Advanced Nickel-Catalyzed Synthesis for High-Purity Benzimidazole and Quinazoline Intermediates
Introduction to Green Heterocyclic Synthesis
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for constructing privileged scaffolds such as benzimidazoles and quinazolines, which serve as critical cores in numerous active pharmaceutical ingredients (APIs). Patent CN109970659B introduces a groundbreaking methodology that leverages a nitrogen-doped hierarchical porous biomass-based carbon material-supported nickel catalyst to achieve oxidative coupling dehydrogenation. This innovation represents a paradigm shift from traditional noble metal catalysis to earth-abundant transition metal systems, addressing both economic and environmental imperatives in modern chemical manufacturing. By utilizing a one-pot synthetic strategy, this technology eliminates the need for intermediate isolation, thereby streamlining the production workflow and enhancing overall process efficiency for reliable pharmaceutical intermediate supplier operations.
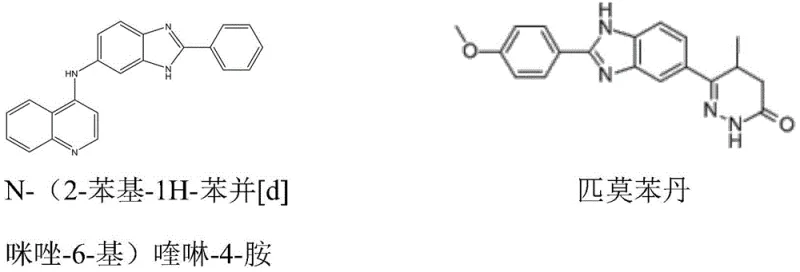
The significance of this technological advancement cannot be overstated, as benzimidazole and quinazoline derivatives are ubiquitous in medicinal chemistry, exhibiting a wide spectrum of biological activities including anticancer, anti-inflammatory, and antihypertensive properties. The ability to synthesize these complex heterocycles using inexpensive raw materials like alcohols and diamines, coupled with a reusable heterogeneous catalyst, offers a compelling value proposition for large-scale industrial applications. This approach not only aligns with the principles of green chemistry by minimizing waste generation but also provides a robust platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring a stable supply chain for downstream drug development projects globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole and quinazoline compounds has relied heavily on methods that suffer from significant operational and economic drawbacks, limiting their utility in large-scale manufacturing environments. Traditional protocols often necessitate the use of expensive and scarce noble metal catalysts such as iridium, platinum, or palladium, which drastically inflate the raw material costs and introduce severe challenges regarding residual metal removal in the final product. Furthermore, many conventional routes require harsh reaction conditions, including high temperatures and pressures, or the use of stoichiometric amounts of toxic oxidants and strong bases, leading to poor atom economy and substantial waste generation. These factors collectively contribute to extended lead times for high-purity pharmaceutical intermediates and complicate the regulatory approval process due to stringent impurity control requirements.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a specially engineered nickel catalyst supported on a nitrogen-doped porous carbon matrix derived from renewable biomass sources. This system operates under markedly milder conditions, typically between 50°C and 150°C, and employs molecular oxygen from the air as the terminal oxidant, producing only water as a byproduct. The heterogeneous nature of the catalyst allows for straightforward separation via filtration, facilitating catalyst recycling and reuse without significant loss of catalytic activity over multiple cycles. This one-pot oxidative coupling dehydrogenation strategy effectively merges the activation of inert alcohol substrates with cyclization, bypassing the need for pre-functionalized aldehydes and reducing the number of unit operations required for cost reduction in API manufacturing.
Mechanistic Insights into Ni-Catalyzed Oxidative Coupling Dehydrogenation
The core of this innovative synthesis lies in the unique capability of the nickel nanoparticles dispersed on the nitrogen-doped carbon support to facilitate the dehydrogenation of alcohols into reactive carbonyl intermediates. The nitrogen doping within the carbon lattice plays a crucial role in stabilizing the nickel species and preventing agglomeration, thereby maintaining a high density of active sites throughout the reaction course. Once the alcohol is oxidized to an aldehyde in situ, it undergoes condensation with the amine groups of the o-phenylenediamine or 2-aminobenzylamine substrates to form an imine intermediate. Subsequent intramolecular cyclization and further dehydrogenation steps yield the aromatic benzimidazole or quinazoline ring system, driven by the thermodynamic stability of the final aromatic product and the continuous removal of hydrogen.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions associated with over-oxidation or polymerization, which are common pitfalls in homogeneous catalytic systems. The hierarchical pore structure of the biomass-derived carbon support ensures efficient mass transfer of reactants to the active metal sites while restricting the formation of bulky byproducts that could otherwise foul the catalyst surface. This precise control over the reaction environment results in high selectivity for the target heterocyclic products, simplifying downstream purification processes and ensuring that the final material meets the stringent purity specifications required for pharmaceutical applications. The robustness of the catalyst against air and moisture further enhances its practical utility in diverse manufacturing settings.
How to Synthesize Benzimidazole and Quinazoline Efficiently
The implementation of this synthesis route is designed to be operationally simple, requiring standard laboratory or plant equipment without the need for specialized high-pressure reactors or inert atmosphere gloveboxes. The process begins with the combination of the diamine substrate, the alcohol coupling partner, the supported nickel catalyst, and a base such as potassium tert-butoxide in a suitable solvent like toluene. Detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometric ratios and thermal profiles optimized to maximize yield and minimize reaction time. This streamlined protocol enables rapid screening of substrate scope and facilitates the quick transition from gram-scale optimization to kilogram-scale production campaigns.
- Mix o-phenylenediamine or 2-aminobenzylamine with the corresponding alcohol, supported nickel catalyst, toluene solvent, and potassium tert-butoxide in a sealed reactor.
- Heat the reaction mixture to a temperature between 50°C and 150°C and maintain for 4 to 24 hours to allow oxidative coupling dehydrogenation to proceed.
- Cool the reaction to room temperature, filter off the heterogeneous catalyst, and purify the filtrate via silica gel column chromatography to isolate the target heterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology translates into tangible strategic benefits that extend far beyond simple unit price reductions. By replacing precious metals with abundant nickel and utilizing biomass-derived supports, the raw material cost structure is fundamentally altered, decoupling production economics from the volatile markets of rare earth elements. The elimination of expensive ligands and stoichiometric oxidants further contributes to substantial cost savings, while the simplified workup procedure reduces solvent consumption and energy usage associated with distillation and purification steps. These efficiencies collectively enhance the margin profile of the final intermediates, allowing for more competitive pricing strategies in the global marketplace.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with a non-precious nickel system removes the necessity for costly metal scavenging resins and complex purification trains designed to meet ppm-level metal limits. This qualitative shift in process design inherently lowers the cost of goods sold (COGS) by reducing both material inputs and processing time. Additionally, the ability to recycle the heterogeneous catalyst multiple times without regeneration significantly amortizes the catalyst cost over a larger volume of production, driving down the effective cost per kilogram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like nickel and renewable biomass precursors mitigates the geopolitical and supply risks associated with sourcing platinum group metals from limited geographic regions. The robustness of the catalyst formulation ensures consistent batch-to-batch performance, reducing the likelihood of production delays caused by catalyst failure or variability. This stability is critical for maintaining continuous supply lines to downstream customers, ensuring that critical drug substances are available without interruption during clinical trials or commercial launch phases.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the generation of hazardous waste streams, aligning with increasingly strict environmental regulations and corporate sustainability goals. The process generates water as the primary byproduct, simplifying effluent treatment requirements and reducing the environmental footprint of the manufacturing site. Furthermore, the mild reaction conditions and lack of high-pressure requirements make the technology highly scalable, allowing for seamless translation from pilot plant studies to multi-ton commercial production facilities with minimal engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis platform. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Can the nickel catalyst be recycled without losing activity?
A: Yes, the patent demonstrates that the nitrogen-doped porous carbon-supported nickel catalyst maintains high activity and stability over at least five consecutive recycling runs without significant deactivation, ensuring consistent production quality.
Q: Does this method avoid heavy metal contamination in the final API?
A: The use of a heterogeneous supported catalyst allows for simple filtration to remove the metal species from the reaction mixture, significantly reducing the risk of residual nickel contamination compared to homogeneous noble metal systems.
Q: What is the substrate scope for this synthesis method?
A: The method exhibits broad substrate tolerance, successfully converting various substituted o-phenylenediamines and benzyl alcohols containing electron-donating or electron-withdrawing groups into the corresponding benzimidazoles and quinazolines with high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole and Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology in reshaping the landscape of heterocyclic intermediate production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzimidazole or quinazoline intermediate meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality chemical solutions that drive your success in the competitive pharmaceutical market.
