Advanced Manufacturing of Crisaborole Intermediates via Efficient Palladium Catalysis
Introduction to Next-Generation Crisaborole Synthesis
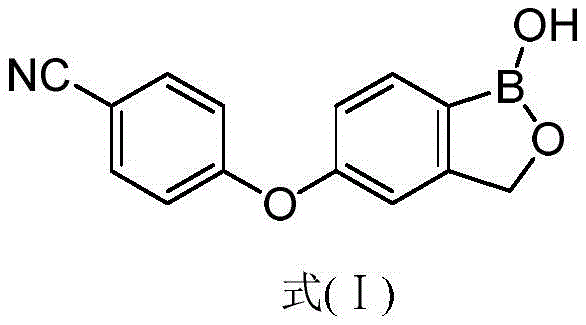
The pharmaceutical landscape for dermatological treatments has been significantly transformed by the introduction of Crisaborole, a potent phosphodiesterase 4 (PDE4) inhibitor approved for the treatment of mild-to-moderate atopic dermatitis. As demand for this active pharmaceutical ingredient (API) continues to rise, the industry requires robust, scalable, and cost-effective synthetic methodologies to ensure supply chain stability. Patent CN108047261B, published in late 2020, discloses a groundbreaking preparation method that addresses the longstanding challenges associated with synthesizing 5-(4-cyanophenoxy)-1,3-dihydro-1-hydroxy-2,1-benzoxaborole. This technical insight report analyzes the proprietary chemistry detailed in the patent, highlighting how the strategic use of hydroxyl-protected intermediates facilitates a high-yield palladium-catalyzed borylation process. By shifting away from harsh organolithium reagents and low-yielding cyclization protocols, this novel approach offers a viable pathway for reliable pharmaceutical intermediate suppliers to enhance production capacity while maintaining stringent quality standards.
The core innovation lies in the stabilization of the reactive benzylic position prior to the critical carbon-boron bond formation. Traditional methods often struggle with the compatibility of the free hydroxyl group and the nitrile functionality under strong basic or nucleophilic conditions. The disclosed method circumvents these issues by introducing a versatile protecting group strategy, wherein the hydroxyl moiety is temporarily masked as an ester or ether. This modification not only enhances the solubility of the substrate in organic media but also prevents decomposition pathways that typically plague earlier synthetic attempts. For procurement managers and R&D directors alike, understanding this mechanistic shift is crucial for evaluating the long-term viability and cost structure of Crisaborole manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for Crisaborole have been plagued by significant inefficiencies that hinder commercial scalability. One prominent strategy, documented in earlier patents such as CN101914109B, relies on the generation of boronic acid intermediates via organolithium chemistry. This approach necessitates the use of alkyl lithium reagents at cryogenic temperatures, which poses severe safety risks and operational complexities on a large scale. Furthermore, the presence of the electrophilic cyano group on the aromatic ring creates a high propensity for nucleophilic attack by the lithium species, leading to a complex mixture of by-products and drastically reduced yields. Another reported pathway, found in academic literature like Bioorganic & Medicinal Chemistry Letters, utilizes a direct cyclization strategy that suffers from a single-step yield of merely 30 percent. Such low efficiency results in excessive waste generation, inflated raw material costs, and difficult purification processes, making these conventional methods economically unviable for high-volume API production.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN108047261B introduces a streamlined two-step sequence centered around a protected intermediate. The novelty begins with the conversion of 2-halo-5-(4-cyanophenoxy)benzyl alcohol into a protected derivative, such as the acetate ester (Formula III). This protected substrate then undergoes a palladium-catalyzed Miyaura borylation with bis(pinacolato)diboron. Unlike the unstable organolithium intermediates, the resulting pinacol boronate ester (Formula II) is a stable, isolable solid that can be purified to high standards before the final cyclization. This decoupling of the borylation and ring-closing steps allows for independent optimization of each stage, ensuring that impurities do not carry through to the final API. The ability to isolate and characterize the boronate intermediate provides a critical quality control checkpoint, significantly reducing the risk of batch failure and ensuring consistent product quality for downstream formulation.

Mechanistic Insights into Pd-Catalyzed Borylation
The heart of this improved synthesis is the transition metal-catalyzed coupling reaction that installs the boron functionality onto the aromatic core. The mechanism proceeds through a classic palladium catalytic cycle, initiating with the oxidative addition of the aryl halide (Formula III) to the Pd(0) species. The use of robust ligands, such as those found in Pd(dppf)Cl2 or tetrakis(triphenylphosphine)palladium, stabilizes the metal center and facilitates this critical step even in the presence of the electron-withdrawing cyano group. Following oxidative addition, the palladium complex undergoes transmetallation with the diboron reagent, activated by the base (e.g., potassium acetate or carbonate). This step transfers the boron moiety to the palladium center, setting the stage for reductive elimination, which releases the desired aryl boronate ester and regenerates the active catalyst. The choice of solvent, ranging from 1,4-dioxane to toluene, plays a pivotal role in solubilizing the inorganic base and maintaining the homogeneity of the reaction mixture, thereby maximizing turnover frequency.
Furthermore, the protection of the benzylic hydroxyl group is mechanistically essential for suppressing competitive side reactions. In the absence of protection, the free alcohol could potentially coordinate to the palladium center, inhibiting catalysis, or participate in base-mediated elimination reactions. By masking the alcohol as an acetyl or silyl group, the electronic and steric environment of the substrate is tuned to favor the desired cross-coupling pathway. Experimental data from the patent indicates that when the acetyl protecting group is employed, the borylation step achieves yields as high as 86 percent, a substantial improvement over unprotected analogues. This high conversion rate minimizes the burden on downstream purification, allowing for simple crystallization or filtration techniques to isolate the intermediate, which aligns perfectly with the requirements for cost reduction in pharmaceutical intermediate manufacturing.
How to Synthesize Crisaborole Efficiently
The practical implementation of this synthesis involves a logical progression of protection, borylation, and deprotection-cyclization. The process begins with the acylation of the starting benzyl alcohol using acetic anhydride in the presence of a catalyst like DMAP, yielding the acetate intermediate in near-quantitative yield. This intermediate is then subjected to the borylation conditions described previously, utilizing mild heating (e.g., 85°C) to drive the reaction to completion. The resulting boronate ester is isolated as a stable solid, ready for the final transformation. The concluding step involves treating the boronate ester with either acid or base to simultaneously remove the protecting group and induce intramolecular cyclization, forming the characteristic B-O bond of the benzoxaborole ring. Detailed standardized synthesis steps follow below for technical reference.
- Protect the hydroxyl group of 2-halo-5-(4-cyanophenoxy)benzyl alcohol (Formula IV) using an acylating agent like acetic anhydride to form Formula III.
- React Formula III with bis(pinacolato)diboron in the presence of a palladium catalyst and base to generate the pinacol boronate ester (Formula II).
- Perform acid or base-mediated cyclization of Formula II to remove the protecting group and form the benzoxaborole ring, yielding Crisaborole (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented methodology translates into tangible operational benefits that extend beyond mere chemical yield. The shift from cryogenic organolithium chemistry to moderate-temperature palladium catalysis eliminates the need for specialized low-temperature reactors and hazardous pyrophoric reagents. This simplification of the reaction infrastructure significantly lowers capital expenditure requirements and reduces the complexity of safety protocols, thereby enhancing overall plant throughput. Moreover, the stability of the protected boronate intermediate allows for flexible production scheduling; the intermediate can be manufactured in large batches and stored, decoupling the upstream synthesis from the final API production and mitigating risks associated with supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like n-butyllithium, coupled with the use of commodity chemicals such as acetic anhydride and potassium acetate, drives down the raw material cost profile. Additionally, the high yields observed in both the protection and borylation steps minimize waste disposal costs and maximize the utilization of expensive palladium catalysts through efficient recycling protocols. The simplified workup procedures, often requiring only filtration and crystallization rather than complex chromatography, further reduce labor and solvent consumption, contributing to substantial cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to market volatility. The ability to produce a stable, isolable intermediate provides a strategic buffer, allowing manufacturers to build inventory of the key precursor without concerns regarding degradation. This stability is crucial for meeting the rigorous delivery timelines demanded by global pharmaceutical clients, ensuring continuity of supply even during periods of high demand or raw material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup options described in the patent facilitate easy scale-up from kilogram to multi-ton production scales without significant re-engineering of the process. The reduction in hazardous waste generation, particularly the avoidance of lithium salts and aggressive quenching steps, aligns with modern green chemistry principles and environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and supports the sustainability goals of forward-thinking pharmaceutical companies seeking eco-friendly supply partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Crisaborole intermediates. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's potential. Understanding these nuances is essential for technical teams assessing the feasibility of technology transfer or process validation.
Q: Why is the hydroxyl protection step critical in this synthesis?
A: Protecting the benzylic hydroxyl group prevents unwanted side reactions during the palladium-catalyzed borylation step, significantly improving reaction yield and purity compared to unprotected routes.
Q: What are the advantages of using acetic anhydride as the protecting reagent?
A: Using acetic anhydride to form the acetyl-protected intermediate offers superior crystallinity and ease of purification compared to other protecting groups like THP, leading to higher overall process efficiency.
Q: Can this process be scaled for industrial production?
A: Yes, the patent describes mild reaction conditions (0-150°C) and simple post-treatment methods like filtration and crystallization, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crisaborole Supplier
As the global demand for non-steroidal treatments for atopic dermatitis grows, securing a dependable source of high-quality Crisaborole intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in organoboron chemistry to deliver superior pharmaceutical intermediates. Our state-of-the-art facilities are equipped to handle complex catalytic processes, including the palladium-mediated borylation routes described in recent patents. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our intermediates meet the exacting standards required for FDA-regulated drug products.
We invite procurement directors and R&D teams to collaborate with us to optimize their supply chains for Crisaborole production. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and process constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can drive efficiency and reliability in your API production, ensuring a steady flow of high-purity intermediates for your critical dermatological formulations.
