Advanced Chiral Spiro Aminophosphine Ligands for Industrial Asymmetric Hydrogenation
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in Chinese Patent CN110724164B, which discloses a novel class of 3-substituted chiral spiro aminophosphine ligands on a pyridine ring. This technology represents a significant leap forward in the design of tridentate ligands, specifically engineered to overcome the limitations of previous generations in asymmetric catalytic hydrogenation. By integrating a chiral spiro indane skeleton with a strategically substituted pyridine group, these ligands facilitate the construction of quaternary carbon stereocenters with unprecedented efficiency. For R&D directors and process chemists, this patent offers a robust solution for synthesizing optically active beta-aryl fatty acids and their derivatives, which are critical scaffolds in numerous bioactive molecules. The ability to achieve such high levels of stereocontrol using earth-abundant or precious metal complexes underscores the practical value of this innovation for modern fine chemical manufacturing.
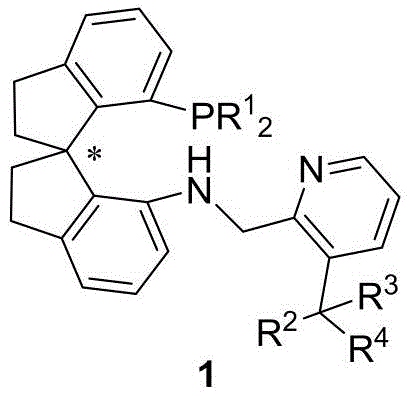
Historically, the synthesis of chiral compounds containing arylalkyl-substituted tertiary carbon centers at the beta-position has been a formidable challenge. Conventional methods often relied on the asymmetric hydrogenation of alpha,beta-unsaturated carboxylic acids or ester derivatives. However, these traditional approaches suffer from significant drawbacks that hinder their industrial applicability. The substrate scope is frequently narrow, often restricted to specific Z or E configurations, and the reaction conditions can be excessively harsh, requiring hydrogen pressures ranging from 50 to 100 atm. Furthermore, these legacy methods typically demand high catalyst loadings, often exceeding 1 mol%, which escalates production costs due to the expense of precious metals like iridium. In contrast, the novel approach detailed in the patent utilizes beta-arylalkylene malonates as substrates, which do not possess the restrictive Z or E configuration issues. This new methodology operates under much milder conditions and achieves remarkable turnover numbers, thereby addressing the critical pain points of cost, safety, and versatility that have long plagued the synthesis of these valuable chiral building blocks.
The mechanistic superiority of this technology lies in the precise engineering of the ligand's steric environment. The core innovation involves the introduction of sterically significant substituents at the 3-position of the pyridine ring within the spiro aminophosphine framework. When coordinated with an iridium salt, this tridentate ligand forms a highly rigid and chiral pocket around the metal center. This specific geometry is crucial for effective chirality transfer during the catalytic cycle. The bulky groups at the pyridine 3-position act as a stereochemical gatekeeper, directing the approach of the substrate and ensuring that hydride transfer occurs exclusively from the desired face. This results in exceptional enantioselectivity, with reported values reaching up to 99% ee. Moreover, the stability of the resulting iridium complex allows for sustained catalytic activity over extended periods, contributing to the observed high turnover numbers. This level of control is essential for minimizing the formation of unwanted diastereomers and enantiomers, simplifying downstream purification processes and ensuring the final product meets stringent regulatory purity specifications.
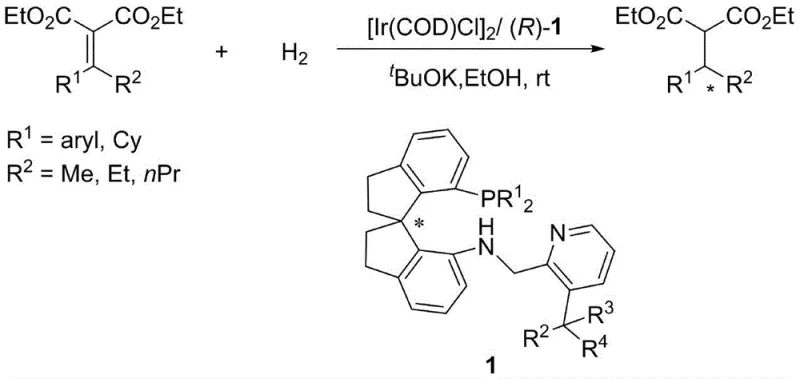
For process development teams looking to implement this technology, the synthesis of the ligand itself is designed for operational simplicity and scalability. The preparation involves a straightforward reductive amination between a chiral spiro indane amine precursor and a substituted pyridine aldehyde. This reaction proceeds efficiently in common organic solvents such as dichloroethane or alcohols, utilizing mild reducing agents like sodium triacetoxyborohydride. The robustness of this synthetic route means that the ligand can be produced in multi-kilogram quantities without requiring exotic reagents or cryogenic conditions. Once synthesized, the ligand is readily complexed with iridium precursors to generate the active catalyst in situ. This ease of preparation, combined with the high performance of the resulting catalyst, makes it an attractive candidate for integration into existing manufacturing workflows. The following section outlines the standardized procedural steps derived from the patent data to guide your laboratory or pilot plant operations.
From a commercial and supply chain perspective, the adoption of this ligand technology offers transformative advantages. The most immediate benefit is the drastic reduction in manufacturing costs associated with catalyst consumption. Because the system supports turnover numbers as high as 19,000, the required loading of the expensive iridium catalyst can be reduced to trace levels, sometimes as low as 0.005 mol%. This directly translates to substantial cost savings in raw materials, making the production of high-value chiral intermediates more economically viable. Additionally, the mild reaction conditions—often proceeding at room temperature and moderate hydrogen pressures—enhance supply chain reliability by reducing the need for specialized high-pressure equipment and lowering energy consumption. This operational flexibility allows for faster batch turnover and reduces the risk of production delays caused by equipment maintenance or safety incidents. Furthermore, the atom-economic nature of the hydrogenation reaction aligns perfectly with modern environmental compliance standards, minimizing waste generation and simplifying the handling of byproducts. These factors collectively contribute to a more resilient and sustainable supply chain for critical pharmaceutical ingredients.
Understanding the specific technical nuances of this technology is vital for making informed procurement and development decisions. The following frequently asked questions address common inquiries regarding the performance, scope, and implementation of these chiral ligands. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for your strategic planning.
- Condense the chiral spiro indane amine precursor with a substituted pyridine aldehyde in an organic solvent like DCE at elevated temperatures.
- Perform reductive amination using a mild reducing agent such as sodium triacetoxyborohydride to form the secondary amine linkage.
- Purify the resulting ligand via silica gel column chromatography to ensure high optical purity suitable for catalytic applications.
Frequently Asked Questions (FAQ)
Q: What distinguishes this 3-substituted ligand from conventional SpiroPAP ligands?
A: The introduction of sterically significant substituents at the 3-position of the pyridine ring provides enhanced chirality transfer control, resulting in superior enantioselectivity (up to 99% ee) compared to earlier generations.
Q: How does this technology impact catalyst loading costs?
A: The ligand enables exceptionally high turnover numbers (TON up to 19,000), allowing for drastically reduced catalyst loading (as low as 0.005 mol%), which significantly lowers the cost associated with precious iridium metals.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process operates under mild reaction conditions (room temperature to 80°C, moderate hydrogen pressure) and utilizes robust substrates like beta-arylalkylene malonates, making it highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Aminophosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory innovations can be seamlessly transitioned to full-scale manufacturing. We are committed to delivering high-purity chiral ligands and intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of specific ligand analogues or large-scale supply of the standard compounds described in CN110724164B, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to engage with our technical procurement team to discuss how this cutting-edge hydrogenation technology can optimize your specific synthesis routes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits for your process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring that you have the reliable partner needed to bring your next-generation therapeutics to market efficiently.
