Advanced Synthesis of Nitroacetophenone: A Scalable Route for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Nitroacetophenone: A Scalable Route for High-Purity Pharmaceutical Intermediates
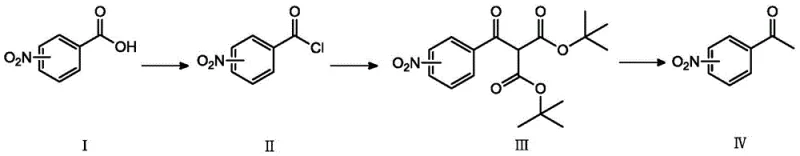
The global demand for high-purity nitroacetophenone derivatives continues to surge, driven by their critical role as key building blocks in the synthesis of complex active pharmaceutical ingredients (APIs) such as linagliptin, epinephrine, and chloramphenicol. Traditional manufacturing routes have long been plagued by severe limitations, including harsh reaction conditions, low selectivity, and significant environmental burdens. However, a groundbreaking technical disclosure found in patent CN109232259B introduces a transformative three-step synthetic strategy that fundamentally alters the production landscape. This novel methodology utilizes nitrobenzoic acid as a stable starting material, employing a sequence of acyl chlorination, condensation with di-tert-butyl malonate, and controlled hydrolysis to achieve superior yields ranging from 75% to 85%. For R&D directors and procurement specialists alike, this patent represents a pivotal shift away from dangerous nitration processes and expensive metal-catalyzed oxidations, offering a robust pathway for the reliable supply of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitroacetophenone has relied heavily on two primary methodologies, both of which present substantial operational and economic challenges for large-scale manufacturers. The first conventional route involves the direct nitration of acetophenone using a mixture of concentrated sulfuric and nitric acids at cryogenic temperatures, typically between -20°C and -15°C. This process is inherently hazardous due to the exothermic nature of nitration and generates a complex mixture of ortho-, meta-, and para-isomers that are notoriously difficult to separate, leading to significant product loss and extensive wastewater treatment requirements. The second traditional approach utilizes the oxidation of nitroethylbenzene mediated by composite transition metal catalysts. While this avoids mixed acids, it introduces a different set of problems: the catalysts are often multi-step preparations with prohibitive costs, and the removal of trace heavy metals from the final API intermediate requires additional, costly purification steps to meet stringent regulatory standards. These legacy methods result in inconsistent batch quality and inflated production costs, creating bottlenecks for supply chain reliability.
The Novel Approach
In stark contrast to these archaic techniques, the method disclosed in CN109232259B leverages a sophisticated acylation-condensation strategy that operates under remarkably mild conditions. By converting nitrobenzoic acid into its corresponding acid chloride and subsequently reacting it with di-tert-butyl malonate, the process bypasses the need for cryogenic cooling or precious metal catalysts entirely. The reaction proceeds efficiently at moderate temperatures of 55-65°C, utilizing common organic solvents such as toluene and ethyl acetate which are easily recovered and recycled. This approach not only simplifies the operational workflow but also drastically reduces the generation of hazardous waste, aligning perfectly with modern green chemistry principles. The strategic choice of reagents ensures high conversion rates and minimizes the formation of difficult-to-remove impurities, thereby streamlining the downstream purification process and enhancing the overall economic viability of the synthesis.
Mechanistic Insights into Di-tert-butyl Malonate Condensation
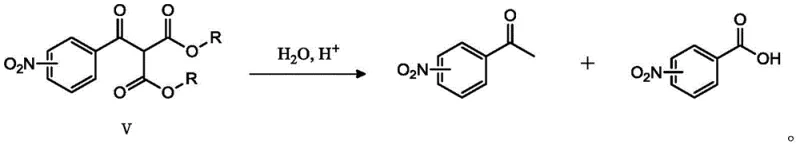
The core innovation of this synthetic route lies in the specific selection of di-tert-butyl malonate as the nucleophile, a decision grounded in profound mechanistic advantages over traditional diethyl or dimethyl malonates. In standard malonic ester syntheses, the hydrolysis of the intermediate diester often leads to a mixture of the desired ketone and the starting carboxylic acid due to competing reaction pathways. However, the inventors discovered that when the ester groups are bulky tert-butyl moieties, the hydrolysis mechanism shifts decisively. Under acidic conditions, the tert-butyl esters undergo cleavage to form unstable intermediate species that rapidly decarboxylate to yield the target nitroacetophenone as the dominant product. This steric effect effectively suppresses the reversion to nitrobenzoic acid, which is the primary side reaction observed when primary alkyl esters are used. This mechanistic nuance is critical for achieving the reported high yields and purity levels without requiring complex chromatographic separations.
Furthermore, the inclusion of inorganic salts, specifically calcium chloride, during the condensation step plays a vital role in driving the reaction equilibrium forward. The calcium ions likely coordinate with the carbonyl oxygens, activating the electrophilic acid chloride and stabilizing the transition state, which accelerates the reaction rate significantly. Additionally, the use of a catalytic amount of DMF in the initial acyl chlorination step facilitates the formation of the Vilsmeier-Haack intermediate, ensuring rapid and complete conversion of the nitrobenzoic acid to the acid chloride. This level of control over the reaction kinetics allows for the direct use of the crude acid chloride in the subsequent step without rigorous purification, saving both time and solvent. The cumulative effect of these mechanistic optimizations is a process that is not only chemically elegant but also industrially robust, capable of delivering consistent quality across multiple batches.
How to Synthesize Nitroacetophenone Efficiently
Implementing this advanced synthesis protocol requires precise adherence to the reaction parameters outlined in the patent to ensure optimal results. The process begins with the activation of the nitrobenzoic acid, followed by the crucial coupling with the malonate derivative, and concludes with a controlled hydrolytic workup. Each step has been engineered to maximize atom economy while minimizing safety risks. Operators should note that the temperature control during the hydrolysis phase is particularly important; refluxing in a mixture of water, acetic acid, and a catalytic amount of sulfuric acid ensures complete deprotection and decarboxylation. For a comprehensive understanding of the specific molar ratios, solvent volumes, and workup procedures required to replicate these results in a pilot or production setting, please refer to the standardized technical guide below.
- Convert nitrobenzoic acid to nitrobenzoyl chloride using thionyl chloride and a catalytic amount of DMF in toluene at reflux.
- React the crude acyl chloride with di-tert-butyl malonate in ethyl acetate using triethylamine and calcium chloride as promoters at 60°C.
- Hydrolyze the resulting diester intermediate in a mixture of water, acetic acid, and sulfuric acid under reflux to yield the final ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic benefits that extend far beyond simple chemical yield. The elimination of expensive transition metal catalysts and the avoidance of hazardous mixed-acid nitration directly translate to a significantly reduced cost of goods sold (COGS). By utilizing commodity chemicals like nitrobenzoic acid and di-tert-butyl malonate, manufacturers can secure raw material supply chains that are less susceptible to geopolitical volatility or specialized vendor bottlenecks. Furthermore, the mild reaction conditions reduce energy consumption associated with cryogenic cooling or high-pressure oxidation, contributing to lower utility costs and a smaller carbon footprint. These factors combine to create a more resilient and cost-effective supply chain for high-purity nitroacetophenone.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the removal of precious metal catalysts and the simplification of the purification train. Traditional oxidation methods require costly catalysts and extensive metal scavenging steps to meet ppm-level impurity specifications for pharmaceutical use. By replacing this with an organic condensation route, the process eliminates the capital expenditure for metal removal resins and the operational cost of catalyst replenishment. Additionally, the ability to recycle solvents like toluene and ethyl acetate further drives down variable costs, making the overall manufacturing process substantially more economical compared to legacy nitration or oxidation technologies.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as nitrobenzoic acid and malonic acid derivatives are widely available bulk chemicals with stable global supply networks. Unlike specialized oxidants or custom-synthesized catalysts that may have long lead times, these inputs can be procured from multiple qualified vendors, reducing the risk of production stoppages due to material shortages. The robustness of the chemistry also means that production schedules are less likely to be disrupted by batch failures or extended reaction times, ensuring a steady flow of intermediates to downstream API synthesis lines.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method offers a distinct advantage by avoiding the generation of large volumes of acidic wastewater typical of mixed-acid nitration. The waste streams generated are primarily organic and saline, which are easier and cheaper to treat in standard effluent treatment plants. This compliance ease facilitates faster regulatory approvals for new manufacturing sites and reduces the liability associated with hazardous chemical handling. The scalability is further supported by the use of standard glass-lined or stainless steel reactors, allowing for seamless technology transfer from laboratory scale to multi-ton commercial production without the need for specialized high-pressure equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitroacetophenone synthesis route. These insights are derived directly from the experimental data and technical disclosures within patent CN109232259B, providing clarity on the practical application of this technology. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: Why is di-tert-butyl malonate preferred over diethyl malonate in this synthesis?
A: Di-tert-butyl malonate prevents the formation of nitrobenzoic acid as a major byproduct during hydrolysis. The steric bulk of the tert-butyl group favors decarboxylation to the ketone (nitroacetophenone) rather than reversion to the starting acid, significantly improving yield and purity.
Q: What are the safety advantages of this method compared to traditional nitration?
A: This method avoids the use of hazardous mixed acids (sulfuric/nitric acid) typically required for direct nitration of acetophenone. By starting with nitrobenzoic acid, the process eliminates the risks associated with exothermic nitration reactions and simplifies waste treatment.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates under mild conditions (55-65°C) and uses common solvents like toluene and ethyl acetate. The absence of expensive transition metal catalysts and the simplicity of the workup make it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the methodology described in CN109232259B and possesses the technical capability to execute this three-step synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of nitroacetophenone meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain for nitroacetophenone and related intermediates. By leveraging our expertise in process optimization, we can help you achieve significant efficiencies and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless partnership for your upcoming development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
