Advanced Catalyst-Free Synthesis of 3-Arylpropynoic Acids for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing critical building blocks. Patent CN108558635B introduces a groundbreaking methodology for the preparation of 3-arylpropynoic acids and their corresponding ester derivatives, addressing long-standing challenges in alkyne carboxylation. This technology leverages a base-promoted reaction between phenylacetylene derivatives and carbon dioxide in dimethyl sulfoxide (DMSO) at moderate temperatures ranging from 40-70°C. Unlike traditional approaches that rely on toxic carbon monoxide or expensive transition metal catalysts, this innovation utilizes readily available inorganic weak bases such as cesium carbonate or organic superbases like DBU. The process operates under atmospheric pressure, significantly reducing equipment requirements and safety hazards associated with high-pressure gas handling. For R&D directors and procurement managers alike, this represents a paradigm shift towards greener chemistry that does not compromise on yield or substrate scope, enabling the production of high-purity pharmaceutical intermediates with exceptional atom economy.
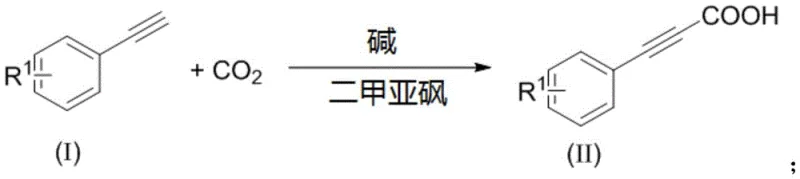
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of propiolic acid derivatives has been fraught with significant operational and economic hurdles that impact the supply chain reliability for reliable pharmaceutical intermediates supplier networks. Traditional routes often involve the addition of formaldehyde followed by oxidation, which introduces toxicity concerns and requires rigorous purification steps to remove residual aldehydes. Alternatively, carbonylation using carbon monoxide necessitates high-pressure reactors and poses severe safety risks due to the extreme toxicity of CO gas, leading to increased capital expenditure for specialized containment infrastructure. Furthermore, direct carboxylation methods developed in academic literature frequently depend on transition metal catalysts such as copper, silver, or rare earth complexes. These metal-based systems not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final product, a critical failure point for API manufacturing where strict ppm limits are enforced. Additionally, many catalytic systems require elevated CO2 pressures exceeding 10 atmospheres, limiting their scalability in standard multipurpose chemical plants and complicating the commercial scale-up of complex organic intermediates.
The Novel Approach
The methodology disclosed in CN108558635B offers a robust solution by eliminating the need for any transition metal or rare earth catalyst, thereby fundamentally altering the cost structure of production. By employing simple bases like cesium carbonate or DBU in DMSO, the reaction achieves high conversion rates under ambient pressure conditions, making it accessible for facilities without high-pressure autoclaves. This approach demonstrates excellent functional group tolerance, successfully accommodating substrates with electron-withdrawing groups such as nitro and fluoro substituents, as well as electron-donating groups like methoxy and methyl, which often deactivate metal catalysts in conventional systems. The ability to perform the reaction at temperatures between 60-70°C ensures optimal activation energy is met without thermal degradation of sensitive moieties. Moreover, the process supports a one-pot synthesis for esters, where the intermediate acid salt reacts in situ with alkyl halides or tosylates, streamlining the workflow and reducing solvent consumption. This operational simplicity translates directly into cost reduction in fine chemical manufacturing by minimizing unit operations and waste generation.
Mechanistic Insights into Base-Promoted Carboxylation
The core of this technological advancement lies in the efficient activation of the terminal alkyne C-H bond through deprotonation by a strong base, generating a nucleophilic acetylide anion stabilized by the polar aprotic solvent DMSO. In the absence of metal coordination, the cesium cation or the organic cation from DBU plays a crucial role in stabilizing the carboxylate intermediate formed upon nucleophilic attack on the electrophilic carbon of carbon dioxide. This metal-free mechanism avoids the formation of stable metal-acetylide complexes that often require harsh acidic workups or ligand exchange steps to release the product. The choice of DMSO as the solvent is critical, as comparative studies within the patent data indicate that solvents like ethanol, THF, or toluene fail to support the reaction, likely due to insufficient solvation of the ionic intermediates or competitive protonation. The reaction kinetics are optimized at 60°C, where the balance between activation energy and thermal stability yields the highest isolated yields, often exceeding 90% for a wide range of substrates. This mechanistic clarity allows process chemists to predict outcomes accurately and troubleshoot potential deviations without relying on empirical trial-and-error associated with complex catalytic cycles.
Impurity control is inherently superior in this system due to the absence of metal catalysts which can promote side reactions such as Glaser coupling or polymerization of the alkyne. The use of stoichiometric amounts of base ensures complete conversion of the starting material while minimizing the formation of oligomeric byproducts. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups like nitro or halo substituents, which might be reduced or displaced under the harsher conditions required for metal-catalyzed processes. The workup procedure involves simple acidification with hydrochloric acid, which precipitates the free acid or allows for extraction, ensuring that the final product meets stringent purity specifications required for downstream pharmaceutical applications. The robustness of this base-mediated pathway ensures consistent batch-to-batch quality, a key metric for supply chain heads managing long-term contracts for high-purity OLED material or drug precursors.
How to Synthesize 3-Arylpropynoic Acid Efficiently
To implement this synthesis effectively, operators must maintain strict anhydrous and oxygen-free conditions using an inert gas blanket such as argon, as moisture can quench the reactive acetylide species and reduce yield. The standard protocol involves charging the reactor with the base and solvent, followed by the addition of the phenylacetylene substrate and pressurization with CO2 to 1 atm. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React phenylacetylene derivatives with CO2 in DMSO using Cs2CO3 or DBU at 60-70°C under inert atmosphere.
- Acidify the reaction mixture with HCl to isolate the 3-arylpropynoic acid product.
- For esters, add alkyl halides or tosylates directly to the reaction mixture for a one-pot transformation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patent offers substantial advantages by decoupling production from the volatile pricing of precious metals and specialized ligands. The elimination of copper, silver, or rare earth catalysts removes a significant cost driver and mitigates supply chain risks associated with the geopolitical scarcity of these materials. Since the reagents involved—cesium carbonate, DBU, DMSO, and CO2—are commodity chemicals available from multiple global vendors, procurement managers can leverage competitive bidding to secure favorable pricing and ensure supply continuity. The atmospheric pressure operation reduces the need for expensive high-pressure reactors, lowering the barrier to entry for contract manufacturing organizations and allowing for faster technology transfer between sites. This flexibility enhances supply chain reliability by enabling distributed manufacturing strategies that reduce lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for costly metal scavenging resins and extensive purification steps to meet residual metal limits, resulting in significant operational savings. The one-pot procedure for ester synthesis further reduces costs by saving on solvent usage, labor hours, and equipment occupancy time compared to multi-step isolation processes. Additionally, the high atom economy of using CO2 as a C1 building block maximizes raw material efficiency, ensuring that a greater proportion of input mass is converted into saleable product. These factors combine to create a leaner manufacturing process that improves overall margin potential without sacrificing product quality.
- Enhanced Supply Chain Reliability: Relying on common inorganic bases and solvents rather than proprietary catalyst kits simplifies inventory management and reduces the risk of production stoppages due to single-source supplier issues. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, increasing the pool of qualified manufacturers capable of executing the process. This broadens the supply base and provides procurement teams with greater negotiating power and contingency options. The robustness of the method across diverse substrates means that a single platform technology can be applied to manufacture a wide portfolio of intermediates, streamlining validation and qualification efforts.
- Scalability and Environmental Compliance: Operating at atmospheric pressure and moderate temperatures significantly lowers the energy footprint of the process compared to high-pressure carbonylation methods, aligning with corporate sustainability goals and reducing utility costs. The absence of toxic carbon monoxide and formaldehyde improves workplace safety and simplifies environmental permitting, accelerating the timeline for new facility approvals. Waste streams are easier to treat as they primarily consist of inorganic salts and organic solvents that can be recovered and recycled, minimizing the environmental impact and disposal fees. This green chemistry profile enhances the brand reputation of manufacturers and meets the increasingly strict environmental, social, and governance (ESG) criteria demanded by downstream pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this base-promoted carboxylation technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process development teams.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented process eliminates the need for expensive transition metals like Copper or Silver, utilizing inexpensive inorganic or organic bases instead.
Q: What are the reaction conditions for carboxylation?
A: The reaction proceeds efficiently at atmospheric pressure (1 atm) of CO2 and moderate temperatures between 40-70°C in DMSO solvent.
Q: Can 3-arylpropynoic esters be synthesized in one pot?
A: Yes, the method allows for a one-pot synthesis where alkyl halides are added directly to the carboxylated intermediate without isolation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylpropynoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free carboxylation technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial plant. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including inert atmosphere operations and efficient solvent recovery systems, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering consistent quality and reliability, helping you secure your supply chain for critical drug substances.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your growth with efficient, high-quality chemical solutions.
