Advanced Pyrimidine-Pyrazole Ruthenium Complex for Scalable Pharmaceutical Intermediate Manufacturing
The chemical industry is currently witnessing a paradigm shift towards sustainable catalytic processes, exemplified by the innovations detailed in patent CN109912661B. This intellectual property introduces a novel pyrimidine-pyrazole metal ruthenium complex that fundamentally alters the landscape of alkyne-aldehyde addition reactions. Traditionally, the synthesis of propargylic alcohols, which are critical building blocks for numerous active pharmaceutical ingredients (APIs), has been plagued by harsh reaction conditions and significant environmental burdens. The disclosed technology leverages the unique electronic properties of a mononuclear hexacoordinate ruthenium(II) center to harness visible light energy, thereby driving catalytic cycles under ambient conditions. For R&D directors and process chemists, this represents a monumental opportunity to transition away from energy-intensive cryogenic protocols toward greener, photo-driven methodologies. The structural integrity of the complex, characterized by precise Ru-N and Ru-Cl bond lengths within an octahedral geometry, ensures exceptional stability and turnover efficiency. By integrating this advanced catalyst into existing synthetic workflows, manufacturers can achieve substantial improvements in process safety and operational simplicity while maintaining the rigorous purity standards required for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic protocols for the nucleophilic addition of terminal alkynes to aromatic aldehydes historically rely on the generation of highly reactive acetylide anions. This traditional approach necessitates the use of stoichiometric amounts of strong bases, such as n-butyllithium or lithium diisopropylamide, under strictly anhydrous and anaerobic conditions. Furthermore, these reactions typically require cryogenic temperatures ranging down to -78°C to prevent side reactions and ensure regioselectivity. Such rigorous operational parameters impose a substantial burden on industrial infrastructure, requiring specialized low-temperature reactors and extensive safety protocols for handling pyrophoric reagents. The energy consumption associated with maintaining cryogenic conditions significantly escalates the operational expenditure, while the sensitivity to moisture often leads to batch failures and inconsistent purity profiles. Consequently, the scalability of these conventional methods is severely restricted, making them less attractive for large-scale manufacturing of pharmaceutical intermediates where cost and reliability are paramount concerns for supply chain managers.
The Novel Approach
In stark contrast, the pyrimidine-pyrazole metal ruthenium complex described in the patent offers a transformative solution by utilizing visible light as the sole energy source to activate the catalytic cycle. This innovative methodology eliminates the need for cryogenic cooling and stoichiometric strong bases, allowing the reaction to proceed smoothly at room temperature with only catalytic amounts of a mild tertiary amine. The mechanism involves the absorption of light quanta by the ruthenium center, facilitating an oxidation-reduction transition state that activates the terminal alkyne without generating hazardous free acetylides. This shift not only drastically reduces the energy footprint of the synthesis but also simplifies the reactor setup, removing the need for complex low-temperature control systems. For procurement teams, this translates to a significant reduction in utility costs and a decrease in the reliance on specialized, high-maintenance equipment. The robustness of the catalyst under ambient conditions further enhances process reliability, ensuring consistent output quality even in varied manufacturing environments, thereby securing the supply chain against technical disruptions.
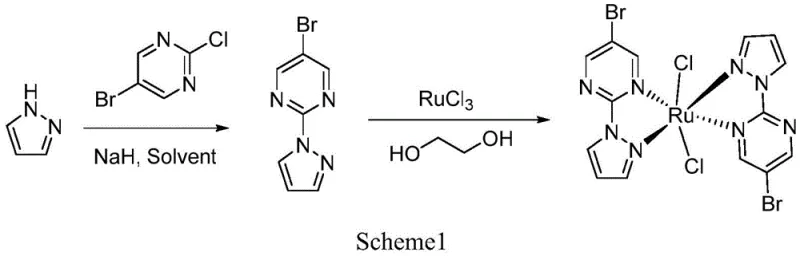
Mechanistic Insights into Visible-Light Driven Ruthenium Catalysis
The efficacy of this catalytic system is rooted in the sophisticated coordination chemistry of the ruthenium(II) center within the pyrimidine-pyrazole ligand framework. The central metal atom adopts a distorted octahedral geometry, coordinated by four nitrogen atoms from two bidentate pyrimidine-pyrazole ligands and two chlorine ions. This specific arrangement creates an electronic environment that is highly conducive to photo-excitation, allowing the complex to absorb visible light and reach an excited state capable of electron transfer. The Ru-N bond lengths, ranging precisely between 1.948(3) and 2.412(24) Å, and the Ru-Cl bond length of approximately 2.135 Å, indicate a stable yet labile coordination sphere that facilitates substrate binding and product release. Upon irradiation, the complex likely undergoes a metal-to-ligand charge transfer (MLCT), generating a transient species that can interact with the terminal alkyne and the tertiary amine base. This interaction lowers the activation energy for the formation of the ruthenium-acetylide intermediate, which subsequently attacks the carbonyl carbon of the aromatic aldehyde. The precise tuning of the ligand electronics ensures that the catalyst remains active over multiple cycles without decomposing, providing the high turnover numbers observed in experimental trials.
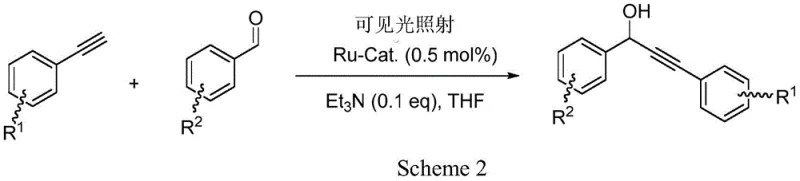
From an impurity control perspective, the mild nature of this photo-driven mechanism offers distinct advantages over traditional base-mediated pathways. Conventional methods often suffer from over-addition, polymerization of the alkyne, or degradation of sensitive functional groups due to the aggressive nature of strong bases and low temperatures. The ruthenium-catalyzed process, operating under neutral to mildly basic conditions with triethylamine, exhibits excellent chemoselectivity. It tolerates various substituents on the aromatic aldehyde, including halogens like iodine, without inducing unwanted side reactions such as dehalogenation or homocoupling of the alkyne. This high selectivity minimizes the formation of by-products, thereby simplifying the downstream purification process. For quality assurance teams, this means a cleaner crude reaction profile, which reduces the load on chromatography columns and crystallization steps. The ability to maintain high purity without extensive workup procedures directly contributes to higher overall yields and reduced solvent waste, aligning perfectly with green chemistry principles and regulatory expectations for pharmaceutical manufacturing.
How to Synthesize Pyrimidine-Pyrazole Ruthenium Complex Efficiently
The preparation of this high-performance catalyst is designed for scalability and reproducibility, involving a straightforward two-step sequence that avoids exotic reagents or inaccessible equipment. The process begins with the synthesis of the organic ligand via a nucleophilic substitution reaction, followed by the coordination of the ligand to the ruthenium metal center in a mixed solvent system. The conditions are optimized to maximize yield while ensuring the formation of the correct crystalline phase, which is critical for catalytic activity. Detailed standard operating procedures for the synthesis, including specific molar ratios, temperature profiles, and purification techniques, are essential for achieving the benchmark performance described in the patent literature. Manufacturers aiming to adopt this technology should adhere strictly to the specified reaction times and solvent volumes to ensure the structural integrity of the final complex.
- Suspend sodium hydride in toluene, add pyrazole, and react with 5-bromo-2-chloropyrimidine to form the pyrimidine-pyrazole ligand.
- Dissolve the ligand in ethylene glycol/water, add ruthenium trichloride, and heat to 120°C to crystallize the yellow needle-shaped complex.
- Utilize the complex (0.5 mol%) with triethylamine under visible light irradiation to catalyze the addition of terminal alkynes to aromatic aldehydes.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this visible-light driven ruthenium catalyst presents compelling economic and logistical benefits for organizations managing the production of fine chemicals and pharmaceutical intermediates. By shifting from cryogenic, base-heavy processes to ambient, photo-driven catalysis, companies can realize substantial cost savings across multiple vectors of their operation. The elimination of expensive cooling infrastructure and the reduction in energy consumption directly lower the variable costs associated with manufacturing. Furthermore, the simplified reaction setup reduces the capital expenditure required for plant upgrades, allowing existing facilities to be repurposed for this advanced chemistry with minimal modification. For supply chain heads, the robustness of the catalyst and the availability of commercially sourced starting materials like ruthenium trichloride and pyrazole derivatives ensure a stable and continuous supply of the catalyst itself. This reliability mitigates the risk of production delays caused by reagent shortages or equipment failures, fostering a more resilient supply network.
- Cost Reduction in Manufacturing: The transition to this catalytic system eliminates the need for stoichiometric amounts of expensive and hazardous strong bases, replacing them with catalytic quantities of inexpensive tertiary amines. Additionally, the removal of cryogenic cooling requirements results in drastic energy savings, as reactors no longer need to be maintained at -78°C. The high selectivity of the reaction minimizes waste generation and reduces the volume of solvents required for purification, further driving down disposal and material costs. These cumulative effects lead to a significantly leaner cost structure for the production of propargylic alcohol intermediates.
- Enhanced Supply Chain Reliability: The synthesis of the catalyst utilizes readily available commodity chemicals, reducing dependency on specialized or single-source suppliers. The stability of the final ruthenium complex allows for easier storage and transportation compared to sensitive organometallic reagents that require inert atmosphere handling. This ease of handling simplifies logistics and reduces the risk of spoilage during transit. Moreover, the mild reaction conditions reduce wear and tear on production equipment, extending asset life and minimizing unplanned maintenance downtime that could disrupt supply continuity.
- Scalability and Environmental Compliance: The use of visible light as an energy source aligns with global sustainability goals, reducing the carbon footprint of the manufacturing process. The absence of harsh reagents simplifies waste treatment protocols, making it easier to comply with increasingly stringent environmental regulations. The process is inherently scalable, as light penetration can be managed through reactor design, allowing for seamless transition from laboratory benchtop to multi-ton commercial production without losing efficiency or selectivity.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrimidine-pyrazole ruthenium complex in industrial settings. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Understanding the operational boundaries and capabilities of this catalyst is crucial for R&D teams planning process validation and for procurement officers evaluating vendor qualifications. The answers below clarify the practical aspects of using this technology, from reaction conditions to substrate compatibility.
Q: Does this ruthenium catalyst require cryogenic conditions?
A: No, unlike conventional methods requiring -78°C, this novel complex operates efficiently at room temperature using visible light as an energy source.
Q: What is the typical yield for the catalytic addition reaction?
A: Experimental data indicates excellent yields, reaching up to 99% for phenylacetylene and benzaldehyde substrates under optimized conditions.
Q: Is the catalyst compatible with halogenated substrates?
A: Yes, the catalyst demonstrates high tolerance for functional groups, successfully catalyzing reactions with iodo-substituted aromatic aldehydes without dehalogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine-Pyrazole Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the pyrimidine-pyrazole metal ruthenium complex in modernizing pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this advanced catalytic technology is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of photochemical reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee the performance of every batch. We understand that consistency is key in API manufacturing, and our dedicated technical team works closely with clients to optimize reaction parameters for maximum yield and minimal impurity formation.
We invite you to collaborate with us to leverage this cutting-edge catalytic solution for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and current process constraints. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us help you drive innovation and efficiency in your supply chain with our premium catalytic solutions.
