Advanced Manufacturing of Crisaborole Intermediates via Optimized Suzuki Coupling
Advanced Manufacturing of Crisaborole Intermediates via Optimized Suzuki Coupling
The pharmaceutical landscape for non-steroidal phosphodiesterase 4 (PDE4) inhibitors has been significantly shaped by the introduction of Crisaborole, commercially known as Eucrisa, for the treatment of atopic dermatitis. As demand for this active pharmaceutical ingredient (API) continues to grow, the efficiency and safety of its supply chain become paramount for global procurement strategies. Patent CN113979891A discloses a groundbreaking preparation method for Crisaborole and its critical intermediates, addressing long-standing challenges in synthetic organic chemistry related to safety and scalability. This technical insight report analyzes the proprietary synthesis route which replaces hazardous cryogenic reactions with a robust, palladium-catalyzed protocol, offering a viable pathway for reliable pharmaceutical intermediates supplier partnerships. The core innovation lies in the strategic manipulation of protecting groups and the application of mild Suzuki-Miyaura coupling conditions, ensuring high purity and operational safety.
![Chemical structure of Crisaborole (4-[(1,3-Dihydro-1-hydroxy-2,1-benzoxaborolan-5-yl)oxy]benzonitrile)](/insights/img/crisaborole-intermediate-synthesis-pharma-supplier-20260304183259-01.webp)
The target molecule, Crisaborole, features a unique benzoxaborole ring system fused with a benzonitrile ether moiety, a structural motif that presents specific synthetic hurdles. The disclosed method begins with the transformation of simple benzoic acid derivatives through a sequence of esterification, etherification, reduction, and protection, culminating in a sophisticated boronation step. By rigorously optimizing reaction parameters such as solvent choice, temperature profiles, and catalyst loading, the inventors have achieved a process that not only enhances the overall yield but also drastically simplifies the purification workflow. For R&D directors evaluating process feasibility, this route represents a significant departure from legacy methods, prioritizing green chemistry principles without compromising on the stringent quality standards required for topical dermatological treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxaborole-based APIs has been plagued by reliance on extremely hazardous reagents and fragile protecting group strategies that hinder industrial scale-up. One prominent prior art method, documented in Chinese patent CN 101914109B, utilizes n-butyllithium (n-BuLi) to introduce the boron atom via metal-halogen exchange. This approach necessitates reaction temperatures as low as -78°C to control the exothermic nature of the lithiation and prevent decomposition, requiring specialized cryogenic equipment that imposes substantial capital expenditure (CAPEX) and operational complexity. Furthermore, n-BuLi is a pyrophoric substance that ignites spontaneously upon contact with air, creating severe safety liabilities for manufacturing facilities and complicating logistics for raw material handling.
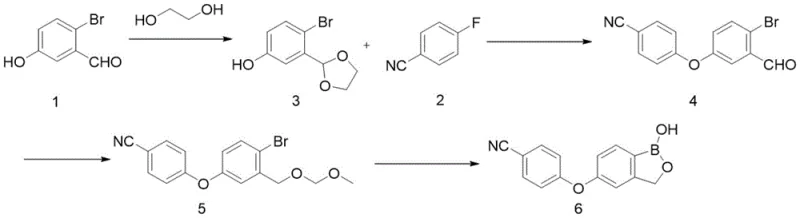
Another conventional strategy, reported in international publication WO 2018207216 A1, attempts to mitigate some risks by employing acetyl protection for the hydroxyl groups. However, this method suffers from inherent chemical instability; the acetyl group is prone to hydrolysis under the alkaline conditions required for the subsequent boronation step. This side reaction generates unwanted byproducts that contaminate the reaction mixture, necessitating extensive downstream purification efforts that erode the overall process yield. The combination of safety hazards associated with organolithium reagents and the yield losses due to protecting group lability creates a bottleneck for cost reduction in pharmaceutical intermediates manufacturing, making these legacy routes economically unattractive for high-volume production.
The Novel Approach
In stark contrast to these problematic legacy routes, the methodology outlined in patent CN113979891A introduces a streamlined and chemically robust synthesis pathway that eliminates the need for cryogenic conditions and unstable protecting groups. The new process leverages a tetrahydropyranyl (THP) ether protection strategy, which exhibits superior stability under the basic conditions of the Suzuki coupling reaction, thereby preserving the integrity of the intermediate throughout the synthesis. Instead of dangerous lithiation, the method employs a palladium-catalyzed cross-coupling reaction using pinacol diboron, a stable and easy-to-handle solid reagent, to install the boron functionality efficiently.
This novel approach allows the critical boronation step to proceed at moderate temperatures ranging from 80°C to 100°C, which is compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The elimination of the -78°C requirement removes the need for expensive liquid nitrogen or specialized refrigeration cycles, directly translating to lower energy consumption and reduced utility costs. Moreover, the use of pinacol diboron in conjunction with catalysts such as (dppf)PdCl2 ensures high selectivity and conversion rates, minimizing the formation of homocoupling byproducts. This shift towards milder, more stable chemistry exemplifies a modern strategy for commercial scale-up of complex pharmaceutical intermediates, aligning perfectly with the industry's move towards safer and more sustainable manufacturing practices.
Mechanistic Insights into Palladium-Catalyzed Boronation and Cyclization
The heart of this innovative synthesis lies in the mechanistic elegance of the Suzuki-Miyaura coupling followed by an acid-mediated cyclization. The process begins with the activation of the aryl bromide intermediate (Compound VI) by the palladium catalyst, forming an oxidative addition complex. This is followed by transmetallation with the pinacol diboron species, facilitated by the presence of a base such as potassium acetate, which activates the diboron reagent to form a reactive boronate species. The subsequent reductive elimination step forge the carbon-boron bond, yielding the aryl boronic ester intermediate with high fidelity. Unlike lithiation methods which are sensitive to trace moisture and oxygen, this catalytic cycle is remarkably tolerant to minor variations in reaction conditions, providing a robust margin of error for process engineers.
Following the successful installation of the boron moiety, the synthesis concludes with a clever one-pot deprotection and cyclization sequence triggered by the addition of inorganic acid, typically hydrochloric acid. The acidic environment serves a dual purpose: it cleaves the acid-labile THP protecting group to reveal the benzylic alcohol, and simultaneously promotes the intramolecular condensation between the newly freed hydroxyl group and the boronic acid functionality. This spontaneous cyclization forms the thermodynamically stable five-membered benzoxaborole ring, driving the equilibrium towards the final product (Compound I). This tandem deprotection-cyclization mechanism not only reduces the number of isolation steps but also ensures that the final product precipitates out of the solution, facilitating easy filtration and drying. Such mechanistic efficiency is crucial for maintaining a clean impurity profile, a key metric for high-purity pharmaceutical intermediates intended for regulatory submission.
How to Synthesize Crisaborole Efficiently
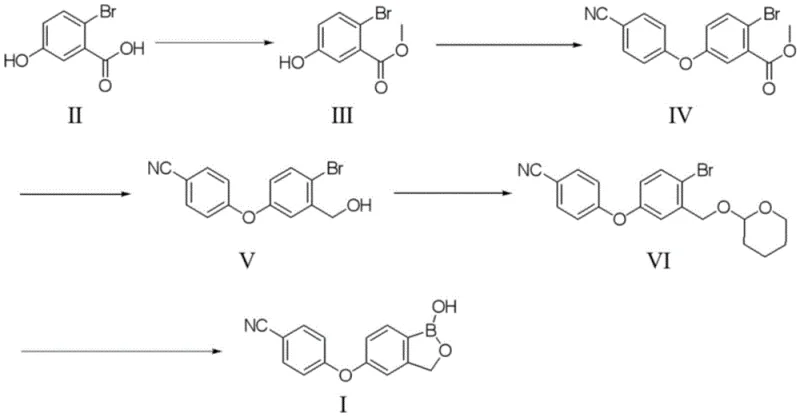
The synthesis of Crisaborole via this patented route involves a logical sequence of transformations starting from commercially available 2-bromo-5-hydroxybenzoic acid. The process is designed to maximize atom economy and minimize waste generation at each stage. Initially, the carboxylic acid is esterified to protect it during subsequent nucleophilic substitutions, followed by the formation of the diaryl ether linkage using 4-fluorobenzonitrile. The ester is then reduced to an alcohol and protected as a THP ether to survive the harsh conditions of the boronation step. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and workup procedures, are outlined in the guide below.
- Perform esterification of 2-bromo-5-hydroxybenzoic acid with methanol under acidic conditions to form the methyl ester intermediate.
- Conduct nucleophilic aromatic substitution with 4-fluorobenzonitrile under alkaline conditions to establish the ether linkage.
- Reduce the ester group to a primary alcohol using alkali metal borohydride, protect the alcohol with dihydropyran, and finally execute Suzuki coupling with pinacol diboron followed by acid cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling economic and logistical advantages that extend beyond simple yield improvements. By fundamentally altering the reaction conditions from cryogenic to moderate temperatures, the process removes significant barriers to entry for contract manufacturing organizations (CMOs) who may lack specialized low-temperature infrastructure. This accessibility broadens the potential supplier base, enhancing supply chain resilience and reducing the risk of production bottlenecks. Furthermore, the replacement of hazardous pyrophoric reagents with stable solids like pinacol diboron simplifies raw material logistics, lowering transportation costs and insurance premiums associated with dangerous goods.
- Cost Reduction in Manufacturing: The elimination of n-butyllithium and the associated cryogenic cooling requirements results in substantial operational cost savings. Without the need for maintaining -78°C environments, energy consumption is drastically reduced, and the lifespan of reactor equipment is extended due to less thermal stress. Additionally, the higher stability of the THP protecting group minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents required for purification, leading to lower waste disposal costs and improved overall process economics.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methanol, potassium carbonate, and pinacol diboron ensures a stable and continuous supply of raw materials, mitigating the risk of shortages often associated with specialized organometallic reagents. The robustness of the palladium-catalyzed step allows for flexible batch scheduling and easier technology transfer between different manufacturing sites, ensuring consistent delivery timelines for downstream API producers. This reliability is critical for maintaining uninterrupted production schedules for finished dermatological formulations.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of less hazardous reagents align closely with green chemistry principles, simplifying the environmental permitting process for new production lines. The process generates fewer toxic byproducts and avoids the generation of lithium salts, which can be challenging to treat in wastewater streams. This environmental compatibility facilitates smoother regulatory approvals and supports corporate sustainability goals, making the manufacturing process more attractive for long-term investment and reducing lead time for high-purity pharmaceutical intermediates market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of adopting this methodology for large-scale production.
Q: Why is the new synthesis route safer than traditional methods?
A: Traditional methods often rely on n-butyllithium (n-BuLi) at cryogenic temperatures (-78°C), which poses significant flammability and explosion risks. The new route utilizes mild Suzuki-Miyaura coupling conditions at 90°C, eliminating the need for hazardous pyrophoric reagents and extreme cooling infrastructure.
Q: How does this process improve yield compared to acetyl protection methods?
A: Previous routes using acetyl protection suffered from hydrolysis under alkaline conditions during boronation, leading to byproduct formation and reduced yields. This optimized protocol employs a tetrahydropyranyl (THP) protecting group which offers superior stability during the coupling reaction, thereby maximizing the conversion to the final boronic acid species.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. By avoiding low-temperature reactions and utilizing robust palladium catalysis with readily available reagents like pinacol diboron, the method facilitates easier heat management and reactor operation, making it highly compatible with commercial-scale manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crisaborole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to ensure the consistent supply of high-quality dermatological APIs. Our technical team has thoroughly analyzed the methodology described in patent CN113979891A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this optimized process to life. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of Crisaborole intermediate performs flawlessly in your final formulation.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your Crisaborole production program.
