Revolutionizing Pharmaceutical Intermediate Production with Metal-Free Carboxylation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for critical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN108558635B, which details a novel preparation method for 3-arylpropynoic acids and their corresponding ester derivatives. This technology represents a paradigm shift from traditional transition-metal-catalyzed processes to a more sustainable, base-promoted carboxylation strategy. By utilizing carbon dioxide as a renewable C1 building block under atmospheric pressure, this method addresses key pain points regarding catalyst cost, heavy metal contamination, and operational safety. For R&D directors and procurement specialists alike, understanding the implications of this metal-free approach is vital for optimizing supply chains and reducing the overall cost of goods sold (COGS) in complex molecule manufacturing.
Historically, the synthesis of 3-arylpropynoic acids has relied on methodologies that present substantial logistical and economic hurdles. Conventional routes often involve the use of toxic C1 sources such as formaldehyde or carbon monoxide, which necessitate stringent safety protocols and specialized handling equipment to mitigate health risks. Alternatively, direct carboxylation methods utilizing Grignard or lithium reagents require cryogenic conditions and strictly anhydrous environments, limiting their practicality for large-scale operations due to high energy consumption and safety concerns. Furthermore, established catalytic systems frequently depend on precious or rare earth metals like copper, silver, or lanthanides. These catalysts not only inflate raw material costs but also introduce the risk of heavy metal residues in the final API, requiring expensive and time-consuming purification steps to meet regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on transition metal catalysts in existing literature creates a bottleneck for cost-effective manufacturing. For instance, copper-based systems often require complex ligand architectures to maintain activity, and silver catalysts may necessitate elevated CO2 pressures (up to 15 atmospheres) to achieve acceptable yields, demanding high-specification reactor vessels. Additionally, the sensitivity of these catalytic cycles to moisture and oxygen often leads to inconsistent batch-to-batch reproducibility. From a supply chain perspective, the volatility of rare earth metal prices and the geopolitical constraints on their sourcing add another layer of risk. The need for multi-step protection and deprotection strategies when using highly reactive organometallic reagents further compounds the issue, resulting in lower overall atom economy and increased waste generation, which contradicts modern green chemistry principles.
The Novel Approach
In stark contrast, the methodology disclosed in CN108558635B offers a streamlined, one-pot solution that operates under remarkably mild conditions. The core innovation lies in the use of simple organic or inorganic bases, such as DBU or Cesium Carbonate, in dimethyl sulfoxide (DMSO) to activate terminal alkynes towards nucleophilic attack on CO2. 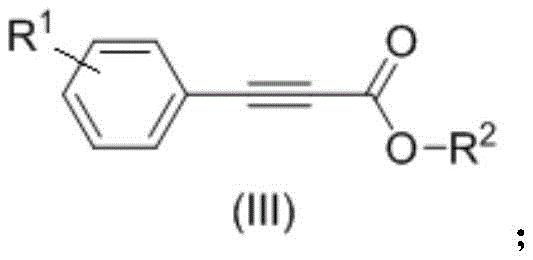 This process proceeds at atmospheric pressure (1 atm) and moderate temperatures ranging from 40°C to 70°C, eliminating the need for high-pressure autoclaves. The reaction demonstrates exceptional substrate tolerance, successfully converting phenylacetylenes bearing electron-withdrawing groups like nitro and halogens, as well as electron-donating groups, into the desired acids with yields reaching up to 98%. Furthermore, the protocol allows for the direct, in-situ conversion of the intermediate carboxylate salts into esters by adding alkyl halides, bypassing isolation steps and significantly enhancing process efficiency.
This process proceeds at atmospheric pressure (1 atm) and moderate temperatures ranging from 40°C to 70°C, eliminating the need for high-pressure autoclaves. The reaction demonstrates exceptional substrate tolerance, successfully converting phenylacetylenes bearing electron-withdrawing groups like nitro and halogens, as well as electron-donating groups, into the desired acids with yields reaching up to 98%. Furthermore, the protocol allows for the direct, in-situ conversion of the intermediate carboxylate salts into esters by adding alkyl halides, bypassing isolation steps and significantly enhancing process efficiency.
Mechanistic Insights into Base-Promoted Carboxylation
The mechanistic elegance of this transformation stems from the ability of strong non-nucleophilic bases to deprotonate the terminal alkyne, generating a reactive acetylide anion stabilized by the polar aprotic solvent, DMSO. Unlike transition metal catalysis which involves oxidative addition and reductive elimination cycles, this base-mediated pathway relies on direct nucleophilic addition of the acetylide species to the electrophilic carbon of CO2. 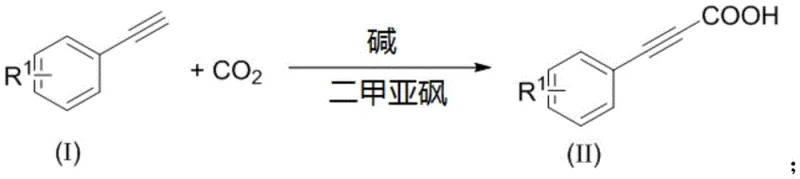 The choice of solvent is critical; experimental data within the patent indicates that while solvents like ethanol, THF, or toluene fail to support the reaction, DMSO provides the necessary dielectric environment to solvate the ionic intermediates and facilitate CO2 insertion. The resulting cesium or ammonium carboxylate salt is stable under the reaction conditions, preventing the reverse decarboxylation reaction that often plagues high-temperature processes. This stability allows the reaction to proceed to near-quantitative conversion without the need for excess driving forces.
The choice of solvent is critical; experimental data within the patent indicates that while solvents like ethanol, THF, or toluene fail to support the reaction, DMSO provides the necessary dielectric environment to solvate the ionic intermediates and facilitate CO2 insertion. The resulting cesium or ammonium carboxylate salt is stable under the reaction conditions, preventing the reverse decarboxylation reaction that often plagues high-temperature processes. This stability allows the reaction to proceed to near-quantitative conversion without the need for excess driving forces.
From an impurity control perspective, the absence of metal catalysts fundamentally simplifies the purification profile. Traditional metal-catalyzed routes often generate homocoupling byproducts (Glaser coupling) or metal-complexed impurities that are difficult to remove. In this metal-free system, the primary byproducts are simply the conjugate acids of the base used, which are water-soluble and easily removed during the aqueous workup. The high selectivity observed across a wide range of substrates, including those with sensitive functional groups like nitro and fluoro substituents, suggests that the basic conditions are sufficiently mild to avoid side reactions such as nucleophilic aromatic substitution or ester hydrolysis during the in-situ esterification step. This clean reaction profile ensures that the final product meets stringent purity specifications required for pharmaceutical applications with minimal downstream processing.
How to Synthesize 3-Arylpropynoic Acid Efficiently
To implement this technology effectively, operators must adhere to strict anhydrous and anaerobic conditions to prevent the quenching of the reactive acetylide intermediate by moisture or oxygen. The standard protocol involves charging the reactor with the base and substrate in DMSO, followed by multiple purge cycles with CO2 to ensure an inert atmosphere. Heating the mixture to the optimal range of 60-70°C maximizes the reaction rate while maintaining high selectivity. For the synthesis of esters, the subsequent addition of alkylating agents such as n-butyl bromide or benzyl bromide allows for the direct formation of the ester bond without isolating the free acid. This seamless integration of carboxylation and esterification into a single operational sequence drastically reduces solvent usage and processing time, making it an ideal candidate for continuous manufacturing or large-batch production.
- Charge a reaction vessel with a base such as Cesium Carbonate (Cs2CO3) or DBU and the terminal phenylacetylene substrate in anhydrous DMSO under inert atmosphere.
- Purge the system with Carbon Dioxide (CO2) gas to replace air, ensuring a pressure of 1 atmosphere, and heat the mixture to 40-70°C for 12-24 hours.
- For esters, add alkyl halides or tosylates directly to the reaction mixture without isolation; for acids, acidify the mixture with HCl and extract with organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free carboxylation technology translates into tangible strategic advantages beyond mere technical feasibility. The elimination of transition metal catalysts removes a significant variable from the raw material cost structure, insulating the production process from the price volatility associated with copper, silver, or palladium markets. Moreover, the use of carbon dioxide as a feedstock offers a virtually inexhaustible and low-cost source of carbon, aligning with corporate sustainability goals and potentially qualifying for green manufacturing incentives. The simplified equipment requirements—specifically the ability to run at atmospheric pressure rather than high pressure—reduce capital expenditure (CAPEX) on specialized reactors and lower maintenance costs, thereby improving the overall return on investment for production facilities.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and complex ligands significantly lowers the direct material costs per kilogram of product. Additionally, the high atom economy and near-quantitative yields minimize waste disposal costs and maximize the utilization of starting materials. The one-pot nature of the esterification process further reduces operational expenses by cutting down on solvent consumption, filtration steps, and drying times, leading to a substantially leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like DMSO, Cesium Carbonate, and CO2, the supply chain becomes more resilient to disruptions compared to dependencies on specialized catalytic systems. The mild reaction conditions reduce the risk of thermal runaways or pressure-related incidents, ensuring consistent uptime and delivery schedules. This reliability is crucial for maintaining the continuity of supply for downstream API manufacturers who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and lack of hazardous high-pressure requirements. The absence of heavy metals simplifies wastewater treatment and effluent management, ensuring easier compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand value of the final pharmaceutical products by associating them with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-promoted carboxylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters. Understanding these nuances is essential for R&D teams evaluating this route for pilot scale-up and for quality assurance teams establishing control strategies for the new manufacturing process.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process (CN108558635B) explicitly eliminates the need for transition metals like Copper or Silver, or rare earth catalysts, relying instead on inexpensive organic or inorganic bases.
Q: What are the pressure requirements for the CO2 carboxylation step?
A: The reaction operates efficiently at normal atmospheric pressure (1 atm), removing the need for specialized high-pressure autoclaves required by traditional methods.
Q: Can this method tolerate sensitive functional groups on the aromatic ring?
A: Yes, the mild reaction conditions (40-70°C) and base-promoted mechanism demonstrate excellent substrate universality, successfully accommodating electron-withdrawing groups like nitro and halogens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylpropynoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free carboxylation technology described in CN108558635B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific solvent and safety requirements of this chemistry, while our rigorous QC labs guarantee that every batch meets stringent purity specifications, free from the heavy metal residues often associated with traditional catalytic methods.
We invite global partners to leverage our technical expertise to optimize their supply chains for 3-arylpropynoic acid derivatives. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your drug development programs.
