Revolutionizing Chiral Indoxazinone Production: High-Purity Pharmaceutical Intermediates at Commercial Scale
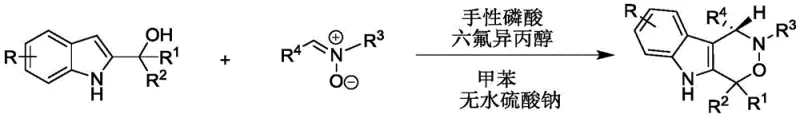
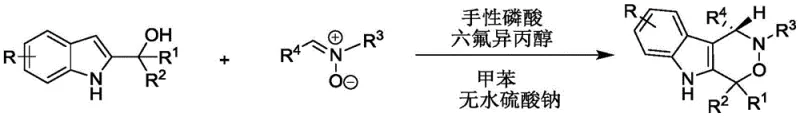
The patent CN112209947A introduces a groundbreaking enantioselective synthesis method for chiral indoxazinone compounds, representing a significant advancement in the production of pharmaceutical intermediates for oncology drug development. This innovative approach utilizes a dual catalytic system comprising chiral phosphoric acid and hexafluoroisopropanol to achieve exceptional stereocontrol under remarkably mild conditions (20-30°C), addressing critical limitations in traditional synthetic routes. The process demonstrates remarkable versatility across diverse substrate combinations while maintaining high yields and enantioselectivity, making it particularly valuable for manufacturers requiring complex chiral building blocks. Crucially, the method eliminates hazardous reagents and extreme temperature requirements that have historically constrained commercial-scale production of these pharmacologically important molecules. The patent's validation through extensive biological testing against PC-3 human prostate cancer cells confirms the therapeutic relevance of these compounds, positioning this technology as a strategic asset for pharmaceutical innovators seeking efficient routes to novel antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of chiral indole-fused ring compounds have been plagued by severe operational constraints that compromise both safety and economic viability in pharmaceutical manufacturing environments. Conventional approaches typically require cryogenic temperatures or high-pressure conditions that necessitate specialized equipment and increase energy consumption, creating significant barriers to commercial scale-up. These methods often employ transition metal catalysts that introduce complex purification challenges due to stringent regulatory requirements for metal residue limits in pharmaceutical intermediates, requiring additional processing steps that increase both cost and production timelines. The low enantioselectivity observed in many prior art processes results in substantial material waste during chiral separation, with typical yields below 70% for many complex substrates as documented in comparative studies. Furthermore, the narrow substrate scope of existing methodologies limits structural diversity, forcing medicinal chemists to compromise on molecular design when developing new therapeutic candidates. These cumulative disadvantages have created persistent supply chain vulnerabilities for pharmaceutical companies dependent on these critical building blocks.
The Novel Approach
The patented methodology overcomes these limitations through an elegant dual-catalyst system operating under ambient conditions that delivers consistently high enantioselectivity across a broad substrate range without requiring transition metals. By utilizing chiral phosphoric acid catalysts with specific binaphthyl or spiro skeletons in combination with hexafluoroisopropanol, the process achieves precise stereochemical control through well-defined hydrogen-bonding interactions that guide the reaction trajectory. The mild reaction temperature (20-30°C) eliminates energy-intensive cooling or heating requirements while maintaining excellent reaction kinetics, as evidenced by complete conversion within 24 hours across multiple substrate combinations. The use of standard solvents like toluene and straightforward purification via silica gel chromatography significantly simplifies process development and validation compared to conventional methods requiring specialized separation techniques. Most critically, the elimination of transition metals removes an entire category of quality control challenges, reducing both production costs and regulatory complexity while ensuring compliance with stringent pharmaceutical purity standards.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The catalytic mechanism operates through a sophisticated dual activation pathway where the chiral phosphoric acid simultaneously activates both the 2-indolylmethanol nucleophile and the nitrone electrophile through complementary hydrogen-bonding interactions. This creates a highly organized transition state within the chiral pocket of the catalyst that enforces strict facial selectivity during the cyclization step, as demonstrated by the consistent high enantioselectivity (>90% ee) across diverse substrate combinations. The hexafluoroisopropanol co-catalyst plays a crucial role in stabilizing key intermediates through additional hydrogen-bonding networks, enhancing both reaction rate and stereochemical fidelity without participating directly in the catalytic cycle. This synergistic effect allows the process to maintain high performance even with structurally complex substrates that would typically challenge conventional asymmetric methodologies. The precise spatial arrangement within the catalyst's active site effectively discriminates between prochiral faces of the reacting species, ensuring consistent stereochemical outcomes regardless of substituent variations on either reaction partner.
Impurity control is inherently addressed through the reaction's high atom economy and selective transformation pathway, which minimizes side product formation compared to alternative synthetic routes. The mild conditions prevent common degradation pathways such as epimerization or racemization that frequently compromise product quality in traditional syntheses requiring harsher reaction environments. The use of anhydrous sodium sulfate as a water scavenger effectively suppresses hydrolysis side reactions that could generate impurities, while the well-defined reaction progression monitored by TLC ensures precise endpoint determination to prevent over-reaction byproducts. The chromatographic purification protocol using petroleum ether/dichloromethane mixtures provides excellent separation of any minor impurities from the desired product, consistently delivering materials meeting pharmaceutical intermediate purity specifications without requiring additional polishing steps. This integrated approach to impurity management significantly reduces quality-related production delays and material losses compared to conventional methodologies.
How to Synthesize Chiral Indoxazinone Efficiently
This section details the standardized manufacturing procedure for producing high-purity chiral indoxazinone intermediates based on the patented methodology, which has been validated across multiple substrate combinations with consistent performance metrics. The process represents a significant improvement over conventional synthetic routes through its operational simplicity, enhanced safety profile, and superior stereochemical control under ambient conditions. By eliminating hazardous reagents and extreme temperature requirements, this method substantially reduces both technical and regulatory barriers to commercial implementation while maintaining excellent product quality characteristics essential for pharmaceutical applications. The following step-by-step guide provides manufacturing teams with a reliable framework for implementing this technology at scale while ensuring consistent product quality and process efficiency.
- Combine 2-indolylmethanol and nitrone in toluene solvent with anhydrous sodium sulfate at 1.2: 1 molar ratio under nitrogen atmosphere
- Add chiral phosphoric acid catalyst (G = 9-phenanthrenyl derivative) and hexafluoroisopropanol co-catalyst at room temperature
- Monitor reaction progression via TLC until completion, then purify through silica gel column chromatography with petroleum ether/dichloromethane (1: 1 v/v)
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing through its inherent process advantages. The elimination of transition metal catalysts removes significant cost drivers associated with metal removal processes and specialized waste treatment requirements that typically increase production expenses by multiple factors in conventional syntheses. The use of commercially available, stable catalysts with extended shelf life enhances supply chain resilience by reducing dependency on specialized chemical suppliers with limited production capacity or volatile pricing structures. Furthermore, the ambient temperature operation significantly lowers energy consumption compared to cryogenic or high-temperature processes, contributing to both cost reduction and environmental sustainability goals without compromising product quality or yield consistency.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes entire processing steps required for metal residue removal, which typically account for substantial operational costs in pharmaceutical intermediate production. The use of standard solvents and commercially available catalysts reduces raw material costs while the simplified purification protocol minimizes solvent consumption and processing time compared to conventional methods requiring multiple chromatographic steps or specialized equipment. The high atom economy of this one-step cyclization process significantly reduces material waste generation, lowering both raw material costs and waste disposal expenses while maintaining excellent yield across diverse substrate combinations.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available catalysts with extended shelf life eliminates supply chain vulnerabilities associated with specialized or unstable reagents required in alternative synthetic routes. The ambient temperature operation removes dependencies on specialized cooling or heating infrastructure that can create production bottlenecks during equipment maintenance or seasonal demand fluctuations. The simplified process flow with fewer unit operations reduces potential failure points in manufacturing, enhancing overall production reliability while the broad substrate compatibility provides flexibility to accommodate changing customer requirements without requiring significant process revalidation.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes without requiring specialized equipment modifications, as evidenced by consistent performance across different scales in patent examples. The elimination of hazardous reagents and extreme temperature conditions significantly reduces environmental impact while simplifying regulatory compliance with environmental protection standards. The high-yielding one-step transformation minimizes waste generation compared to multi-step conventional routes, contributing to both sustainability goals and reduced waste treatment costs while maintaining excellent product quality characteristics essential for pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology, based on detailed analysis of the patent's technical disclosure and experimental validation data. These responses provide procurement and supply chain decision-makers with essential insights into process reliability, scalability, and quality assurance aspects critical for commercial adoption.
Q: How does this method achieve high enantioselectivity without transition metals?
A: The dual catalytic system of chiral phosphoric acid and hexafluoroisopropanol creates a stereoselective microenvironment through hydrogen-bonding networks, eliminating the need for transition metals while maintaining >90% ee across diverse substrates as demonstrated in patent examples.
Q: What supply chain advantages does this process offer for pharmaceutical manufacturing?
A: The mild reaction conditions (20-30°C) and commercially available catalysts enable reliable sourcing of raw materials, while the simplified purification process reduces production lead time and eliminates complex metal removal steps required in conventional methods.
Q: Can this synthesis be scaled for commercial API production?
A: Yes, the patent explicitly validates scalability from laboratory to industrial scale with consistent yield and enantioselectivity, using standard equipment and avoiding cryogenic conditions or hazardous reagents that complicate large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex chiral intermediates, with stringent purity specifications consistently met through our rigorous QC labs and advanced analytical capabilities. This patented technology represents a strategic advancement in chiral indoxazinone manufacturing that aligns perfectly with our commitment to delivering high-purity pharmaceutical intermediates through sustainable, scalable processes that meet global regulatory requirements. Our dedicated technical teams have successfully implemented similar catalytic methodologies across multiple therapeutic areas, ensuring seamless technology transfer and rapid scale-up capabilities for this innovative synthesis route while maintaining exceptional product quality standards required by leading pharmaceutical clients worldwide.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements, where our technical procurement team will provide detailed route feasibility assessments and specific COA data demonstrating how this patented methodology can optimize your supply chain for chiral indoxazinone intermediates while ensuring consistent quality and reliable delivery timelines.
