often demands harsh temperatures and pressures, this innovation utilizes visible light quanta to access excited states, enabling efficient energy transfer and electron transport processes. For R&D directors and process chemists, this technology offers a compelling alternative for constructing carbon-carbon bonds, specifically through the addition of terminal alkynes to aromatic aldehydes, a critical step in synthesizing propargylic alcohol intermediates found in numerous bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the nucleophilic addition of terminal alkynes to aldehydes has been a staple reaction in organic synthesis, yet it is plagued by significant operational drawbacks that hinder industrial scalability and cost-efficiency. Traditional protocols typically rely on strong bases to generate acetylide anions, necessitating strictly anhydrous and oxygen-free environments to prevent catalyst deactivation or side reactions. Furthermore, these reactions often require cryogenic conditions, frequently operating at temperatures as low as -78°C, which imposes a massive energy burden on manufacturing facilities and requires specialized, expensive cooling infrastructure. The reliance on stoichiometric amounts of activating reagents not only increases raw material costs but also generates substantial chemical waste, complicating downstream purification and wastewater treatment processes. For supply chain managers, these rigorous conditions translate into longer batch cycles, higher utility costs, and increased safety risks associated with handling pyrophoric reagents and cryogenic fluids, making the conventional approach increasingly untenable for modern, sustainable manufacturing.
The Novel Approach
The pyrimidine-pyrazole ruthenium complex described in the patent offers a paradigm shift by enabling this transformation under ambient conditions using visible light as the sole energy source. This photocatalytic system eliminates the need for cryogenic cooling and strong stoichiometric bases, allowing the reaction to proceed efficiently at room temperature with only catalytic amounts of a mild tertiary amine, such as triethylamine. 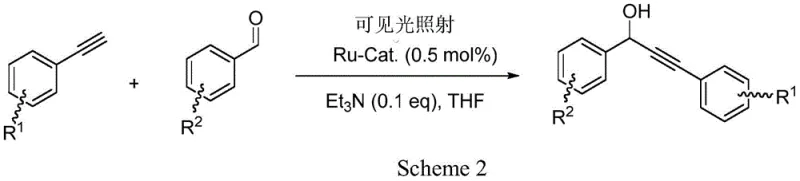 As illustrated in the reaction scheme, the catalyst activates the terminal alkyne through a photo-induced mechanism that bypasses the high-energy barriers associated with thermal activation. This approach not only drastically simplifies the operational procedure but also significantly enhances the safety profile of the process by removing hazardous reagents and extreme temperature requirements. For procurement teams, this translates to a reduction in the complexity of raw material sourcing and a decrease in the overall cost of goods sold (COGS) due to lower energy consumption and simplified waste management protocols.
As illustrated in the reaction scheme, the catalyst activates the terminal alkyne through a photo-induced mechanism that bypasses the high-energy barriers associated with thermal activation. This approach not only drastically simplifies the operational procedure but also significantly enhances the safety profile of the process by removing hazardous reagents and extreme temperature requirements. For procurement teams, this translates to a reduction in the complexity of raw material sourcing and a decrease in the overall cost of goods sold (COGS) due to lower energy consumption and simplified waste management protocols.
Mechanistic Insights into Pyrimidine-Pyrazole Ruthenium Photocatalysis
The efficacy of this catalytic system stems from the precise molecular architecture of the complex, where a central Ruthenium(II) ion is coordinated in a distorted octahedral geometry by two pyrimidine-pyrazole ligands and two chloride ions. The synthesis of this active species involves a straightforward two-step protocol, beginning with the formation of the organic ligand followed by metal coordination. 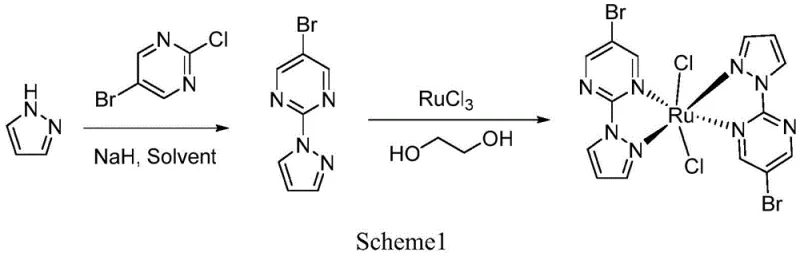 As shown in the synthesis pathway, the ligand is constructed via a nucleophilic substitution between pyrazole and 5-bromo-2-chloropyrimidine, followed by complexation with ruthenium trichloride in an ethylene glycol-water mixture. The resulting complex exhibits excellent stability and solubility characteristics, allowing it to function effectively in common organic solvents like tetrahydrofuran. The nitrogen-rich environment provided by the pyrimidine and pyrazole rings facilitates strong metal-ligand bonding, which stabilizes the ruthenium center against decomposition while maintaining the electronic flexibility required for photo-redox cycling. This structural integrity ensures that the catalyst remains active over extended reaction periods, minimizing the need for frequent replenishment and reducing the potential for metal contamination in the final product.
As shown in the synthesis pathway, the ligand is constructed via a nucleophilic substitution between pyrazole and 5-bromo-2-chloropyrimidine, followed by complexation with ruthenium trichloride in an ethylene glycol-water mixture. The resulting complex exhibits excellent stability and solubility characteristics, allowing it to function effectively in common organic solvents like tetrahydrofuran. The nitrogen-rich environment provided by the pyrimidine and pyrazole rings facilitates strong metal-ligand bonding, which stabilizes the ruthenium center against decomposition while maintaining the electronic flexibility required for photo-redox cycling. This structural integrity ensures that the catalyst remains active over extended reaction periods, minimizing the need for frequent replenishment and reducing the potential for metal contamination in the final product.
From a mechanistic perspective, the absorption of visible light promotes an electron from the metal-centered d-orbitals to the ligand-centered pi-star orbitals (MLCT transition), generating a long-lived excited state capable of engaging in single-electron transfer processes. This excited state interacts with the terminal alkyne and the amine base to generate a reactive radical species that adds to the carbonyl group of the aromatic aldehyde. The mild nature of this activation mode allows for exceptional functional group tolerance, accommodating sensitive substituents such as halogens (fluorine, chlorine, bromine, iodine) without inducing unwanted side reactions like dehalogenation. For quality control teams, this selectivity is paramount, as it ensures a cleaner impurity profile and simplifies the purification process, ultimately leading to higher yields of the desired propargylic alcohol intermediates with minimal byproduct formation.
How to Synthesize Pyrimidine-Pyrazole Ruthenium Complex Efficiently
The preparation of this high-performance catalyst is designed for scalability and reproducibility, utilizing commercially available starting materials and standard laboratory equipment. The process avoids the use of exotic reagents or ultra-high vacuum techniques, making it accessible for both pilot-scale and commercial manufacturing environments. The initial ligand synthesis achieves high yields through simple recrystallization, while the subsequent metal coordination step produces the final complex as distinct yellow needle crystals that are easily isolated by filtration.
- Suspend sodium hydride in toluene, add pyrazole, then react with 5-bromo-2-chloropyrimidine to form the ligand.
- Dissolve the ligand in ethylene glycol/water, add ruthenium trichloride, and heat to 120°C to form the yellow needle crystals.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this visible-light catalytic technology offers profound strategic advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing and supply chain resilience. By shifting from energy-intensive thermal processes to ambient light-driven reactions, manufacturers can achieve significant operational expenditure (OpEx) savings through reduced utility consumption and the elimination of cryogenic infrastructure. The simplified reaction conditions also mean that existing reactor fleets can be utilized with minimal modification, avoiding the need for capital-intensive equipment upgrades. Furthermore, the use of mild reagents and the generation of less hazardous waste streams align with increasingly stringent environmental regulations, reducing the compliance burden and associated disposal costs for large-scale production facilities.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling and strong stoichiometric bases directly lowers the variable costs associated with each production batch. By operating at room temperature with low catalyst loading (0.5 mol%), the process minimizes energy usage and raw material consumption, leading to substantial cost savings over the lifecycle of the product. Additionally, the high selectivity of the catalyst reduces the formation of byproducts, which decreases the load on downstream purification units and improves overall material throughput.
- Enhanced Supply Chain Reliability: The robustness of the pyrimidine-pyrazole ruthenium complex ensures consistent performance across different batches, mitigating the risk of production delays caused by catalyst instability or sensitivity. The use of common solvents and readily available reagents simplifies the procurement process, reducing dependency on specialized suppliers and enhancing the agility of the supply chain. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this technology highly scalable from gram to ton quantities without compromising safety or efficiency. The reduction in hazardous waste generation supports corporate sustainability goals and facilitates easier regulatory approval for new manufacturing sites. This environmental compatibility positions the technology as a future-proof solution for green chemistry initiatives within the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the patent data to provide accurate guidance for process development and procurement strategies.
Q: What are the storage conditions for this ruthenium complex?
A: The complex forms stable yellow needle crystals and should be stored in a cool, dry place away from direct light to maintain catalytic activity, although it demonstrates robust stability under reaction conditions.
Q: Does this catalyst require specialized light equipment?
A: No specialized high-energy equipment is needed; the patent specifies that standard LED lamps (e.g., 21W) are sufficient to drive the photocatalytic cycle effectively at room temperature.
Q: Can this method tolerate functional groups like halogens?
A: Yes, the catalytic system shows excellent tolerance for halogenated substrates, such as 4-iodobenzaldehyde, yielding high conversion rates without dehalogenation side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine-Pyrazole Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic systems like the pyrimidine-pyrazole ruthenium complex in modernizing pharmaceutical intermediate synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of catalyst or intermediate meets the exacting standards required by global multinational corporations.
We invite you to collaborate with our technical team to explore how this visible-light catalytic route can optimize your specific manufacturing challenges. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your production volume. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring a seamless integration of this cutting-edge technology into your supply chain.
