Scalable Synthesis of Chiral Indoxazinones for Advanced Oncology Drug Development
Scalable Synthesis of Chiral Indoxazinones for Advanced Oncology Drug Development
The pharmaceutical industry is constantly seeking robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN112209947A, which discloses a novel class of chiral indoxazinone compounds and their efficient synthesis. This technology addresses the longstanding challenge of achieving high enantioselectivity in indole-fused ring systems without resorting to expensive transition metal catalysts or extreme reaction conditions. The disclosed compounds, characterized by a fused indole-oxazinone core, have demonstrated promising cytotoxic activity against PC-3 human prostate cancer cells, positioning them as valuable candidates for anticancer drug discovery. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral indole-fused ring compounds has been plagued by significant operational hurdles that impede commercial viability. Traditional routes often rely on harsh reaction conditions, including extreme temperatures or the use of hazardous reagents, which not only increase safety risks but also complicate process control and waste management. Furthermore, many existing methods suffer from poor enantioselectivity, yielding racemic mixtures that require costly and inefficient resolution steps to isolate the biologically active enantiomer. The sensitivity of indole substrates to oxidative degradation and polymerization under vigorous conditions further limits the scope of applicable substrates, often resulting in low yields and inconsistent product quality. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating bottlenecks for supply chain managers aiming to secure reliable sources of complex intermediates.
The Novel Approach
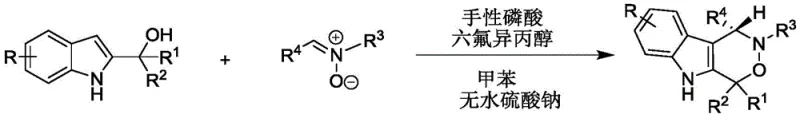
In stark contrast, the methodology outlined in CN112209947A introduces a remarkably mild and efficient organocatalytic strategy. By utilizing 2-indolemethanol and nitrones as readily available starting materials, the process achieves direct construction of the chiral indoxazinone skeleton in a single step. The reaction operates under ambient thermal conditions, specifically between 20-30°C, eliminating the need for energy-intensive heating or cryogenic cooling. The use of toluene as a solvent and anhydrous sodium sulfate as an additive simplifies the reaction matrix, while the dual catalytic system ensures exceptional stereocontrol. This approach not only broadens the substrate scope to include diverse electronic and steric variations but also delivers products with high yields and excellent enantiomeric excess (ee), effectively solving the purity and efficiency issues inherent in older technologies.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
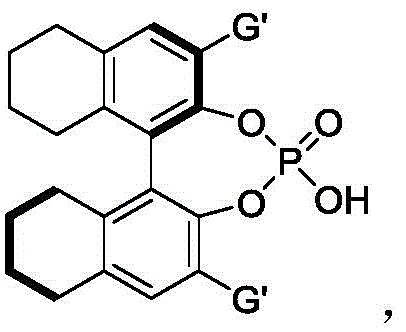
The success of this synthetic route lies in the sophisticated interplay between the chiral phosphoric acid catalyst and hexafluoroisopropanol (HFIP). The chiral phosphoric acid, derived from binaphthyl or spiro skeletons, acts as a bifunctional catalyst, simultaneously activating the electrophilic nitrone and the nucleophilic indole moiety through a hydrogen-bonding network. This precise spatial arrangement directs the approach of the reactants, enforcing a specific trajectory that favors the formation of one enantiomer over the other. The addition of HFIP further enhances this effect by strengthening the hydrogen bonds and stabilizing the transition state, thereby boosting both the reaction rate and the stereoselectivity. This synergistic catalysis allows for the formation of two new stereocenters with high fidelity, a feat that is often difficult to achieve with metal-based catalysts which may suffer from leaching or toxicity issues.
From an impurity control perspective, the mild nature of this organocatalytic cycle minimizes side reactions such as polymerization or decomposition of the sensitive indole ring. The absence of heavy metals eliminates the risk of metal contamination, a critical parameter for pharmaceutical intermediates intended for clinical use. The reaction pathway is clean, with the primary byproduct being water, which is sequestered by the anhydrous sodium sulfate, driving the equilibrium towards product formation. This mechanistic elegance translates directly into a simpler purification profile, reducing the burden on downstream processing and ensuring that the final API intermediate meets stringent regulatory standards for residual solvents and impurities.
How to Synthesize Chiral Indoxazinone Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the reaction mixture in toluene, where the stoichiometry of 2-indolemethanol to nitrone is carefully controlled, typically ranging from 1:1 to 2:1, with a preferred ratio of 1.2:1 to maximize conversion. The addition of the catalytic system is critical; a loading of approximately 10 mol% chiral phosphoric acid and 60 mol% HFIP has been shown to provide optimal results. The reaction proceeds at room temperature (30°C) and is monitored via TLC until completion, usually within 24 hours. Following the reaction, the workup involves a simple filtration to remove the drying agent, followed by concentration and purification via silica gel column chromatography using a petroleum ether and dichloromethane mixture.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone in toluene with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol as co-catalysts to initiate the asymmetric cyclization.
- Stir the reaction at 20-30°C until completion, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The shift from harsh, metal-catalyzed processes to this mild organocatalytic method fundamentally alters the cost structure and risk profile of manufacturing these complex intermediates. By operating at ambient temperatures and using common solvents like toluene, the process significantly reduces energy consumption and eliminates the need for specialized cryogenic infrastructure. This simplicity translates into lower capital expenditure (CAPEX) for reactor setups and reduced operational expenditure (OPEX) regarding utility costs. Furthermore, the high atom economy and selectivity mean that less raw material is wasted, directly contributing to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver and the associated expense of metal scavenging steps required to meet regulatory limits. Additionally, the high yields (up to 98%) and excellent enantioselectivity (up to 96% ee) minimize the loss of valuable starting materials and reduce the need for recycling or reprocessing off-spec batches. The use of commercially available and relatively inexpensive reagents like 2-indolemethanol and nitrones ensures a stable and predictable raw material cost base, shielding the supply chain from volatility associated with exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of this reaction under mild conditions makes it less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or mixing efficiency. This reliability is crucial for maintaining consistent supply schedules for downstream API synthesis. The broad substrate scope demonstrated in the patent allows for the production of a diverse library of analogs from a common set of precursors, enabling flexible inventory management and rapid response to changing R&D demands. The simplicity of the workup procedure also shortens the overall production cycle time, enhancing the agility of the supply chain.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards and the use of standard organic solvents. The generation of minimal waste, primarily aqueous waste from the drying agent and solvent residues that can be recovered, aligns with green chemistry principles and simplifies environmental compliance. This eco-friendly profile is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet sustainability goals. The process safety is inherently high due to the mild operating conditions, reducing the risk of accidents and ensuring business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral indoxazinone compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for integration into their supply chains.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes mild reaction conditions (20-30°C) and avoids harsh reagents, resulting in higher safety, lower energy costs, and excellent enantioselectivity (up to 96% ee).
Q: What is the biological activity of the synthesized compounds?
A: The synthesized chiral indoxazinones exhibit significant cytotoxic activity against PC-3 human prostate cancer cells, with IC50 values as low as 40.08 μg/mL for specific derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available solvents like toluene, simple workup procedures (filtration and chromatography), and operates at ambient temperatures, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN112209947A for the development of novel anticancer agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the high enantiomeric excess and chemical purity required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data for our catalog of chiral indole derivatives or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in delivering high-quality, cost-effective solutions for the global healthcare market.
