Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as the critical backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component synthesis strategy for carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in process chemistry, addressing the longstanding challenges associated with constructing carbonyl-bridged systems through traditional coupling methods. By leveraging a palladium-catalyzed cascade reaction, this invention enables the efficient assembly of diversified substituted double heterocyclic compounds containing trifluoromethyl and carbonyl groups in a single operational step. The strategic importance of this patent lies in its ability to bypass hazardous reagents while maintaining high atom economy and substrate compatibility, making it an ideal candidate for the reliable pharmaceutical intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks has relied on three primary strategies, each fraught with significant industrial drawbacks that hinder large-scale adoption. The first approach involves the direct coupling of two pre-formed heterocyclic substrates, a method often plagued by low reactivity due to steric hindrance and the requirement for harsh activation conditions that can degrade sensitive functional groups. The second strategy employs oxidative cyclization of substrates containing dual nucleophiles with activated methyl-substituted heterocycles, which frequently necessitates the use of stoichiometric oxidants and generates substantial amounts of toxic waste, complicating downstream purification and environmental compliance. Furthermore, existing transition metal-catalyzed tandem cyclization reactions, while more efficient, often struggle with the specific integration of carbonyl bridges, requiring high-pressure carbon monoxide gas which poses severe safety risks and infrastructure costs for manufacturing facilities. These conventional pathways often result in poor regioselectivity and limited scope, restricting the chemical diversity accessible to medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. This method ingeniously combines trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives in a one-pot reaction, facilitated by a PdCl2/TFP catalyst system and a formic acid/acetic anhydride mixture that serves as a safe carbon monoxide surrogate. 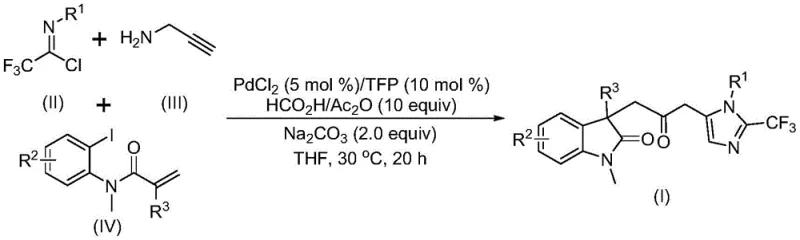 . This transformative process eliminates the need for handling toxic CO gas cylinders, thereby drastically simplifying the safety protocols required for production. The reaction proceeds efficiently at a low temperature of 30°C in tetrahydrofuran, demonstrating exceptional functional group tolerance that allows for the incorporation of various substituents such as halogens, alkyl groups, and nitro groups without compromising yield. This streamlined workflow not only accelerates the synthesis timeline but also significantly reduces the operational complexity, positioning it as a superior choice for cost reduction in API manufacturing.
. This transformative process eliminates the need for handling toxic CO gas cylinders, thereby drastically simplifying the safety protocols required for production. The reaction proceeds efficiently at a low temperature of 30°C in tetrahydrofuran, demonstrating exceptional functional group tolerance that allows for the incorporation of various substituents such as halogens, alkyl groups, and nitro groups without compromising yield. This streamlined workflow not only accelerates the synthesis timeline but also significantly reduces the operational complexity, positioning it as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of bond-forming events orchestrated by the palladium center. The cycle initiates with the oxidative addition of zero-valent palladium into the carbon-iodine bond of the acrylamide substrate, generating a reactive aryl-palladium species. This intermediate subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a stable divalent alkyl-palladium intermediate that sets the stage for the indolinone ring closure. Crucially, the presence of the formic acid and acetic anhydride mixture facilitates the in-situ generation of carbon monoxide, which inserts into the palladium-carbon bond to form an acyl-palladium species. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate, which undergoes isomerization to become susceptible to nucleophilic attack. The final cyclization event is driven by the activation of this amidine by the acyl-palladium complex, resulting in the formation of the imidazole ring and the regeneration of the active palladium catalyst. This concerted mechanism ensures high regioselectivity and minimizes the formation of side products, which is critical for maintaining a clean impurity profile in pharmaceutical intermediates.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in suppressing thermal degradation pathways that often lead to complex byproduct mixtures in high-temperature processes. The use of a specific ligand system, trifuryl phosphine, stabilizes the palladium center against aggregation into inactive palladium black, ensuring consistent catalytic turnover throughout the 12 to 20-hour reaction window. Furthermore, the choice of tetrahydrofuran as the solvent provides an optimal balance of solubility for the diverse organic substrates and the inorganic base, sodium carbonate, preventing heterogeneous mass transfer limitations that could otherwise lead to incomplete conversion. The post-treatment process is equally designed for purity, involving a simple filtration step followed by silica gel column chromatography, which effectively removes palladium residues and unreacted starting materials. This rigorous control over the reaction environment and workup procedure ensures that the final carbonyl-bridged biheterocyclic compounds meet the stringent purity specifications required for downstream drug development applications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale discovery and pilot-scale production, requiring standard equipment and readily available reagents. The protocol begins with the precise weighing of the palladium catalyst and ligand, followed by the addition of the base and the carbon monoxide source mixture into the reaction vessel containing the organic solvent. Once the substrates are introduced, the mixture is stirred at the controlled temperature, allowing the cascade reaction to proceed to completion as monitored by standard analytical techniques. For detailed standardized synthesis steps and specific molar ratios optimized for different substrates, please refer to the guide below.
- Combine palladium chloride catalyst, trifuryl phosphine ligand, sodium carbonate base, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture and stir thoroughly.
- Maintain the reaction at 30°C for 12 to 20 hours, then filter, purify via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic advantages that directly impact the bottom line and operational resilience. The shift away from hazardous gaseous reagents to liquid surrogates fundamentally alters the risk profile of the manufacturing process, reducing the need for specialized high-pressure containment systems and lowering insurance and compliance overheads. Moreover, the reliance on commodity chemicals such as propargylamine and acrylamide, which are produced in vast quantities globally, ensures a stable and continuous supply chain that is less susceptible to the volatility often seen with exotic or custom-synthesized building blocks. This stability is further enhanced by the robustness of the catalyst system, which tolerates a wide range of substrate variations, allowing for flexible sourcing of raw materials without the need for extensive re-validation of the process.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and dangerous carbon monoxide gas infrastructure, which traditionally requires significant capital investment in safety monitoring and delivery systems. By utilizing a formic acid and acetic anhydride mixture as the carbonyl source, the process leverages cheap, bulk-available chemicals that significantly lower the raw material cost per kilogram of the final product. Additionally, the mild reaction conditions of 30°C result in substantially reduced energy consumption compared to high-temperature reflux protocols, contributing to lower utility costs over the lifecycle of the production campaign. The high efficiency of the palladium catalyst, used at low loading levels, combined with the high yields observed in optimized examples, ensures that the cost of goods sold remains competitive even for complex molecular architectures.
- Enhanced Supply Chain Reliability: The synthetic route is built upon a foundation of commercially available starting materials that are not subject to strict regulatory controls or geopolitical supply constraints, ensuring a reliable pharmaceutical intermediate supplier experience. The simplicity of the one-pot procedure minimizes the number of unit operations required, reducing the potential for bottlenecks and equipment downtime that can disrupt production schedules. Furthermore, the broad substrate scope means that a single manufacturing line can be adapted to produce a library of analogues by simply swapping the aryl or alkyl substituents, providing agility in responding to changing market demands or clinical trial requirements without the need for new facility qualification.
- Scalability and Environmental Compliance: Scaling this reaction from gram-scale to commercial tonnage is facilitated by the absence of gas-liquid mass transfer limitations associated with CO gas, making the scale-up predictable and safe. The use of tetrahydrofuran, a common industrial solvent with established recovery and recycling protocols, aligns with green chemistry principles and simplifies waste management procedures. The straightforward workup involving filtration and chromatography avoids the generation of difficult-to-treat aqueous waste streams often associated with strong acid or base quenches, thereby reducing the environmental footprint and disposal costs. This alignment with sustainability goals not only meets current regulatory standards but also future-proofs the supply chain against increasingly stringent environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation technology, derived directly from the experimental data and scope defined in the patent literature. Understanding these nuances is essential for R&D teams planning to integrate this chemistry into their existing pipelines or for procurement teams evaluating the feasibility of long-term supply contracts. The answers provided reflect the specific conditions and outcomes reported in the patent, ensuring accuracy and relevance for technical decision-making.
Q: What is the carbon monoxide source in this synthesis?
A: The process utilizes a mixture of formic acid and acetic anhydride as a safe, in-situ carbon monoxide substitute, eliminating the need for toxic CO gas cylinders.
Q: What are the optimal reaction conditions?
A: The reaction proceeds efficiently at a mild temperature of 30°C in tetrahydrofuran (THF) solvent, typically requiring 12 to 20 hours for completion.
Q: What yields can be expected?
A: Depending on the substrate substituents, isolated yields range significantly, with optimized examples demonstrating yields up to 92%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis route for accelerating the development of next-generation therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific safety and processing requirements of organometallic catalysis, while our rigorous QC labs guarantee that every batch meets stringent purity specifications and complies with international regulatory standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug discovery programs with reliable quality and consistent performance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient pathway for your target molecules. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a discussion on route feasibility assessments for your proprietary candidates, ensuring your supply chain is optimized for speed, cost, and reliability.
