Scalable Synthesis of 1-Substituted-1,2,3-Triazoles for High-Purity Pharmaceutical Intermediates
Scalable Synthesis of 1-Substituted-1,2,3-Triazoles for High-Purity Pharmaceutical Intermediates
The landscape of heterocyclic chemistry has been profoundly transformed by the advent of efficient click chemistry methodologies, particularly those enabling the rapid construction of nitrogen-rich scaffolds essential for modern drug discovery. Among these, the 1,2,3-triazole ring system stands out as a privileged structure due to its exceptional metabolic stability, hydrogen-bonding capability, and dipole moment, which facilitate strong interactions with biological targets. The patent CN101792438B, titled "Method for synthesizing 1-substituted-1,2,3-tolyltriazole," introduces a robust and versatile synthetic protocol that addresses critical bottlenecks in the production of these valuable intermediates. By leveraging a copper-catalyzed cycloaddition strategy involving organic azides and propiolic acid, this technology offers a pathway to generate 1-substituted-1,2,3-triazoles with remarkable efficiency and operational simplicity. For R&D directors and procurement specialists in the fine chemical sector, this represents a significant opportunity to optimize supply chains for complex pharmaceutical building blocks, ensuring consistent quality while mitigating the risks associated with traditional, harsher synthetic routes.
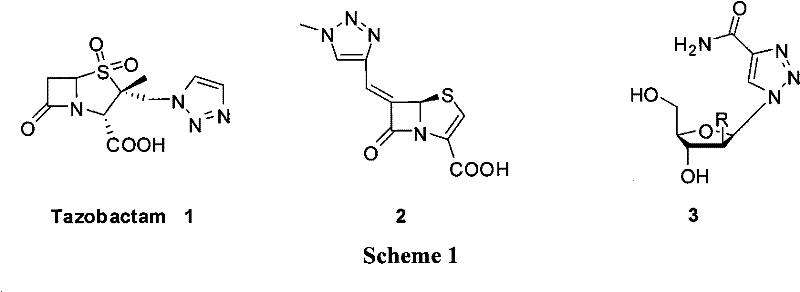
The significance of the 1,2,3-triazole motif cannot be overstated in the context of contemporary medicinal chemistry. As illustrated in the structural diversity of known bioactive molecules, this heterocycle serves as a core component in a wide array of therapeutic agents, ranging from beta-lactamase inhibitors like Tazobactam to potent antitumor and antifungal candidates. The ability to introduce this ring system reliably and cost-effectively is paramount for the development of next-generation APIs. The methodology described in the referenced patent provides a generalized solution that accommodates a broad spectrum of substituents, including electron-withdrawing and electron-donating groups on the aromatic ring, as well as heterocyclic variants. This universality makes it an ideal candidate for library synthesis in drug discovery campaigns as well as for the kilogram-scale manufacturing of key intermediates required for commercial drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles relied heavily on thermal Huisgen 1,3-dipolar cycloaddition reactions between azides and alkynes. While chemically sound, this classical approach suffers from severe practical drawbacks that hinder its utility in industrial settings. The reaction typically requires elevated temperatures, often exceeding 100°C, and prolonged reaction times, which can lead to thermal decomposition of sensitive functional groups and the formation of complex mixtures of 1,4- and 1,5-regioisomers. Separating these isomers is notoriously difficult and resource-intensive, requiring extensive chromatographic purification that drastically reduces overall yield and increases waste generation. Furthermore, alternative strategies involving transition metal catalysts such as palladium have been explored to improve selectivity; however, the reliance on precious metals introduces substantial cost burdens and necessitates rigorous metal scavenging steps to meet the stringent residual metal limits imposed by regulatory bodies for pharmaceutical ingredients. These factors collectively contribute to high manufacturing costs and extended lead times, creating a pressing need for more sustainable and economical alternatives.
The Novel Approach
In stark contrast to these legacy methods, the process detailed in patent CN101792438B utilizes a copper(I)-catalyzed system that operates under remarkably mild conditions, often at room temperature or with moderate heating. The core innovation lies in the specific combination of a copper source, such as cuprous iodide or copper sulfate, with sodium ascorbate as a reducing agent to maintain the active catalytic species in situ. This system facilitates the reaction between organic azides and propiolic acid to directly yield 1-substituted-1,2,3-triazoles with high regioselectivity. The versatility of this method is evidenced by its compatibility with a diverse array of solvents, including polar aprotic solvents like DMF and DMSO, as well as aqueous mixtures, allowing for flexibility in process optimization. Moreover, the workup procedure is exceptionally straightforward, involving simple liquid-liquid extraction followed by standard column chromatography, which streamlines the isolation of the pure product. This approach not only eliminates the formation of unwanted regioisomers but also avoids the use of expensive noble metals, thereby offering a clear economic advantage for large-scale production.
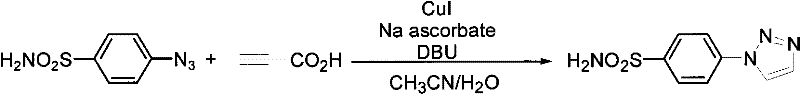
Mechanistic Insights into Cu-Catalyzed Azide-Alkyne Cycloaddition
The efficacy of this synthesis relies on the well-established mechanism of the Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC), often referred to as the premier "click chemistry" reaction. In this catalytic cycle, the copper(I) species coordinates with the terminal alkyne, in this case, propiolic acid, to form a copper-acetylide intermediate. This activation significantly enhances the nucleophilicity of the alkyne, facilitating a concerted [3+2] cycloaddition with the organic azide. The presence of sodium ascorbate is critical, as it continuously reduces any oxidized copper(II) species back to the active copper(I) state, ensuring the catalytic cycle proceeds without interruption. This redox buffering prevents catalyst deactivation and minimizes the formation of oxidative byproducts, which is a common issue in aerobic conditions. The specific use of propiolic acid in this patent is particularly noteworthy; while typical CuAAC reactions yield 1,4-disubstituted triazoles, the conditions described promote the formation of 1-substituted triazoles, suggesting a potential decarboxylation pathway or a specific mechanistic variation that eliminates the carboxylic acid moiety post-cyclization. This unique transformation simplifies the molecular architecture, directly providing the desired 1-substituted scaffold without the need for additional decarboxylation steps, thereby enhancing atom economy and process efficiency.
From an impurity control perspective, the mechanism offers inherent advantages that are highly valued by quality assurance teams. The high specificity of the copper-acetylide interaction ensures that side reactions, such as polymerization of the alkyne or non-specific azide reduction, are minimized. The reaction tolerates a wide range of functional groups, including nitro, halo, hydroxyl, and sulfonamide moieties, without the need for protecting groups. This chemoselectivity reduces the number of synthetic steps required to build complex molecules, directly correlating to a cleaner impurity profile in the final API intermediate. The ability to run the reaction in solvents like water or aqueous acetonitrile further aids in the suppression of organic side products, as the polarity of the medium favors the desired polar transition states. Consequently, the resulting crude product is of high quality, requiring minimal purification effort to achieve pharmaceutical-grade standards, which is a decisive factor in reducing the cost of goods sold (COGS) for high-value intermediates.
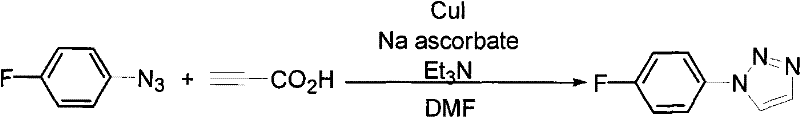
How to Synthesize 1-Substituted-1,2,3-Triazoles Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The patent outlines a generalized procedure that is adaptable to various substrates, making it a robust tool for process chemists. The key to success lies in maintaining the correct molar ratios between the catalyst, the reducing agent, and the reactants, as deviations can lead to incomplete conversion or catalyst precipitation. The following guide summarizes the critical operational parameters derived from the patent examples, providing a foundation for scaling this technology.
- Combine cuprous halide or copper sulfate catalyst, sodium ascorbate, organic azide, and propiolic acid in a suitable solvent such as DMF or DMSO.
- Stir the reaction mixture magnetically at temperatures ranging from room temperature to 200°C for a duration between 0.5 hours and 3 days depending on substrate reactivity.
- Upon completion, extract with ethyl acetate or dichloromethane, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift from precious metal catalysts to abundant copper salts represents a fundamental change in the cost structure of triazole manufacturing. Copper is orders of magnitude cheaper than palladium or ruthenium, and its widespread availability insulates the supply chain from the volatility often seen in the precious metals market. Furthermore, the elimination of expensive ligands and the ability to use commodity solvents like DMF or even water reduces the raw material expenditure significantly. The simplified workup procedure, which avoids complex distillation or specialized scavenging resins, lowers the operational overhead and reduces the consumption of consumables such as silica gel and filtration media. These cumulative savings contribute to a more competitive pricing model for the final intermediate, allowing downstream partners to optimize their own margins.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the replacement of high-cost catalytic systems with inexpensive copper sources. By utilizing cuprous iodide or copper sulfate, the direct material cost for the catalyst is drastically reduced compared to palladium-based alternatives. Additionally, the mild reaction conditions, often proceeding at room temperature or moderate heat, result in substantial energy savings over the course of a production campaign. The high yields reported in the patent examples, frequently exceeding 80% and reaching up to 94% for certain substrates, mean that less raw material is wasted, further driving down the effective cost per kilogram of the product. This efficiency allows for a leaner manufacturing process with reduced waste disposal costs, aligning with both financial and environmental goals.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials. Organic azides and propiolic acid are commercially accessible commodities, reducing the risk of bottlenecks associated with bespoke reagents. The robustness of the reaction across different solvent systems provides flexibility; if a specific solvent faces supply constraints, the process can often be adapted to use alternatives like acetonitrile/water mixtures or DMSO without re-validating the entire chemistry. This adaptability ensures continuity of supply even in fluctuating market conditions. Moreover, the scalability of the reaction from milligram to multi-kilogram scales has been demonstrated through the variety of examples provided, giving confidence that the process can meet increasing demand without requiring fundamental re-engineering of the production line.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener profile than many traditional heterocyclic syntheses. The avoidance of high-temperature thermal cycloadditions reduces the energy footprint of the process. The potential to use aqueous solvent systems minimizes the volume of volatile organic compounds (VOCs) released into the atmosphere, facilitating compliance with increasingly stringent environmental regulations. The copper catalyst, while requiring removal, is easier to manage and recycle compared to heavy noble metals, and the overall waste stream is less hazardous. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, which is increasingly important for partnerships with major pharmaceutical companies committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of this Cu-catalyzed method over traditional thermal cycloaddition?
A: This method operates under significantly milder conditions, often at room temperature, eliminating the need for high-energy thermal inputs. It utilizes inexpensive copper catalysts instead of precious metals like palladium, drastically reducing raw material costs while maintaining high regioselectivity and yield.
Q: Which solvents are compatible with this synthesis protocol for industrial scale-up?
A: The patent demonstrates versatility across a wide range of polar aprotic and protic solvents, including DMF, DMSO, acetonitrile/water mixtures, and even pure water in specific cases. This flexibility allows process chemists to select solvents based on solubility profiles and environmental safety regulations without compromising reaction efficiency.
Q: How does the impurity profile compare to other triazole synthesis routes?
A: The use of sodium ascorbate as a reducing agent ensures the copper remains in the active Cu(I) state, minimizing side reactions associated with oxidative degradation. The simple workup procedure involving extraction and standard column chromatography effectively removes metal residues and unreacted starting materials, resulting in a high-purity final product suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results described in patent CN101792438B can be seamlessly translated into industrial reality. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 1-substituted-1,2,3-triazole meets the exacting standards required for pharmaceutical applications. Our commitment to process excellence means we can deliver these complex heterocycles with consistent quality, supporting your R&D timelines and commercial manufacturing needs with reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this copper-catalyzed route can optimize your budget. Please contact us to request specific COA data for our available triazole derivatives and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the delivery of life-saving medicines to the market through superior chemical innovation.
