Scalable Enzymatic Production of High-Purity Eluxadoline Intermediates for Global Pharma Supply Chains
The pharmaceutical landscape for treating Irritable Bowel Syndrome (IBS-D) has been significantly shaped by the approval of Eluxadoline, commercially known as Viberzi. As demand for this therapeutic agent grows, the efficiency of its supply chain becomes a critical focal point for global manufacturers. Patent CN106636241B introduces a groundbreaking enzymatic preparation method for a key Eluxadoline intermediate, specifically addressing the bottlenecks associated with traditional chemical synthesis. This innovation shifts the paradigm from harsh chemical transformations to a green, biocatalytic approach, utilizing nitrile hydratase to convert a nitrile precursor into the essential amide functionality found in the drug's backbone. By leveraging specific biological catalysts, this technology promises to deliver high-purity intermediates with superior stereochemical integrity, directly impacting the quality and availability of the final Active Pharmaceutical Ingredient (API).
The structural complexity of Eluxadoline requires precise construction of its phenylalanine core, a task that has historically relied on multi-step chemical sequences. 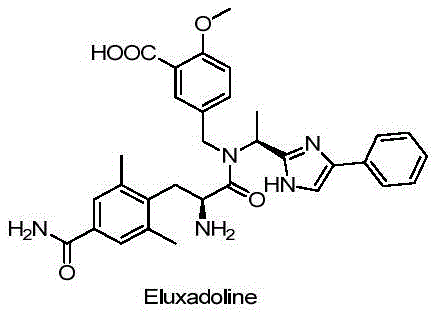 . The intermediate designated as Compound I serves as a pivotal building block in this architecture. Traditional routes often struggle with maintaining optical purity while navigating the reactivity of sensitive functional groups. The patented method described in CN106636241B offers a robust solution by decoupling the formation of the carbon skeleton from the installation of the amide group, allowing each step to be optimized independently for maximum efficiency and minimal waste generation.
. The intermediate designated as Compound I serves as a pivotal building block in this architecture. Traditional routes often struggle with maintaining optical purity while navigating the reactivity of sensitive functional groups. The patented method described in CN106636241B offers a robust solution by decoupling the formation of the carbon skeleton from the installation of the amide group, allowing each step to be optimized independently for maximum efficiency and minimal waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described by Chaozhong Cai et al., typically rely on the modification of expensive natural amino acid derivatives like N-Boc-2,6-dimethyl-L-tyrosine. 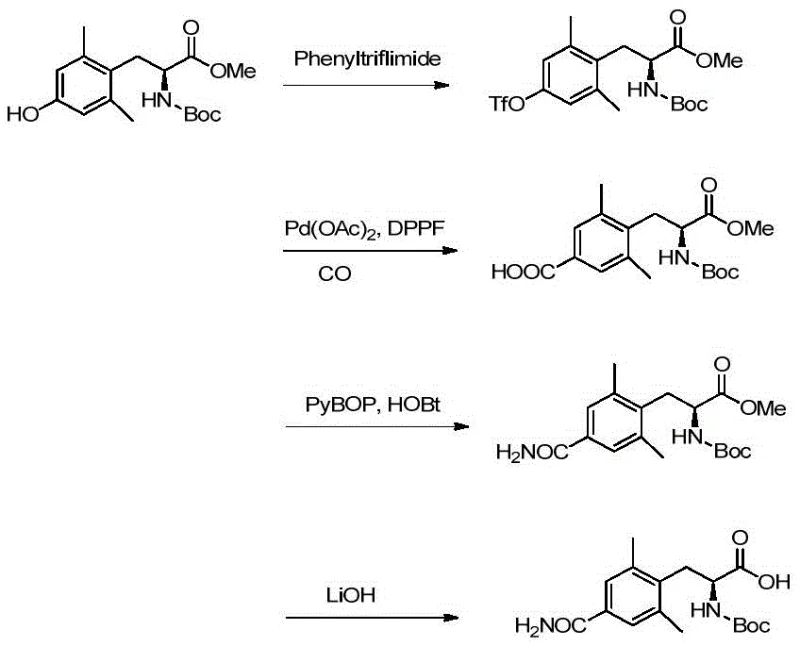 . These legacy processes are fraught with inefficiencies, including the high cost of chiral starting materials and a linear sequence of four distinct reaction steps that cumulatively erode the overall yield. Furthermore, the chemical hydrolysis of nitriles to amides, if attempted directly, often requires aggressive conditions involving strong acids or bases and elevated temperatures. Such harsh environments pose a significant risk of racemization at the chiral center and can lead to over-hydrolysis, producing unwanted carboxylic acid byproducts that are difficult to separate, thereby complicating downstream purification and increasing production costs.
. These legacy processes are fraught with inefficiencies, including the high cost of chiral starting materials and a linear sequence of four distinct reaction steps that cumulatively erode the overall yield. Furthermore, the chemical hydrolysis of nitriles to amides, if attempted directly, often requires aggressive conditions involving strong acids or bases and elevated temperatures. Such harsh environments pose a significant risk of racemization at the chiral center and can lead to over-hydrolysis, producing unwanted carboxylic acid byproducts that are difficult to separate, thereby complicating downstream purification and increasing production costs.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a highly selective biocatalytic strategy. 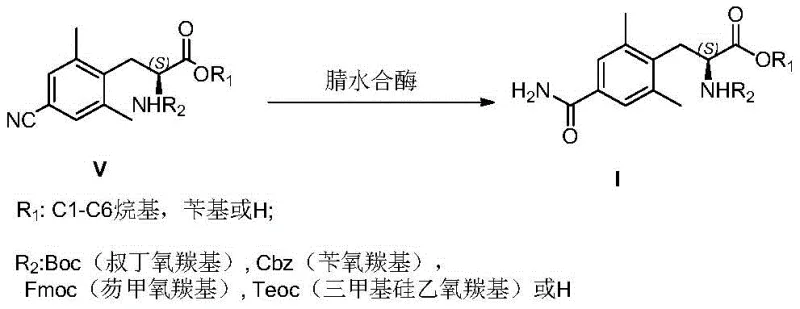 . This method employs nitrile hydratase enzymes to effect the hydration of the nitrile group in Compound V under exceptionally mild aqueous conditions, typically at temperatures between 30°C and 40°C and a neutral pH range of 6 to 8. This biological specificity ensures that only the nitrile group is transformed into the primary amide, leaving other sensitive moieties such as the Boc-protecting group and the ester functionality completely intact. The result is a streamlined process that bypasses the need for hazardous reagents and extreme thermal inputs, offering a cleaner reaction profile that simplifies work-up procedures and significantly enhances the safety profile of the manufacturing operation.
. This method employs nitrile hydratase enzymes to effect the hydration of the nitrile group in Compound V under exceptionally mild aqueous conditions, typically at temperatures between 30°C and 40°C and a neutral pH range of 6 to 8. This biological specificity ensures that only the nitrile group is transformed into the primary amide, leaving other sensitive moieties such as the Boc-protecting group and the ester functionality completely intact. The result is a streamlined process that bypasses the need for hazardous reagents and extreme thermal inputs, offering a cleaner reaction profile that simplifies work-up procedures and significantly enhances the safety profile of the manufacturing operation.
Mechanistic Insights into Nitrile Hydratase Catalysis
The core of this technological advancement lies in the precise mechanism of nitrile hydratase enzymes, which facilitate the addition of water across the carbon-nitrogen triple bond. Unlike chemical catalysts that might lack discrimination, these enzymes possess an active site tailored to recognize the specific steric and electronic environment of the substrate, Compound V. The reaction proceeds through a coordinated mechanism where the nitrile nitrogen binds to a metal center within the enzyme, activating the carbon atom for nucleophilic attack by a water molecule. This controlled environment prevents the formation of reactive intermediates that could lead to side reactions. Consequently, the process achieves near-quantitative conversion rates with exceptional chemoselectivity, ensuring that the delicate balance of functional groups required for the subsequent coupling steps in Eluxadoline synthesis is preserved without the need for additional protection-deprotection cycles.
Furthermore, the enzymatic pathway provides a robust mechanism for impurity control, which is paramount for regulatory compliance in pharmaceutical manufacturing. Chemical hydrolysis often generates a spectrum of byproducts, including the corresponding carboxylic acid and unreacted starting material, which can be structurally similar and challenging to remove. The enzymatic process, however, exhibits a "stop-at-amide" characteristic, effectively suppressing over-hydrolysis to the acid. Additionally, the mild pH conditions prevent epimerization at the alpha-carbon, maintaining the critical (S)-configuration of the phenylalanine derivative with an enantiomeric excess consistently exceeding 99%. This high level of stereochemical fidelity reduces the burden on chiral chromatography or recrystallization steps, directly contributing to a more efficient and cost-effective purification workflow.
How to Synthesize Eluxadoline Intermediate Efficiently
The synthesis of the target intermediate involves a convergent strategy where the aromatic core and the amino acid side chain are assembled prior to the biocatalytic step. The process begins with the generation of an organozinc reagent from an iodo-alanine derivative, which is then coupled with a substituted iodobenzene using palladium catalysis to form the nitrile precursor. 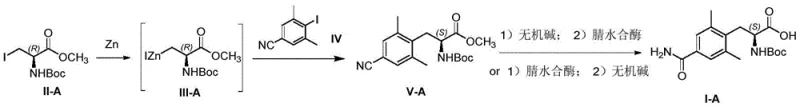 . Once the nitrile substrate is secured, the enzymatic transformation is executed in a buffered aqueous system. Detailed operational parameters, including enzyme loading, substrate concentration, and pH control strategies, are critical for maximizing space-time yield. For the complete standardized operating procedure and specific reaction conditions validated at scale, please refer to the detailed synthesis guide below.
. Once the nitrile substrate is secured, the enzymatic transformation is executed in a buffered aqueous system. Detailed operational parameters, including enzyme loading, substrate concentration, and pH control strategies, are critical for maximizing space-time yield. For the complete standardized operating procedure and specific reaction conditions validated at scale, please refer to the detailed synthesis guide below.
- Preparation of the nitrile precursor (Compound V) via zinc-mediated Reformatsky reaction followed by palladium-catalyzed coupling.
- Suspension of the nitrile substrate in a phosphate buffer solution adjusted to pH 6-8.
- Addition of nitrile hydratase enzyme powder (e.g., NHT101) and stirring at 30-40°C until conversion is complete, followed by protein denaturation and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits beyond mere technical novelty. The shift from a four-step chemical sequence reliant on expensive chiral pool materials to a shorter, biocatalytic route fundamentally alters the cost structure of the intermediate. By utilizing readily available achiral or racemic starting materials that are resolved or dynamically kinetic resolved during the process, manufacturers can decouple their supply from the volatile pricing of specialized natural amino acid derivatives. This flexibility allows for more stable long-term contracting and reduces exposure to raw material shortages that frequently plague the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of harsh chemical reagents and the reduction in the number of unit operations directly lower the variable costs associated with production. The mild reaction conditions obviate the need for specialized corrosion-resistant equipment and extensive energy consumption for heating or cooling, leading to substantial savings in utility costs. Moreover, the high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, which reduces the consumption of solvents and chromatography media during purification, further driving down the cost of goods sold (COGS) without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process enhances supply chain resilience by simplifying the manufacturing workflow. Fewer reaction steps mean fewer opportunities for batch failures or yield losses, resulting in more predictable production timelines. The use of stable enzyme preparations, which can be stored and handled with relative ease compared to sensitive organometallic catalysts, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, mitigating the risk of stockouts that could disrupt the availability of the final medication to patients.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach offers significant advantages for large-scale operations. The use of aqueous buffers and the absence of heavy metal catalysts in the final step simplify waste treatment and disposal, aligning with increasingly stringent environmental regulations. The process has already demonstrated successful scaling from gram to hectogram quantities in the patent examples, indicating a clear path to metric-ton production. This scalability ensures that the technology can meet growing market demand while maintaining a reduced environmental footprint, a key factor for pharmaceutical companies aiming to meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the advantages of the enzymatic method over traditional chemical hydrolysis?
A: The enzymatic method operates under mild conditions (30-40°C, pH 6-8) compared to harsh acidic or basic hydrolysis, preventing racemization and side reactions like over-hydrolysis to carboxylic acids, thereby ensuring higher purity and yield.
Q: Which specific enzymes are suitable for this transformation?
A: Commercially available nitrile hydratases such as NHT101 to NHT124 from Shangke Biomedical have been demonstrated to effectively catalyze the hydration of the specific nitrile intermediate with high stereoselectivity.
Q: Is this process scalable for industrial production?
A: Yes, the patent data demonstrates successful scaling from gram to hectogram levels (e.g., 150g scale) with consistent high yields (96-98%) and excellent enantiomeric excess (>99% ee), indicating strong potential for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eluxadoline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic processes in modern pharmaceutical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN106636241B can be seamlessly translated into industrial reality. We are committed to delivering high-purity Eluxadoline intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex enzymatic reactions with the same precision and reliability as traditional chemical synthesis, providing a secure source for your critical supply chain needs.
We invite you to collaborate with us to leverage this advanced technology for your Eluxadoline projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic route can optimize your budget. Please contact us to request specific COA data from our pilot batches and comprehensive route feasibility assessments, allowing you to make informed decisions about integrating this efficient and sustainable manufacturing method into your supply chain.
