Advanced Copper-Catalyzed Synthesis of 1-Substituted-1,2,3-Triazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously demand efficient, scalable routes for constructing heterocyclic scaffolds, particularly the 1,2,3-triazole motif which is ubiquitous in modern drug design. Patent CN101792438A introduces a robust methodology for the synthesis of 1-substituted-1,2,3-triazoles, addressing critical bottlenecks in intermediate manufacturing. This technology leverages a copper-catalyzed decarboxylative coupling strategy, utilizing propiolic acid and organic azides to generate high-value structural units found in blockbuster drugs like Tazobactam. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains for pharmaceutical intermediates by replacing expensive noble metal catalysts with cost-effective copper systems while maintaining high purity standards.
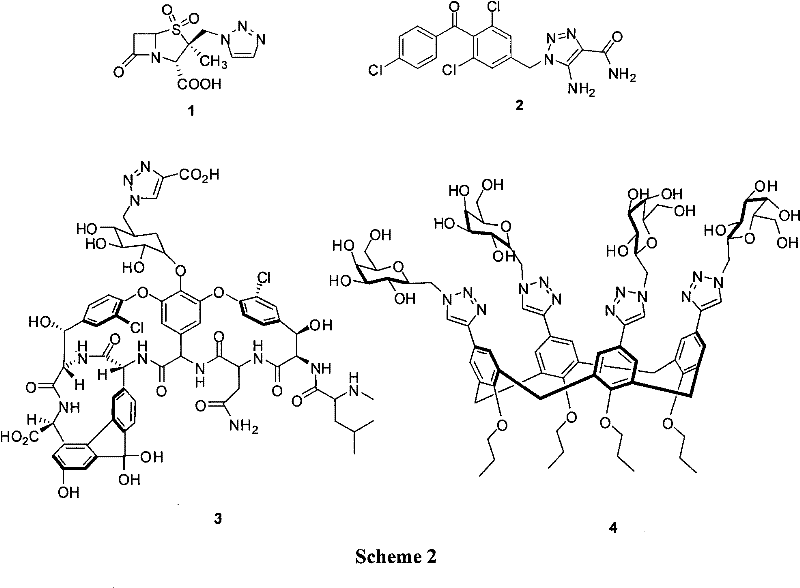
The strategic value of this synthesis lies in its ability to produce 1-substituted triazoles directly, bypassing the need for complex protecting group strategies or harsh thermal conditions that often degrade sensitive functional groups. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for ensuring batch-to-batch consistency and regulatory compliance. The following analysis dissects the technical merits of this approach, highlighting its potential for cost reduction in pharmaceutical intermediate manufacturing and its scalability for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,3-triazole ring has relied heavily on thermal 1,3-dipolar cycloaddition between azides and alkynes, a process that often requires elevated temperatures and prolonged reaction times, leading to poor regioselectivity and the formation of isomeric mixtures. Furthermore, alternative catalytic methods employing ruthenium or palladium complexes, while effective for specific regioisomers, introduce significant economic and environmental burdens due to the high cost of precious metals and the stringent requirements for residual metal removal in final drug substances. Traditional routes often involve multi-step sequences to install the triazole moiety, increasing the overall process mass intensity and generating substantial chemical waste, which complicates the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology described in CN101792438A revolutionizes this landscape by employing a copper-catalyzed decarboxylative coupling of terminal alkynoic acids, specifically propiolic acid, with organic azides. This innovative route operates under mild conditions, typically ranging from room temperature to moderate heating, and utilizes inexpensive copper salts such as cuprous iodide or copper sulfate alongside sodium ascorbate as a reducing agent. The reaction demonstrates exceptional tolerance for various functional groups on the aromatic azide component, including nitro, halo, and ester substituents, allowing for the direct synthesis of diverse 1-substituted triazoles without the need for extensive protection-deprotection sequences. This streamlined approach not only enhances atom economy but also simplifies downstream processing, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Coupling
The core of this synthesis involves a copper-mediated cycle where the active Cu(I) species coordinates with the terminal alkyne of propiolic acid to form a copper-acetylide intermediate. Unlike standard Click chemistry which yields 1,4-disubstituted triazoles, this specific protocol facilitates a decarboxylation event subsequent to or concurrent with the cycloaddition, ultimately yielding the 1-substituted product. The presence of sodium ascorbate ensures the maintenance of the copper in its active +1 oxidation state, preventing oxidative deactivation and ensuring consistent catalytic turnover throughout the reaction duration. This mechanistic pathway is highly advantageous for R&D teams as it minimizes the formation of side products associated with thermal decomposition or alternative cycloaddition pathways.
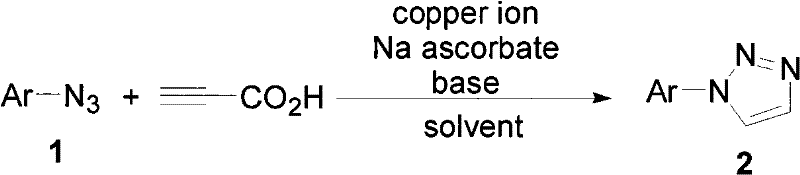
Impurity control is inherently managed through the high regioselectivity of the copper catalyst, which directs the formation of the triazole ring exclusively at the 1-position relative to the aryl group from the azide. The use of mild bases such as DBU or triethylamine in certain embodiments further assists in neutralizing acidic byproducts and facilitating the decarboxylation step without promoting hydrolysis of sensitive esters or amides present in the substrate. For quality assurance, this predictable reactivity profile ensures that the impurity spectrum remains narrow and well-defined, simplifying the validation of purification protocols such as column chromatography or crystallization, which is critical for meeting the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize 1-Substituted-1,2,3-Triazoles Efficiently
The operational simplicity of this patent makes it highly attractive for process chemists aiming to establish robust manufacturing protocols. The general procedure involves charging a reactor with the copper catalyst, sodium ascorbate, the specific organic azide, and propiolic acid in a polar aprotic solvent like DMF or DMSO. The reaction mixture is then stirred magnetically, with temperature and time adjusted based on the electronic nature of the azide substrate; electron-deficient azides may react faster at lower temperatures, while electron-rich variants might require mild heating. Following the reaction, a standard aqueous workup involving extraction with ethyl acetate or dichloromethane effectively isolates the crude product, which can be further purified to analytical grade.
- Combine cuprous halide or copper sulfate catalyst, sodium ascorbate, organic azide, and propiolic acid in a suitable solvent system such as DMF or DMSO.
- Maintain the reaction mixture under magnetic stirring at temperatures ranging from room temperature to 200°C for a duration of 0.5 hours to 3 days.
- Upon completion, extract the product with ethyl acetate or dichloromethane, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from precious metal catalysts to base metal copper systems offers immediate and tangible benefits for cost management and supply chain resilience. The raw materials specified in this patent, including propiolic acid and various substituted phenyl azides, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even during market fluctuations, a critical factor for supply chain heads managing long-term production schedules for active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or ruthenium catalysts drastically lowers the direct material cost per kilogram of the intermediate. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing process. The avoidance of complex purification steps required to remove trace noble metals further decreases operational expenditures, allowing for significant margin improvement in the final product pricing.
- Enhanced Supply Chain Reliability: By utilizing widely available copper salts and common organic solvents, the manufacturing process becomes less susceptible to geopolitical supply disruptions often associated with rare earth or precious metal mining. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent systems, provides flexibility in production planning, enabling manufacturers to adapt quickly to changing capacity requirements without compromising product quality.
- Scalability and Environmental Compliance: The protocol's compatibility with standard reactor setups and its operation under relatively mild conditions facilitate seamless scale-up from laboratory to pilot and commercial scales. The reduced reliance on toxic heavy metals aligns with increasingly stringent environmental regulations and green chemistry principles, minimizing the environmental footprint of the manufacturing process and simplifying regulatory filings related to elemental impurities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium systems?
A: This method utilizes inexpensive copper catalysts instead of precious palladium or ruthenium metals, significantly reducing raw material costs and simplifying heavy metal removal processes for pharmaceutical grade compliance.
Q: Can this synthesis protocol be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly highlights the method's suitability for industrialization due to mild reaction conditions, simple workup procedures involving standard extraction, and the use of commercially available solvents.
Q: What is the regioselectivity profile of this triazole synthesis?
A: The process specifically yields 1-substituted-1,2,3-triazoles through a decarboxylative mechanism, avoiding the mixture of 1,4- and 1,5-isomers often seen in non-catalyzed thermal cycloadditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role of efficient heterocycle synthesis in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1-substituted-1,2,3-triazole intermediates meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and reduce overall project costs.
