Scalable Synthesis of L-Alanine Derivatives for Eluxadoline via Advanced Negishi Coupling Technology
Scalable Synthesis of L-Alanine Derivatives for Eluxadoline via Advanced Negishi Coupling Technology
The pharmaceutical landscape for treating diarrhea-predominant irritable bowel syndrome (IBS-D) has been significantly shaped by the approval of Eluxadoline, marketed under the trade name Viberzi. As a mixed opioid receptor agonist and antagonist, this molecule requires highly specific chiral intermediates to ensure efficacy and safety. Patent CN110092735B, published in May 2021, introduces a groundbreaking preparation method for a critical L-alanine derivative, specifically (S)-2-tert-butyloxycarbonylamino-3-(4-carbamoyl-2,6-dimethylphenyl)propionic acid. This technical disclosure represents a pivotal shift from traditional, low-yield synthetic pathways to a robust, palladium-catalyzed Negishi coupling strategy. For R&D directors and procurement specialists alike, this patent offers a blueprint for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with superior atom economy. The innovation lies not just in the chemical transformation but in the strategic protection of functional groups that historically plagued earlier synthesis attempts.
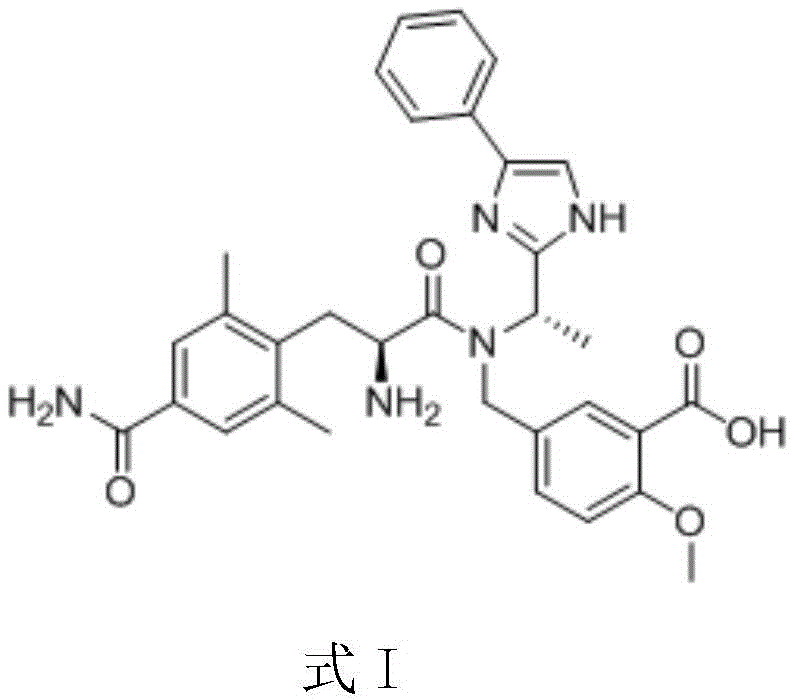
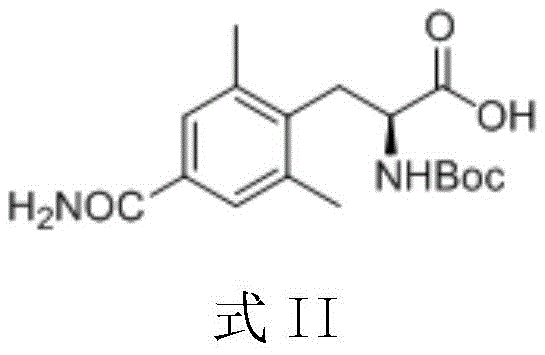
Understanding the molecular architecture of Eluxadoline is crucial for appreciating the complexity of its synthesis. The target molecule relies heavily on the stereochemical integrity of its alanine-derived side chain. The patent explicitly identifies the compound shown in Formula II as the key intermediate. Historically, accessing this scaffold involved convoluted routes that compromised both cost and quality. The new methodology described in CN110092735B addresses these bottlenecks by streamlining the carbon-carbon bond formation step. By focusing on the preparation of this specific L-alanine derivative, the patent provides a direct pathway to enhancing the supply chain stability for this high-value therapeutic agent. This is particularly relevant for manufacturers seeking cost reduction in API manufacturing where raw material efficiency is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
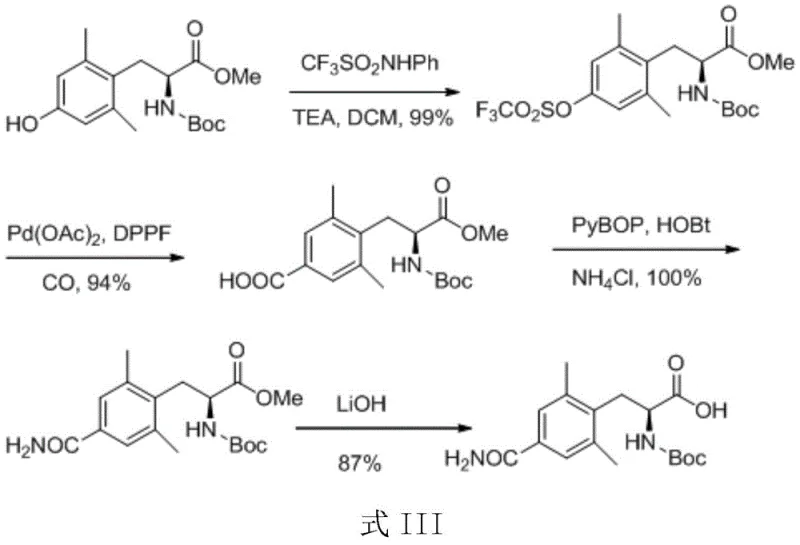
Prior to this invention, the synthesis of the requisite L-alanine derivative was fraught with inefficiencies that hindered commercial viability. One prominent prior art route, disclosed in WO2003092688, utilized Boc-L-2,6-dimethyl-tyrosine methyl ester as a starting material. While chemically feasible, this approach suffered from the high cost and structural complexity of the starting raw material, which is difficult to prepare on a large scale. Furthermore, the intermediate synthesis steps in this legacy route required noble metal catalysts that drove up production costs significantly. Another route, described in WO2006099060, attempted to graft micromolecules onto aromatic rings but resulted in low yields and significant damage to the Boc amide protection, leading to a plethora of byproducts that complicated separation and purification. These conventional methods often struggled to maintain chiral purity, with some steps yielding products with insufficient ee values, ultimately affecting the quality of the final API.
The Novel Approach
In stark contrast, the novel approach detailed in CN110092735B leverages a Negishi coupling reaction to construct the core carbon skeleton efficiently. This method bypasses the need for expensive, hard-to-source tyrosine derivatives by utilizing simpler, more accessible starting materials like compound 3. The innovation effectively solves the problem of low yield and byproduct formation associated with previous coupling attempts. By protecting the carboxyl group as an amide during the critical coupling phase, the process prevents the degradation of the Boc protecting group, a common failure point in earlier methodologies. This strategic modification ensures that the reaction proceeds with high fidelity, minimizing the generation of impurities that are costly to remove. The result is a streamlined process where the overall yield from compound 3 is not lower than 46.7 percent, a substantial improvement over the roughly 20 percent yields observed in some prior art. This leap in efficiency translates directly to enhanced supply chain reliability and reduced waste generation.
Mechanistic Insights into Pd-Catalyzed Negishi Coupling
The heart of this synthetic breakthrough is the palladium-catalyzed Negishi coupling between an organozinc reagent and an aryl halide. The mechanism begins with the activation of zinc powder, typically using 1,2-dibromoethane and trimethylchlorosilane in anhydrous DMF. This activation step is critical for generating the reactive organozinc species from the iodo-compound (compound 3). Once formed, this zinc reagent undergoes transmetallation with the palladium catalyst, which has previously undergone oxidative addition with the aryl iodide (compound 5). The use of specific ligands, such as P(o-tolyl)3, facilitates this cycle, ensuring high turnover numbers and minimizing catalyst loading. The reaction conditions are meticulously controlled, with temperatures maintained between 80-100°C to drive the coupling to completion without compromising the sensitive chiral center. This precise control over reaction parameters is what allows the process to achieve such high levels of stereochemical retention.
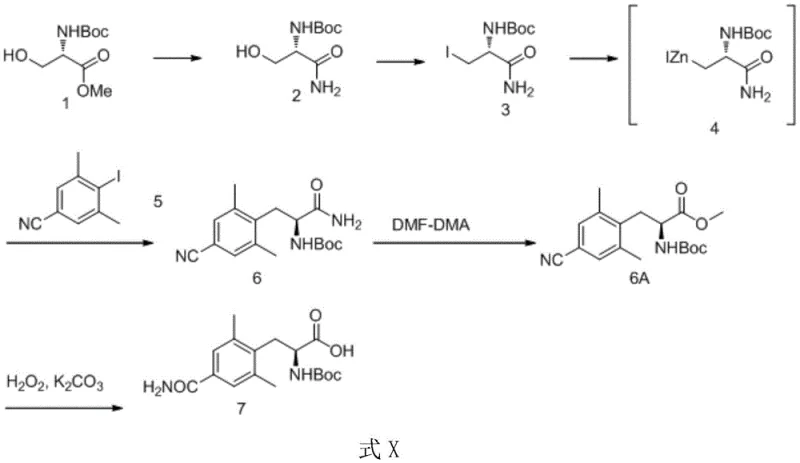
Impurity control is another cornerstone of this mechanistic design. In traditional routes, the presence of free carboxylic acids or esters during the coupling phase often led to side reactions or catalyst poisoning. By converting the carboxyl group into a primary amide (compound 3) prior to the Negishi step, the inventors effectively mask this reactive site. This protection strategy ensures that the palladium catalyst interacts exclusively with the intended carbon-zinc and carbon-iodine bonds. Furthermore, the subsequent conversion of the amide back to the ester (compound 6A) using DMF-DMA is a mild and selective transformation that avoids racemization. Finally, the hydrolysis of the nitrile group to the primary amide using hydrogen peroxide and potassium carbonate is performed under basic conditions that preserve the stereochemistry of the alpha-carbon. This multi-layered approach to impurity management ensures that the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize (S)-2-tert-butyloxycarbonylamino-3-(4-carbamoyl-2,6-dimethylphenyl)propionic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the ammonolysis of a methyl ester to form the primary amide, followed by iodination to create the coupling partner. The core Negishi coupling is then executed in a one-pot fashion, followed by esterification and final hydrolysis. This sequence minimizes the number of isolation steps, which is a key factor in reducing overall processing time and solvent consumption. For technical teams looking to implement this route, the detailed conditions regarding zinc activation and catalyst loading are critical success factors. The patent emphasizes that while six chemical transformations occur, only three distinct purification steps are necessary, highlighting the operational simplicity of the design. Detailed standardized synthesis steps are provided in the guide below.
- Perform ammonolysis on methyl ester precursor followed by iodination to generate the key zinc reagent precursor.
- Activate zinc powder and react with the iodo-compound to form the organozinc intermediate in situ.
- Execute palladium-catalyzed Negishi coupling with 2,5-dimethyl-4-cyanoiodobenzene, followed by esterification and final hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patent are profound. The shift from low-yielding, multi-purification routes to a high-efficiency Negishi coupling process directly impacts the cost of goods sold (COGS). By eliminating the need for complex starting materials like Boc-L-2,6-dimethyl-tyrosine, the raw material costs are significantly reduced. Moreover, the ability to telescope multiple steps without intermediate isolation reduces solvent usage, energy consumption, and labor hours. This operational efficiency translates into substantial cost savings that can be passed down the supply chain. The robustness of the process also means fewer batch failures and more consistent delivery schedules, addressing a major pain point in the sourcing of critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts in certain steps and the reduction in purification cycles lead to a leaner manufacturing process. The high atom economy of the Negishi coupling ensures that a greater proportion of raw materials end up in the final product, minimizing waste disposal costs. Additionally, the use of common solvents like DMF and methanol simplifies solvent recovery and recycling protocols. These factors combined create a manufacturing profile that is inherently more cost-effective than legacy methods, allowing for competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as simple amino acid derivatives and commercially available aryl iodides mitigates the risk of supply disruptions. Unlike routes dependent on specialized, low-volume chiral pool materials, this synthesis can be scaled using commodity chemicals. The high overall yield of over 46 percent ensures that production targets can be met with less feedstock, providing a buffer against raw material price volatility. This reliability is essential for maintaining continuous API production lines and meeting regulatory commitments.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, featuring reactions that are exothermic but manageable under standard industrial conditions. The reduction in the number of purification steps significantly lowers the volume of organic waste generated, aligning with green chemistry principles. The avoidance of harsh reagents and the use of catalytic amounts of palladium further enhance the environmental profile of the synthesis. This makes the process not only economically attractive but also compliant with increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and experimental data presented in the patent documentation. Understanding these nuances is vital for stakeholders evaluating the feasibility of adopting this technology for their own production needs. The answers reflect the empirical results obtained during the patent examples, providing a realistic expectation of performance.
Q: What is the overall yield advantage of this new Negishi coupling route?
A: The patented process achieves an overall yield of not less than 46.7% from compound 3, significantly outperforming prior art routes which often struggle with yields around 20% due to side reactions and purification losses.
Q: How does this method improve chiral purity compared to conventional synthesis?
A: By utilizing a specific amide protection strategy on the carboxyl group during the Negishi reaction, the method prevents the damage of the Boc protecting group, thereby maintaining high optical purity with ee values reaching 100% in optimized embodiments.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability, featuring one-pot reactions that minimize intermediate isolation steps. Only three out of six steps require independent purification, drastically reducing solvent usage and processing time for ton-grade manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-tert-butyloxycarbonylamino-3-(4-carbamoyl-2,6-dimethylphenyl)propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications like Eluxadoline. Our team of expert chemists has extensively analyzed the technological potential of the Negishi coupling route described in CN110092735B. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via HPLC, to guarantee that every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-efficient but also technically robust, ensuring the long-term success of your pharmaceutical formulations.
