Advanced Palbociclib Manufacturing: A Novel Grignard-Based Synthetic Route for Commercial Scale-Up
Advanced Palbociclib Manufacturing: A Novel Grignard-Based Synthetic Route for Commercial Scale-Up
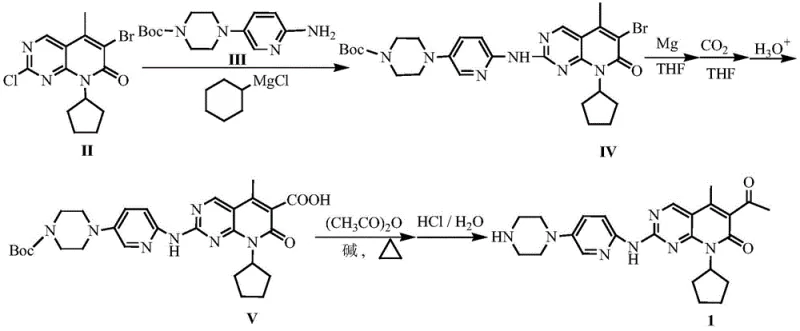
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of high-value oncology therapeutics, particularly for CDK4/6 inhibitors like Palbociclib. Patent CN109867673B, published in March 2021, introduces a transformative synthetic strategy that addresses critical bottlenecks in the existing manufacturing landscape. This intellectual property discloses a streamlined three-step process that leverages cyclohexylmagnesium chloride for coupling, followed by a Grignard carboxylation and a final acetylation-deprotection sequence. By shifting away from toxic organotin reagents and sensitive lithium bases, this method offers a pathway to high-purity intermediates with significantly reduced operational hazards. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable API intermediate supplier capable of delivering consistent quality at scale.
![Chemical structure of Palbociclib showing the pyrido[2,3-d]pyrimidin-7-one core](/insights/img/palbociclib-synthesis-grignard-pharma-supplier-20260303074637-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Palbociclib has been plagued by reliance on hazardous and expensive reagents that complicate industrial scale-up. One prominent prior art route utilizes organic tin reagents in a Stille coupling step; these reagents are not only prohibitively expensive and difficult to source globally but also introduce severe toxicity concerns that require rigorous and costly purification protocols to meet regulatory limits. Another established method depends on lithium hexamethyldisilazide (LHMDS), a base that is extremely sensitive to moisture and air, leading to inconsistent reaction outcomes and the generation of complex byproduct profiles that necessitate silica gel column chromatography, a technique generally unsuitable for multi-ton production. Furthermore, alternative pathways employing isopropyl magnesium chloride generate substantial volumes of flammable propane gas, creating significant safety liabilities in manufacturing plants, while palladium-catalyzed steps often suffer from long reaction times and the formation of structurally similar impurities that are notoriously difficult to separate.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in CN109867673B presents a highly efficient and industrially viable alternative that prioritizes safety and cost-effectiveness. The core innovation lies in the strategic use of cyclohexylmagnesium chloride as a nucleophilic coupling agent, which avoids the generation of flammable gases and utilizes readily available commodity chemicals. The subsequent transformation involves converting a bromo-intermediate into a Grignard reagent using magnesium metal, followed by direct carboxylation with carbon dioxide, a cheap and abundant C1 source, to install the necessary carboxylic acid functionality. This route completely bypasses the need for precious metal catalysts like palladium or toxic tin reagents, thereby simplifying the downstream processing and drastically reducing the environmental footprint of the synthesis. The final steps involve a mild acetylation with acetic anhydride and a safe hydrochloric acid-mediated deprotection, ensuring the final product is free from genotoxic sulfonate impurities often associated with methanesulfonic acid deprotection methods.
Mechanistic Insights into Grignard Carboxylation and Acetylation
The mechanistic elegance of this synthesis begins with the coupling of the halogenated pyridopyrimidine core with the piperazine-pyridine fragment. Under the influence of cyclohexylmagnesium chloride, a transmetallation or activation event occurs that facilitates the nucleophilic attack, forming the critical C-N bond to yield intermediate (IV) with high fidelity. This step is crucial as it establishes the central scaffold of the molecule without the side reactions typical of palladium-catalyzed aminations, such as homocoupling or incomplete conversion. The reaction conditions are optimized to proceed at moderate temperatures, typically around 60°C, which balances reaction kinetics with thermal stability, ensuring that the sensitive heterocyclic core remains intact throughout the transformation. The use of THF as a solvent provides the necessary solvation for the magnesium species while maintaining a manageable viscosity for large-scale stirring and heat transfer.
Following the coupling, the conversion of intermediate (IV) to the carboxylic acid (V) represents a masterclass in atom economy and impurity control. The bromine atom on the pyridopyrimidine ring serves as a handle for the formation of a Grignard reagent upon treatment with magnesium metal. Once the organomagnesium species is generated, the introduction of dry carbon dioxide gas allows for a nucleophilic addition to the electrophilic carbon of CO2, effectively extending the carbon skeleton by one unit to form a carboxylate salt. Subsequent acidic hydrolysis liberates the free carboxylic acid (V). This approach is superior to lithiation strategies because Grignard reagents are generally more tolerant of certain functional groups and easier to handle on a large scale. The final acetylation of the methyl group adjacent to the carbonyl is achieved using acetic anhydride in the presence of an organic base like pyridine, followed by acid-catalyzed deprotection. Crucially, the use of HCl instead of methanesulfonic acid prevents the formation of alkyl mesylates, thereby eliminating a major class of genotoxic impurities and simplifying the mutagenicity assessment of the final drug substance.
How to Synthesize Palbociclib Efficiently
The execution of this synthetic route requires precise control over reaction parameters, particularly temperature and moisture levels, to maximize yield and purity. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing overall cycle time. While the general workflow involves the initial coupling, followed by Grignard formation and carboxylation, and concluding with acetylation and deprotection, the specific stoichiometry and workup procedures are critical for success. Operators must ensure that the Grignard formation is initiated correctly to avoid passivation of the magnesium surface, and the carbonation step must be maintained at low temperatures to prevent side reactions. For a detailed breakdown of the specific molar ratios, solvent volumes, and isolation techniques required to replicate this high-yielding process, please refer to the standardized operating procedures below.
- Couple compound (II) with compound (III) using cyclohexylmagnesium chloride in THF to obtain intermediate (IV).
- React intermediate (IV) with magnesium metal to form a Grignard reagent, followed by carbonation with CO2 and hydrolysis to yield carboxylic acid (V).
- Acetylate compound (V) with acetic anhydride in pyridine, followed by acid-catalyzed deprotection to isolate Palbociclib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route translates directly into enhanced operational resilience and cost efficiency. The elimination of exotic reagents like organic tin compounds and specialized palladium catalysts removes significant volatility from the raw material supply chain, as the new process relies on bulk commodities like magnesium, carbon dioxide, and acetic anhydride which are universally available and price-stable. This shift not only secures the supply line against geopolitical or logistical disruptions affecting specialty chemical markets but also drastically simplifies the vendor qualification process. Furthermore, the removal of toxic heavy metals from the process flow reduces the burden on waste management systems and lowers the costs associated with environmental compliance and hazardous waste disposal, contributing to a more sustainable and economically favorable manufacturing model.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of high-cost catalytic systems with stoichiometric reagents that are inexpensive and widely sourced. By avoiding the use of palladium catalysts and phosphine ligands, which represent a significant portion of the bill of materials in traditional cross-coupling reactions, the overall variable cost of goods sold is substantially decreased. Additionally, the simplified purification requirements, stemming from the cleaner reaction profile and absence of heavy metal residues, reduce the consumption of chromatography media and solvents, further driving down the cost per kilogram of the final API intermediate. This leaner material usage profile ensures that the manufacturing process remains economically viable even at fluctuating market prices for fine chemicals.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on stable, non-hazardous reagents like cyclohexylmagnesium chloride and carbon dioxide enhances the predictability of production schedules. Unlike moisture-sensitive lithium reagents that require specialized storage and handling infrastructure, the reagents used in this process are more forgiving and easier to transport, reducing the risk of batch failures due to reagent degradation. This robustness allows for more flexible inventory management and shorter lead times for raw material procurement. Consequently, manufacturers can maintain higher service levels and respond more agilely to demand surges from downstream pharmaceutical partners, ensuring a continuous flow of high-quality intermediates without the delays often caused by the sourcing of niche catalysts.
- Scalability and Environmental Compliance: The safety profile of this synthesis is inherently superior for large-scale operations, as it eliminates the generation of flammable propane gas and toxic tin byproducts. This reduction in process hazards lowers the capital expenditure required for explosion-proof equipment and specialized ventilation systems, making it easier to scale production from pilot plant to commercial multi-ton reactors. Moreover, the avoidance of genotoxic impurities simplifies the regulatory filing process and reduces the analytical testing burden, accelerating time-to-market. The greener nature of the process, characterized by lower toxicity and better atom economy, aligns perfectly with modern corporate sustainability goals and increasingly stringent environmental regulations, future-proofing the manufacturing asset against evolving compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for industrial application. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing route.
Q: How does this new synthesis route improve upon previous methods involving tin reagents?
A: Unlike prior art routes that utilize toxic and expensive organic tin reagents which are difficult to remove and purchase, this novel method employs cyclohexylmagnesium chloride and standard Grignard chemistry. This substitution eliminates the need for complex heavy metal scavenging steps, significantly simplifying the purification process and reducing environmental hazards associated with tin waste disposal.
Q: What are the safety advantages regarding the nucleophilic reagents used?
A: Previous synthetic pathways often relied on isopropyl magnesium chloride, which generates large amounts of flammable propane gas during the reaction, posing significant safety risks in large-scale workshops. The disclosed method utilizes cyclohexylmagnesium chloride, which avoids the generation of low-boiling-point flammable gases, thereby enhancing operational safety and reducing the complexity of explosion-proof requirements in the manufacturing facility.
Q: Does this process avoid the formation of genotoxic impurities?
A: Yes, a critical advantage of this route is the avoidance of methanesulfonic acid for deprotection. Conventional methods using methanesulfonic acid carry a risk of generating genotoxic methanesulfonate esters. This new protocol utilizes hydrochloric acid for the final deprotection step, effectively mitigating the risk of genotoxic impurity formation and ensuring a safer impurity profile for the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the route disclosed in CN109867673B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to implement this Grignard-based strategy effectively. We are committed to delivering high-purity Palbociclib intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to ensure every batch is free from the genotoxic and heavy metal impurities that plague older synthesis methods. Our facility is equipped to handle the specific safety and processing requirements of this route, ensuring a seamless transition from development to full-scale manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your oncology drug development pipeline.
