Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their pervasive presence in bioactive molecular frameworks and functional materials. A groundbreaking development in this sector is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward by replacing cumbersome multi-step sequences with a streamlined oxidative cyclization protocol. The core innovation lies in the utilization of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) as a catalytic system to promote the coupling of trifluoroethylimide hydrazide with 2-methylquinoline. For R&D directors and procurement specialists, this patent offers a compelling value proposition: it eliminates the need for toxic heavy metal catalysts and严苛 reaction conditions, thereby facilitating easier regulatory approval and safer handling protocols. The ability to synthesize these complex scaffolds with high atom economy and structural diversity positions this method as a critical asset for developing next-generation therapeutic agents and OLED materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional literature methods typically rely on quinoline-2-formic acid as the primary starting material, necessitating a laborious five-step reaction sequence to achieve the final target molecule. This conventional pathway suffers from a dismal total yield of approximately 17%, rendering it economically unviable for large-scale applications. Furthermore, the harsh reaction conditions often required in these legacy processes demand specialized equipment capable of maintaining strict anhydrous and anaerobic environments, which significantly inflates capital expenditure and operational costs. The reliance on such inefficient routes creates bottlenecks in the supply chain, leading to extended lead times and inconsistent batch quality, which are unacceptable for modern pharmaceutical manufacturing standards where purity and reproducibility are paramount.
The Novel Approach
In stark contrast, the methodology described in patent CN113307790B introduces a paradigm shift by utilizing readily available 2-methylquinoline and trifluoroethylimide hydrazide as starting materials. This novel approach employs a TBAI/TBHP promoted oxidative cyclization that occurs in a single pot, dramatically simplifying the workflow. As illustrated in the reaction scheme below, the process bypasses the need for pre-functionalized aldehydes, generating the reactive intermediates in situ. 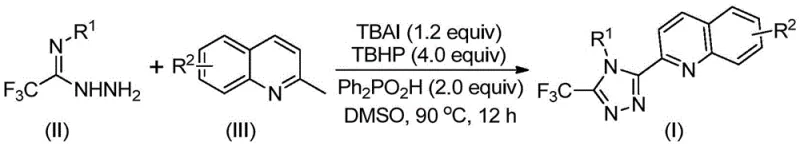 The reaction tolerates a wide range of substituents on both the aryl ring of the hydrazide and the quinoline backbone, allowing for the rapid generation of diverse libraries of compounds. Experimental data from the patent indicates that yields can reach as high as 97% under optimized conditions, a massive improvement over the 17% ceiling of traditional methods. This efficiency not only reduces raw material waste but also minimizes the environmental footprint associated with solvent usage and purification steps, aligning perfectly with green chemistry principles.
The reaction tolerates a wide range of substituents on both the aryl ring of the hydrazide and the quinoline backbone, allowing for the rapid generation of diverse libraries of compounds. Experimental data from the patent indicates that yields can reach as high as 97% under optimized conditions, a massive improvement over the 17% ceiling of traditional methods. This efficiency not only reduces raw material waste but also minimizes the environmental footprint associated with solvent usage and purification steps, aligning perfectly with green chemistry principles.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the iodide salt and the peroxide oxidant. The reaction is believed to proceed via a radical pathway where tetrabutylammonium iodide acts as a catalyst to activate the tert-butyl peroxide. Initially, the methyl group at the 2-position of the quinoline ring is oxidized to an aldehyde functionality in situ, generating 2-quinolinecarbaldehyde without the need for isolation. This transient aldehyde then undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination facilitates an intramolecular electrophilic substitution, followed by aromatization to yield the final 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole core. The inclusion of diphenylphosphoric acid serves as a crucial additive, likely stabilizing the transition states and enhancing the overall reaction kinetics. This mechanism avoids the use of transition metals like copper or palladium, which are common in cross-coupling reactions but pose significant challenges regarding residual metal limits in pharmaceutical products.
From an impurity control perspective, this metal-free mechanism is exceptionally advantageous. The absence of heavy metals eliminates the need for expensive and time-consuming scavenging steps typically required to reduce metal residues below ppm levels. Furthermore, the high selectivity of the oxidative cyclization minimizes the formation of side products, resulting in a cleaner crude reaction profile. The use of DMSO as the preferred solvent ensures excellent solubility of all reactants and intermediates, promoting homogeneous reaction conditions that further suppress the formation of oligomeric byproducts. For quality control teams, this translates to a more predictable impurity profile and simplified analytical validation, ensuring that the final active pharmaceutical ingredient (API) intermediates meet stringent global regulatory specifications with minimal downstream processing.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and industrial production. The protocol involves charging a reactor with the specified molar ratios of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent. The detailed standardized synthesis steps are provided in the guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP) aqueous solution, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization without requiring anhydrous or oxygen-free conditions.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the target 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits beyond mere chemical yield. The shift from a five-step sequence to a one-pot reaction fundamentally alters the cost structure of manufacturing these valuable intermediates. By collapsing multiple unit operations into a single vessel, the process drastically reduces labor hours, energy consumption, and solvent volumes. This consolidation directly translates to significant cost reduction in pharmaceutical intermediate manufacturing, allowing companies to improve their margins or offer more competitive pricing to downstream clients. Additionally, the elimination of toxic heavy metal catalysts removes a major regulatory hurdle, streamlining the path to market for new drug candidates that incorporate this scaffold.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the replacement of expensive, multi-step precursors with commodity chemicals like 2-methylquinoline. Since the reaction does not require precious metal catalysts, the bill of materials is significantly lowered. Furthermore, the high conversion rates observed in the patent examples mean that less raw material is wasted, maximizing the output per kilogram of input. The simplified work-up procedure, which involves basic filtration and chromatography rather than complex extractions or metal scavenging, further drives down operational expenditures. These factors combine to create a highly cost-effective production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commercially sourced reagents. Tetrabutylammonium iodide, tert-butyl peroxide, and diphenylphosphoric acid are bulk chemicals with stable global supply lines, reducing the risk of shortages that often plague specialized catalysts. The robustness of the reaction conditions, which do not demand inert atmospheres or ultra-dry solvents, means that production can be maintained even in facilities with standard infrastructure. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demands.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration in gram-scale reactions with potential for expansion to multi-kilogram batches. The use of DMSO, a solvent with well-established recovery and recycling protocols, supports sustainable manufacturing practices. Moreover, the absence of heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also aligns with the corporate sustainability goals of major multinational corporations, making it a preferred choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-formic acid and five reaction steps with a low total yield of 17%, this novel approach utilizes cheap 2-methylquinoline in a one-pot reaction achieving yields up to 97% without toxic heavy metal catalysts.
Q: Does this process require strict anhydrous or inert atmosphere conditions?
A: No, a significant operational advantage of this patent technology is that the reaction proceeds efficiently in standard organic solvents like DMSO without the need for rigorous anhydrous or oxygen-free environments, simplifying equipment requirements.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of commercially available reagents like TBAI and TBHP, combined with simple post-treatment via filtration and chromatography, makes the process highly scalable from gram-level laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO is essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from benchtop to plant. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the highest industry standards. We understand the critical nature of supply continuity and are committed to delivering high-quality intermediates that support your drug development timelines.
We invite you to leverage our technical expertise to optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this advanced metal-free synthesis can drive value for your organization. Let us be your trusted partner in navigating the complexities of modern pharmaceutical manufacturing.
