Advanced Manufacturing of Palbociclib Intermediates: A High-Yield PdCl2 Catalytic Strategy for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of efficient oncology treatments has placed significant focus on the supply chain integrity of key kinase inhibitors, particularly Palbociclib. As a critical CDK4/6 inhibitor, the commercial viability of Palbociclib relies heavily on the robustness of its synthetic intermediates. Patent CN108117550B introduces a transformative preparation method for the pyrido[2,3-d]pyrimidine compound, designated herein as Compound 1, which serves as a pivotal scaffold in this therapeutic class. This technical disclosure addresses longstanding bottlenecks in the manufacturing of this high-purity pharmaceutical intermediate by leveraging a simplified palladium chloride (PdCl2) catalytic system. Unlike legacy processes that suffer from prolonged reaction times and complex catalyst systems, this innovation demonstrates that high-efficiency cyclization can be achieved under moderate thermal conditions, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
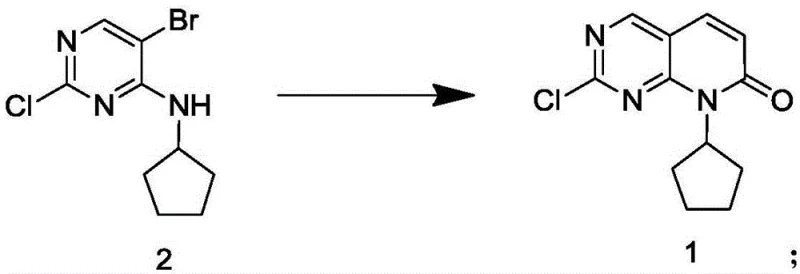
![Chemical structure of Compound 1, a key pyrido[2,3-d]pyrimidine intermediate for Palbociclib](/insights/img/palbociclib-intermediate-synthesis-pdcl2-pharma-supplier-20260309075247-01.png)
The structural complexity of Compound 1 necessitates a synthetic route that balances reactivity with selectivity. The patent elucidates a pathway that not only secures the carbon skeleton effectively but also ensures that the impurity profile remains within stringent limits required for downstream API synthesis. By focusing on the direct coupling of readily available precursors, this method reduces the step count and operational complexity typically associated with constructing the fused ring system. For procurement teams evaluating cost reduction in API manufacturing, the shift towards such streamlined chemistries represents a strategic opportunity to lower the cost of goods sold (COGS) while enhancing supply security.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound 1 has been plagued by inefficiencies inherent to early-generation palladium-catalyzed protocols. Prior art, including methodologies referenced in international patent filings such as WO2014128588 and WO2016030439, often relies on sophisticated catalyst systems like Pd(OAc)2 paired with bulky phosphine ligands or requires harsh oxidative conditions. These conventional approaches frequently demand reaction temperatures exceeding 90°C and extended durations ranging from several hours to multiple days to reach acceptable conversion rates. Furthermore, the reliance on expensive ligand-capped palladium species introduces significant cost burdens and complicates the purification process, as removing trace phosphine oxides and metal residues requires additional processing steps. The yield in these traditional methods typically hovers around 80%, which, when compounded over multi-step syntheses, results in substantial material loss and increased waste generation, posing challenges for commercial scale-up of complex pharmaceutical intermediates.
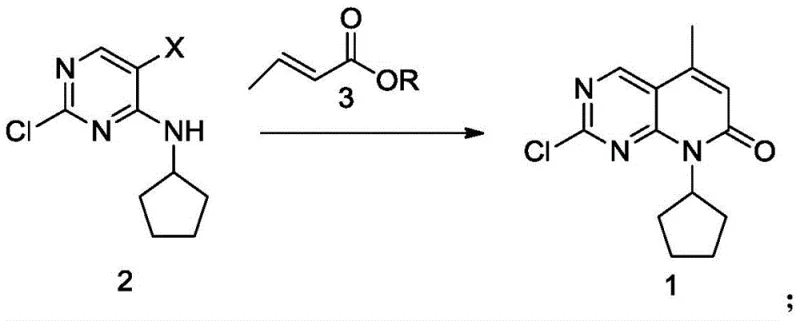
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in CN108117550B utilizes a remarkably simple yet highly effective catalytic system centered on PdCl2. This method enables the direct reaction between Compound 2 (a halogenated pyrimidine) and Compound 3 (an unsaturated carboxylic acid derivative) to form the target bicyclic structure with exceptional efficiency. The elimination of exogenous phosphine ligands is a game-changer, as it drastically reduces the raw material cost and simplifies the reaction matrix. Operating at a moderate temperature range of 60°C to 85°C, preferably between 65°C and 75°C, this process minimizes thermal stress on the reactants, thereby suppressing the formation of thermal degradation by-products. The result is a robust process capable of delivering yields as high as 94.8% with purity levels exceeding 99%, setting a new benchmark for reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into PdCl2-Catalyzed Cyclization
The efficacy of this novel synthesis lies in the unique reactivity of the PdCl2 catalyst within the specific solvent environment of N-methyl-2-pyrrolidone (NMP) or tetrahydrofuran (THF). Mechanistically, the palladium center facilitates the oxidative addition into the carbon-halogen bond of Compound 2, followed by coordination and insertion of the alkene moiety from Compound 3. The presence of a tertiary amine base, such as triethylamine, plays a dual role: it acts as a proton scavenger to drive the equilibrium forward and potentially stabilizes the active palladium species in the absence of traditional ligands. This ligand-free or in-situ ligand generation mechanism allows for a more open coordination sphere around the metal center, enhancing the rate of the migratory insertion step which is often the rate-determining step in such cyclizations. The subsequent reductive elimination restores the aromaticity of the pyridine ring and regenerates the active catalyst, completing the cycle with high turnover frequency.
From an impurity control perspective, the moderate reaction temperature is critical. High-temperature processes often promote non-selective radical pathways or over-oxidation, leading to difficult-to-remove polymeric impurities or regio-isomers. By maintaining the reaction at 70°C (reflux in THF or controlled heating in NMP), the kinetic energy is sufficient to overcome the activation barrier for the desired cyclization without triggering competing decomposition pathways. Furthermore, the stoichiometry of the base and the acid component is finely tuned, with a molar ratio of Compound 2 to base optimized between 1:2 and 1:4. This precise balance ensures complete neutralization of the acid by-product while preventing base-mediated hydrolysis of sensitive functional groups, resulting in a crude product profile that is exceptionally clean and amenable to simple crystallization rather than column chromatography.
How to Synthesize Compound 1 Efficiently
The practical implementation of this synthesis involves dissolving the halogenated precursor and the acrylic acid derivative in a polar aprotic solvent, followed by the addition of the base and the palladium catalyst under an inert atmosphere. The reaction mixture is then heated to the optimal temperature window and monitored via TLC or HPLC until the starting material is fully consumed. Following the cyclization, a post-treatment step involving acetic anhydride is employed to cap any remaining reactive amines or hydroxyls, facilitating the final crystallization. For a detailed, step-by-step breakdown of the standardized operating procedure, including specific workup and purification protocols, please refer to the guide below.
- Dissolve Compound 2 and crotonic acid (Compound 3) in a water-soluble organic solvent like NMP with a base such as triethylamine.
- Add PdCl2 catalyst under nitrogen atmosphere and heat the mixture to 60-85°C for approximately 6 hours until completion.
- Perform post-treatment by reacting with acetic anhydride, followed by crystallization, filtration, and drying to isolate high-purity Compound 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this PdCl2-catalyzed route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the bill of materials. By replacing expensive, air-sensitive ligand-palladium complexes with commodity-grade PdCl2, the direct material cost is significantly reduced. Additionally, the shortened reaction time—from days in some prior art methods to merely 6 hours in this optimized protocol—increases the throughput capacity of existing reactor assets, effectively expanding production capability without capital expenditure. This efficiency gain translates directly into improved margin potential and a more resilient supply chain capable of responding rapidly to market demand fluctuations for oncology APIs.
- Cost Reduction in Manufacturing: The elimination of proprietary phosphine ligands removes a major cost driver from the synthesis. Traditional catalysts often require specialized handling and storage due to air sensitivity, whereas PdCl2 is stable and easy to handle. Furthermore, the high yield (>90%) minimizes the loss of valuable starting materials, ensuring that every kilogram of input generates maximum output. The simplified workup, which avoids complex chromatographic separations in favor of crystallization and filtration, reduces solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity chemicals such as crotonic acid, triethylamine, and NMP ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialty reagents. PdCl2 is a standard industrial chemical with a robust global supply network, mitigating the risk of production stoppages due to catalyst shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility and ensuring consistent batch-to-batch quality for downstream customers.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and atmospheric pressure, reducing the energy footprint and safety risks associated with high-pressure or cryogenic reactions. The use of NMP, while requiring careful handling, allows for high solubility and efficient reaction kinetics, and the solvent can be recovered and recycled effectively. The reduction in reaction time and the high atom economy of the cyclization step align with green chemistry principles, lowering the E-factor (mass of waste per mass of product) and facilitating easier compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational benefits and technical feasibility of the method for potential manufacturing partners.
Q: What are the advantages of using PdCl2 over traditional palladium catalysts for this synthesis?
A: Using simple PdCl2 eliminates the need for expensive phosphine ligands found in traditional catalysts like Pd(PPh3)4, significantly reducing raw material costs and simplifying the removal of metal residues from the final product.
Q: How does this method improve the purity profile compared to prior art?
A: The optimized reaction conditions at moderate temperatures (65-75°C) minimize thermal degradation and side reactions, consistently achieving purities above 98.5% without requiring complex chromatographic purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available solvents like NMP and standard equipment requirements, with short reaction times (approx. 6h) and high yields (up to 94.8%), making it highly scalable for ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Intermediate Supplier
The technological advancements presented in CN108117550B underscore the potential for significant process intensification in the production of kinase inhibitor intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Compound 1 meets the exacting standards required for global pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can be integrated into your supply chain. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your oncology drug development programs.
