Revolutionizing Pharmaceutical Intermediate Production Through Metal-Free Halogen Bond Catalysis for High-Purity Oxazolidinones
Patent CN113072517A introduces a groundbreaking synthetic methodology for five-membered oxygen-containing heterocyclic compounds specifically targeting oxazolidinones and related structures with significant pharmaceutical relevance This innovation leverages a novel halogen bond-catalyzed [3+2] cyclization reaction between epoxides and heterocumulative dienes such as isocyanates or carbon disulfide addressing critical limitations in traditional metal-catalyzed approaches The process operates under remarkably mild conditions at 70-80°C without requiring transition metals thereby eliminating hazardous metal residues that compromise product purity in biomedical applications Furthermore the methodology achieves exceptional selectivity for single product formation avoiding the complex byproduct mixtures that characterize conventional routes This patent represents a paradigm shift in heterocyclic synthesis offering a sustainable pathway to high-value intermediates essential for next-generation drug development The strategic elimination of metal catalysts not only enhances safety profiles but also simplifies downstream purification processes directly translating to cost efficiencies for pharmaceutical manufacturers seeking reliable supply chains
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing five-membered oxygen heterocyclic compounds predominantly rely on transition metal catalysts or metal complexes which introduce significant challenges for pharmaceutical applications due to unavoidable metal residue contamination These residues necessitate extensive and costly purification protocols to meet stringent regulatory requirements for drug substances often resulting in reduced overall yields and increased production timelines Moreover conventional methods frequently operate under harsh conditions exceeding 100°C demanding specialized high-pressure equipment and elevating energy consumption substantially The inherent asymmetry of heterocumulative dienes like isocyanates and carbon disulfide leads to poor regioselectivity in metal-catalyzed systems generating multiple byproducts that complicate product isolation and diminish process efficiency Additionally the use of toxic metals such as palladium or copper creates environmental compliance burdens and supply chain vulnerabilities due to fluctuating raw material costs and geopolitical constraints These cumulative drawbacks have long hindered the commercial viability of such syntheses despite their theoretical appeal in constructing complex molecular architectures required for modern therapeutics
The Novel Approach
The patented methodology overcomes these limitations through an innovative halogen bond-catalyzed system utilizing aryl-substituted 2-halogenated imidazolium salts as non-covalent catalysts operating effectively at reduced temperatures of 70-80°C without any transition metals This organic catalytic system exploits the directional nature of carbon-halogen bonds to precisely control the cyclization pathway ensuring high regioselectivity and exclusive formation of single products without competing byproducts The mild reaction conditions eliminate the need for specialized high-pressure reactors while significantly reducing energy consumption compared to conventional methods Crucially the absence of metal components removes all concerns regarding residual contamination streamlining regulatory compliance and reducing purification costs substantially The catalysts themselves are readily prepared from inexpensive starting materials through straightforward synthetic routes with near quantitative yields enhancing overall process economics This approach represents a sustainable advancement that aligns with green chemistry principles while delivering superior performance metrics essential for pharmaceutical manufacturing scalability
Mechanistic Insights into Halogen Bond-Catalyzed [3+2] Cyclization
The catalytic mechanism centers on the electrophilic character of the halogen atom in aryl-substituted 2-halogenated imidazolium salts which forms a directional halogen bond with the nucleophilic oxygen of epoxides This interaction activates the epoxide ring toward nucleophilic attack by heterocumulative dienes such as isocyanates or carbon disulfide through a well-defined transition state where the halogen bond donor stabilizes developing negative charge during ring opening The carbon-halogen bond's anisotropic electrostatic distribution creates a sigma-hole that facilitates precise molecular recognition between catalyst and substrate ensuring regioselective attack at specific positions to form single stereoisomers This non-covalent catalytic pathway operates through a concerted mechanism that avoids high-energy intermediates responsible for side reactions in conventional systems The halogen bond's hydrophobic nature further enhances compatibility with diverse organic solvents while preventing unwanted hydrolysis pathways that could compromise product integrity during synthesis

The exceptional selectivity achieved in this process stems from the catalyst's ability to control both regiochemistry and stereochemistry through steric and electronic modulation of the transition state Unlike hydrogen bonding catalysts that often produce racemic mixtures the halogen bond's directional properties enforce specific molecular orientations that favor formation of single diastereomers without requiring additional chiral auxiliaries The absence of metal centers eliminates redox pathways that could lead to decomposition or undesired side reactions with sensitive functional groups commonly present in pharmaceutical intermediates Furthermore the catalyst system demonstrates remarkable tolerance to moisture and air compared to traditional organometallic catalysts reducing stringent handling requirements during manufacturing scale-up This combination of precise molecular control and operational robustness ensures consistent production of high-purity heterocyclic compounds meeting pharmaceutical industry specifications without complex purification steps
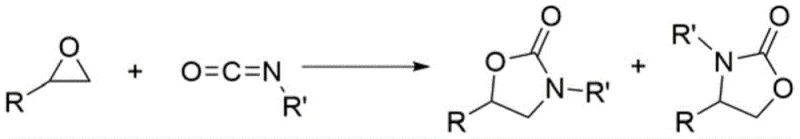
How to Synthesize Oxazolidinone Efficiently
This innovative synthetic route represents a significant advancement over conventional methodologies by enabling efficient production of high-purity oxazolidinones through a streamlined catalytic process The patented approach utilizes readily available epoxide substrates and heterocumulative dienes under mild thermal conditions eliminating the need for expensive transition metals while maintaining excellent yield and selectivity profiles Detailed operational parameters have been optimized through extensive experimentation to ensure reproducibility across different production scales with particular attention paid to solvent selection and catalyst loading ratios that maximize efficiency The following standardized procedure provides step-by-step guidance for implementing this technology in industrial settings while maintaining strict quality control standards essential for pharmaceutical manufacturing
- Dissolve epoxide substrate and catalyst in anhydrous organic solvent such as chlorobenzene under inert atmosphere while maintaining precise stoichiometric ratios.
- Add heterocumulative diene component like isocyanate and heat reaction mixture to controlled temperature range of 70-80°C for optimal cyclization kinetics.
- Purify reaction mixture using column chromatography with petroleum ether/ethyl acetate solvent system to isolate high-purity oxazolidinone product.
Commercial Advantages for Procurement and Supply Chain Teams
This novel catalytic methodology addresses critical pain points in pharmaceutical intermediate supply chains by delivering a robust manufacturing solution that enhances both cost efficiency and operational reliability The elimination of transition metals from the synthesis process removes significant regulatory hurdles associated with metal residue testing while reducing dependency on volatile commodity markets for precious metals Furthermore the simplified reaction pathway minimizes waste generation compared to conventional multi-step approaches aligning with increasingly stringent environmental regulations while lowering disposal costs These advantages collectively position this technology as an ideal solution for procurement teams seeking sustainable supply partners who can deliver consistent quality without compromising on economic viability or environmental responsibility
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated purification steps required to remove metal residues results in substantial cost savings throughout the production process By operating under milder conditions at lower temperatures energy consumption is significantly reduced while maintaining high conversion rates The simplified workup procedure avoids costly chromatographic separations needed to isolate products from complex byproduct mixtures generated by conventional methods further optimizing resource utilization without compromising product quality
- Enhanced Supply Chain Reliability: The use of commercially available starting materials and straightforward catalyst preparation ensures consistent raw material availability without exposure to geopolitical supply risks associated with rare metals The robust nature of the halogen bond catalytic system maintains performance across varying production scales enabling seamless transition from laboratory development to commercial manufacturing without reoptimization delays This reliability translates directly to predictable lead times and reduced risk of production interruptions that could impact critical drug development timelines
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to industrial production volumes while maintaining consistent product quality attributes essential for pharmaceutical applications Operating at lower temperatures reduces energy requirements and equipment stress during scale-up facilitating implementation in standard manufacturing facilities without specialized infrastructure investments The absence of toxic metal byproducts simplifies waste management procedures and aligns with green chemistry principles enhancing regulatory compliance while supporting corporate sustainability initiatives through reduced environmental footprint
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology in pharmaceutical intermediate manufacturing operations Each response is grounded in experimental data from the original patent documentation and reflects practical considerations for successful technology transfer to industrial settings
Q: How does this method eliminate metal residues in pharmaceutical intermediates?
A: The halogen bond catalysis system employs organic catalysts without transition metals ensuring no metal contamination occurs during synthesis which is critical for biomedical applications where regulatory standards strictly prohibit metal residues.
Q: What enables high selectivity in producing single five-membered oxygen heterocyclic compounds?
A: The carbon-halogen bond donor catalyst precisely directs molecular orientation during cyclization minimizing side reactions that generate complex byproduct mixtures common in conventional methods using asymmetric heterocumulative dienes.
Q: How does the reduced reaction temperature enhance commercial scalability?
A: Operating at 80°C instead of over 100°C lowers energy consumption reduces equipment stress and facilitates seamless scale-up from laboratory to industrial production volumes while maintaining consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Supplier
Our company leverages this patented halogen bond catalysis technology to deliver high-purity oxazolidinone intermediates with exceptional consistency across production scales from 100 kgs to 100 MT/annual commercial production volumes NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance Our manufacturing facilities operate under cGMP guidelines with rigorous validation protocols ensuring complete traceability and compliance with global regulatory standards required for pharmaceutical applications This technical expertise combined with our commitment to sustainable manufacturing practices positions us as an ideal partner for developing robust supply chains of complex heterocyclic compounds essential for modern drug discovery programs
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements by contacting our technical procurement team directly Request detailed COA data and route feasibility assessments demonstrating how our patented methodology can optimize your intermediate supply chain while meeting your exact quality specifications and delivery timelines
