Advanced Catalytic Synthesis of Benzimidazole Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of Benzimidazole Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, cost-effective, and environmentally benign synthetic routes for critical heterocyclic scaffolds. Patent CN109265403B introduces a transformative methodology for the synthesis of benzimidazole and its derivatives, utilizing imidazole hydrochloride as a highly efficient organocatalyst. This innovation addresses long-standing challenges in the cyclization of o-phenylenediamines with formamide derivatives, offering a pathway that eliminates the need for harsh mineral acids or expensive transition metal complexes. By leveraging this novel catalytic system, manufacturers can achieve exceptional yields and purity profiles while significantly simplifying downstream processing. The core reaction involves the condensation of substituted o-phenylenediamines with DMF or its derivatives, facilitated by a catalytic amount of imidazole hydrochloride, resulting in the formation of the benzimidazole core with remarkable selectivity.
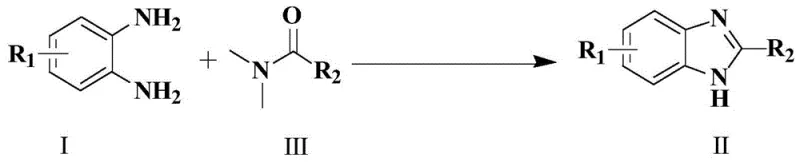
This technological breakthrough is particularly relevant for the production of high-purity pharmaceutical intermediates, where impurity profiles and process robustness are paramount. The method described in the patent not only streamlines the synthetic workflow but also enhances the economic viability of producing these valuable heterocycles. For R&D teams focused on process development, the ability to operate under relatively mild thermal conditions without the requirement for high-pressure autoclaves represents a significant safety and operational advantage. Furthermore, the broad substrate scope demonstrated in the patent data suggests that this protocol is versatile enough to handle a wide array of substitution patterns, making it a reliable solution for diverse drug discovery and development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole derivatives has relied heavily on traditional protocols that often involve severe reaction conditions and hazardous reagents. Conventional methods typically utilize strong mineral acids such as concentrated hydrochloric acid or polyphosphoric acid to drive the cyclization of o-phenylenediamines with carboxylic acids or aldehydes. These processes frequently necessitate elevated temperatures and extended reaction times, which can lead to thermal degradation of sensitive substrates and the formation of complex impurity profiles that are difficult to separate. Additionally, the corrosive nature of these acidic media imposes stringent requirements on reactor materials, increasing capital expenditure and maintenance costs for manufacturing facilities. Alternative oxidative cyclization methods using aldehydes often suffer from poor atom economy and the generation of stoichiometric waste, while transition metal-catalyzed routes introduce the risk of heavy metal contamination, necessitating costly purification steps to meet regulatory limits for pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN109265403B employs imidazole hydrochloride as a mild yet potent organocatalyst to activate DMF and its derivatives for cyclization. This approach effectively bypasses the need for corrosive strong acids or expensive metal catalysts, thereby reducing the environmental footprint and operational hazards associated with benzimidazole production. The reaction proceeds efficiently at temperatures between 120°C and 140°C, which are manageable in standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment. By avoiding the use of dichloromethane and other chlorinated solvents often found in older protocols, this new method aligns better with modern green chemistry principles and regulatory expectations regarding solvent residues. The result is a cleaner reaction profile that facilitates easier isolation and purification, ultimately leading to higher overall process efficiency and reduced production costs for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique ability of imidazole hydrochloride to activate the amide carbonyl of DMF towards nucleophilic attack by the diamine. Mechanistically, the proton from the imidazole hydrochloride likely interacts with the oxygen of the DMF carbonyl group, increasing its electrophilicity and facilitating the initial condensation with one of the amino groups of the o-phenylenediamine. This activation lowers the energy barrier for the formation of the amidine intermediate, which subsequently undergoes intramolecular cyclization and dehydration to form the aromatic benzimidazole ring. The presence of the imidazole moiety may also assist in proton transfer steps during the dehydration process, ensuring smooth progression to the final product without the accumulation of stable intermediates that could lead to side reactions. This subtle yet powerful catalytic effect allows the reaction to proceed with high turnover numbers, meaning that only a small catalytic loading (0.1 to 0.3 equivalents) is required to drive the transformation to completion.
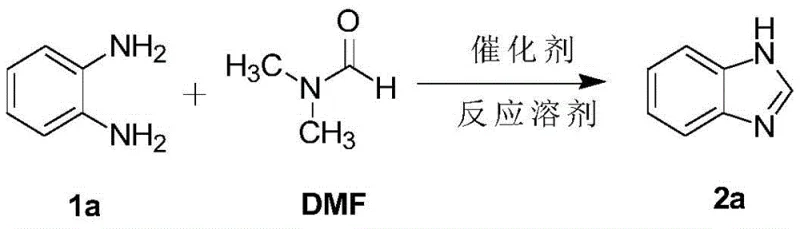
From an impurity control perspective, the mildness of this catalytic system is a distinct advantage for maintaining product integrity. Harsh acidic conditions often promote polymerization or decomposition of the starting diamines, especially when electron-rich substituents are present on the aromatic ring. However, the imidazole hydrochloride system demonstrates excellent chemoselectivity, tolerating a wide range of functional groups including halogens, nitro groups, and alkyl chains without inducing unwanted side reactions. The patent data indicates that even sensitive substrates like 5-bromo-o-phenylenediamine react cleanly to provide the corresponding benzimidazole in high yield, suggesting that the catalyst does not promote dehalogenation or other degradation pathways. This high level of selectivity ensures that the final crude product contains fewer by-products, simplifying the purification workload and improving the overall mass balance of the manufacturing process, which is critical for cost reduction in API manufacturing.
How to Synthesize Benzimidazole Efficiently
The practical implementation of this synthesis is straightforward and relies on readily available starting materials and simple operational procedures. The general protocol involves mixing the substituted o-phenylenediamine with DMF (acting as both solvent and reagent) and a catalytic amount of imidazole hydrochloride. The mixture is then heated to the optimal temperature range, monitored by TLC until conversion is complete, followed by a standard aqueous workup. This simplicity makes the technology highly accessible for process chemists looking to implement robust manufacturing routes. The detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Combine o-phenylenediamine derivative, imidazole hydrochloride catalyst (0.1-0.3 eq), and DMF solvent in a reaction vessel.
- Heat the mixture to 120-140°C and stir for 2-6 hours until TLC indicates complete conversion.
- Quench with water, extract with ethyl acetate, wash organic layers, dry over sodium sulfate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic method offers tangible benefits that extend beyond mere chemical yield. The elimination of corrosive concentrated hydrochloric acid and heavy metal catalysts significantly reduces the complexity of waste treatment and disposal, leading to lower environmental compliance costs. Furthermore, the use of DMF as a dual-purpose solvent and reagent simplifies the bill of materials, reducing the number of SKUs that need to be sourced and managed. The patent documentation explicitly highlights a potential cost reduction, noting that while current market prices for benzimidazole are approximately 120,000 RMB/ton, this inventive method could potentially control production costs around 40,000 RMB/ton, representing a substantial economic advantage. This dramatic improvement in cost structure is driven by the high efficiency of the catalyst, the reduced reaction time, and the simplified downstream processing requirements.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the replacement of stoichiometric amounts of expensive or hazardous reagents with a catalytic quantity of inexpensive imidazole hydrochloride. By avoiding the use of precious metal catalysts or large volumes of corrosive acids, the raw material costs are significantly lowered, and the lifespan of production equipment is extended due to reduced corrosion. Additionally, the high yields reported (often exceeding 90%) minimize the loss of valuable starting materials, directly improving the gross margin of the final product. The simplified workup procedure, which avoids complex extraction or neutralization steps associated with strong acids, further reduces labor and utility costs per batch.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF and o-phenylenediamine ensures a stable and resilient supply chain, as these materials are widely available from multiple global suppliers. Unlike specialized transition metal catalysts which may face supply bottlenecks or price volatility, imidazole hydrochloride is a bulk chemical with consistent availability. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable, with the patent reporting successful execution on a 20g scale with maintained efficiency, indicating a clear path to multi-kilogram and ton-scale production. The absence of high-pressure requirements (unlike some CO2-mediated methods requiring 5MPa) allows the reaction to be run in standard atmospheric reactors, reducing capital investment barriers for scale-up. From an environmental standpoint, the avoidance of chlorinated solvents like dichloromethane and the reduction of acidic waste streams align with increasingly strict global environmental regulations, mitigating the risk of regulatory shutdowns or fines. This green profile enhances the long-term sustainability of the supply chain and improves the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows.
Q: What are the advantages of using imidazole hydrochloride over traditional strong acid catalysts?
A: Unlike traditional methods requiring corrosive concentrated hydrochloric acid or polyphosphoric acid which demand high temperatures and specialized equipment, imidazole hydrochloride operates under milder conditions (120-140°C) with excellent functional group tolerance and avoids equipment corrosion issues.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent demonstrates successful scalability up to 20g scale with maintained high yields (90%), and the process avoids harsh conditions like 5MPa pressure or 150°C extremes, making it highly suitable for commercial scale-up.
Q: Does this method tolerate sensitive functional groups like halogens?
A: The method exhibits excellent functional group tolerance, successfully accommodating electron-withdrawing groups such as bromine and chlorine, as well as electron-donating groups like methyl and methoxy, without significant loss in yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the imidazole hydrochloride catalyzed route are successfully translated into robust industrial processes. We are committed to delivering high-purity benzimidazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex heterocyclic chemistry with precision, guaranteeing supply continuity for your most demanding projects.
We invite you to collaborate with us to leverage this cost-effective synthesis technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods sold.
