Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
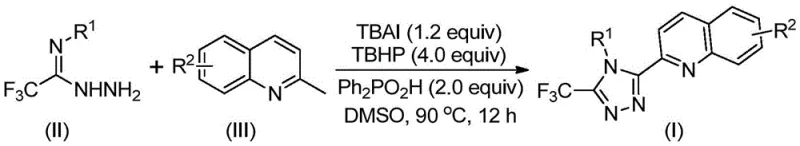
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold which is ubiquitous in bioactive molecular frameworks. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy. By leveraging a metal-free catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), the process achieves high conversion rates under relatively mild thermal conditions. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: the ability to access complex heterocyclic intermediates without the burden of heavy metal contamination or prohibitive raw material costs. The versatility of this method allows for the introduction of diverse substituents on the aryl ring, thereby expanding the chemical space available for drug discovery and functional material development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that hinder commercial viability. The prior art predominantly relies on quinoline-2-carboxylic acid as the foundational starting material. This traditional pathway is notoriously cumbersome, typically necessitating a five-step reaction sequence to arrive at the final target molecule. Furthermore, the cumulative yield of such multi-step processes is often dismally low, reported in literature to be around 17%, which is economically unsustainable for large-scale manufacturing. Beyond the poor atom economy, these conventional routes frequently demand severe reaction conditions, including stringent temperature controls and the use of hazardous reagents. The reliance on carboxylic acid derivatives also limits the structural diversity that can be achieved, as functional group tolerance is often compromised during the activation and coupling steps. For a supply chain manager, these factors translate into long lead times, high waste generation, and significant volatility in production costs, making the traditional approach unsuitable for the demands of modern API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113307790B introduces a direct and elegant solution by utilizing 2-methylquinoline and trifluoroacetimidohydrazide as the primary building blocks. This novel approach bypasses the need for pre-functionalized carboxylic acids, instead employing an oxidative cyclization strategy that constructs the triazole ring in situ. The reaction is promoted by a synergistic combination of tetrabutylammonium iodide and tert-butyl peroxide, which facilitates the transformation under heating conditions of 80 to 100°C. Crucially, this method eliminates the requirement for anhydrous or oxygen-free environments, drastically simplifying the operational protocol. The use of diphenylphosphoric acid as an additive further enhances the reaction efficiency, ensuring high yields across a broad substrate scope. This streamlined process not only reduces the number of unit operations but also significantly lowers the barrier to entry for producing these valuable intermediates, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage in terms of speed and cost.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this synthetic route lies in its sophisticated yet operationally simple mechanistic pathway, which proceeds through a radical-mediated oxidative process. Initially, the tetrabutylammonium iodide and tert-butyl peroxide system acts to oxidize the methyl group of the 2-methylquinoline substrate, effectively converting it into a reactive 2-quinolinecarbaldehyde intermediate in situ. This transient aldehyde then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone species. Following this condensation, the system promotes an oxidative iodination event, which activates the hydrazone for subsequent intramolecular electrophilic substitution. This cyclization step is critical, as it closes the five-membered triazole ring while simultaneously incorporating the trifluoromethyl group at the 5-position. The final aromatization step yields the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product. Understanding this mechanism is vital for R&D teams, as it highlights the dual role of the reagents: acting both as oxidants for C-H activation and as promoters for ring closure, thereby avoiding the need for external transition metal catalysts that often leave trace impurities.
From an impurity control perspective, this metal-free mechanism offers substantial benefits for downstream processing. Traditional transition metal-catalyzed reactions often generate complex mixtures of metal-ligand complexes and require extensive scavenging procedures to meet regulatory purity standards for pharmaceutical ingredients. In this oxidative cyclization, the byproducts are primarily organic salts and reduced forms of the peroxide, which are far easier to remove via standard aqueous workups or silica gel filtration. The absence of heavy metals means that the crude product profile is cleaner, reducing the load on purification columns and minimizing the loss of product during cleanup. Furthermore, the tolerance of the reaction to various functional groups—such as halogens, alkoxy groups, and nitro groups on the aromatic rings—suggests that the radical intermediates are sufficiently selective to avoid non-specific side reactions. This selectivity ensures that the impurity profile remains manageable even when synthesizing diverse analogues, providing a robust platform for generating libraries of high-purity triazole compounds for biological screening.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The protocol involves charging a reaction vessel with the key components: the hydrazide derivative, the methylquinoline substrate, the iodide promoter, the peroxide oxidant, and the phosphoric acid additive, all dissolved in a polar aprotic solvent such as DMSO. The mixture is then heated to a controlled temperature range, typically between 80°C and 100°C, and maintained under stirring for a duration of 8 to 14 hours to ensure full conversion of the starting materials. The simplicity of the workup procedure is a major highlight; once the reaction is deemed complete, the mixture can be filtered and directly subjected to silica gel treatment followed by column chromatography. This straightforward isolation technique underscores the practicality of the method for both small-scale discovery and larger production runs.
- Combine tetrabutylammonium iodide, tert-butyl peroxide, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in an organic solvent like DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive and scarce transition metal catalysts fundamentally alters the cost structure of the manufacturing process. By relying on commodity chemicals like tetrabutylammonium iodide and tert-butyl peroxide, the raw material costs are significantly reduced compared to precious metal-catalyzed alternatives. Moreover, the removal of heavy metals from the process flow eliminates the need for specialized metal scavengers and the associated validation testing, leading to substantial cost savings in quality control and waste disposal. The operational simplicity, characterized by the lack of need for inert atmosphere techniques (such as nitrogen blanketing or gloveboxes), further reduces capital expenditure on specialized equipment and lowers the energy consumption associated with maintaining strict anhydrous conditions. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the substitution of high-cost catalytic systems with inexpensive organic promoters. Traditional methods often rely on palladium or copper catalysts, which not only carry a high price tag but also introduce variability due to metal lot differences. By switching to an iodide/peroxide system, manufacturers can stabilize their input costs and reduce the overall bill of materials. Additionally, the high atom economy of the direct cyclization means less raw material is wasted in forming byproducts, enhancing the overall material efficiency. The simplified purification process also reduces the consumption of solvents and silica gel, further driving down the variable costs per kilogram of product. This holistic reduction in operational expenses allows for more competitive pricing in the global market for API intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents that may have long lead times or single-source suppliers. The reagents utilized in this patented method, including 2-methylquinoline and common oxidants, are widely available from multiple global chemical suppliers, mitigating the risk of supply disruptions. The robustness of the reaction conditions, which do not require ultra-dry solvents or oxygen-free environments, means that production can be sustained even if there are minor fluctuations in utility quality or facility conditions. This resilience ensures that delivery schedules for high-purity pharmaceutical intermediates can be met consistently, fostering stronger relationships with downstream pharmaceutical clients who depend on just-in-time inventory models. The ability to source materials locally in various regions further shortens the logistics chain and reduces transportation risks.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this oxidative cyclization is inherently designed for scalability. The use of DMSO as a solvent, while requiring careful handling, is well-understood in industrial settings, and the reaction exotherm can be managed effectively with standard cooling protocols. From an environmental standpoint, the avoidance of heavy metals aligns perfectly with increasingly stringent global regulations regarding metal residues in pharmaceuticals and industrial effluents. This compliance reduces the regulatory burden and the cost associated with wastewater treatment and hazardous waste disposal. The process generates fewer toxic byproducts, supporting green chemistry initiatives and improving the overall sustainability profile of the manufacturing site, which is a key metric for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The responses cover aspects ranging from substrate scope to purification requirements, ensuring a comprehensive overview of the technology's practical application in a commercial setting.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid which suffer from low yields (around 17%) and harsh multi-step conditions, this novel approach utilizes cheap 2-methylquinoline and operates under mild, metal-free conditions with significantly higher efficiency.
Q: Does this process require expensive transition metal catalysts?
A: No, the process explicitly avoids the use of toxic and expensive heavy metal catalysts. It relies on an organic catalytic system involving tetrabutylammonium iodide and tert-butyl peroxide, which simplifies purification and reduces environmental impact.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent highlights that the reaction does not require strict anhydrous or oxygen-free conditions, uses commercially available starting materials, and allows for easy scale-up from gram-scale to potential tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology disclosed in CN113307790B for the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch is free from heavy metal contaminants and adheres to the highest industry standards. Our capability to handle complex heterocyclic synthesis allows us to support your drug development pipeline with reliable, high-quality materials.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this metal-free method can optimize your specific supply chain economics. We encourage you to contact our technical procurement team today to request specific COA data for our triazole derivatives and to discuss route feasibility assessments tailored to your target molecules. Let us help you accelerate your time-to-market with a supply partner that combines chemical innovation with commercial reliability.
