Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously demand robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs to enhance metabolic stability and bioavailability. Patent CN111978265B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives, addressing critical limitations in existing literature. This technology leverages a ferric chloride-promoted cyclization strategy that operates under remarkably mild conditions, avoiding the stringent anhydrous and oxygen-free environments typically required for such transformations. The significance of this scaffold is underscored by its presence in blockbuster drugs such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox, where the triazole core plays a pivotal role in biological activity.
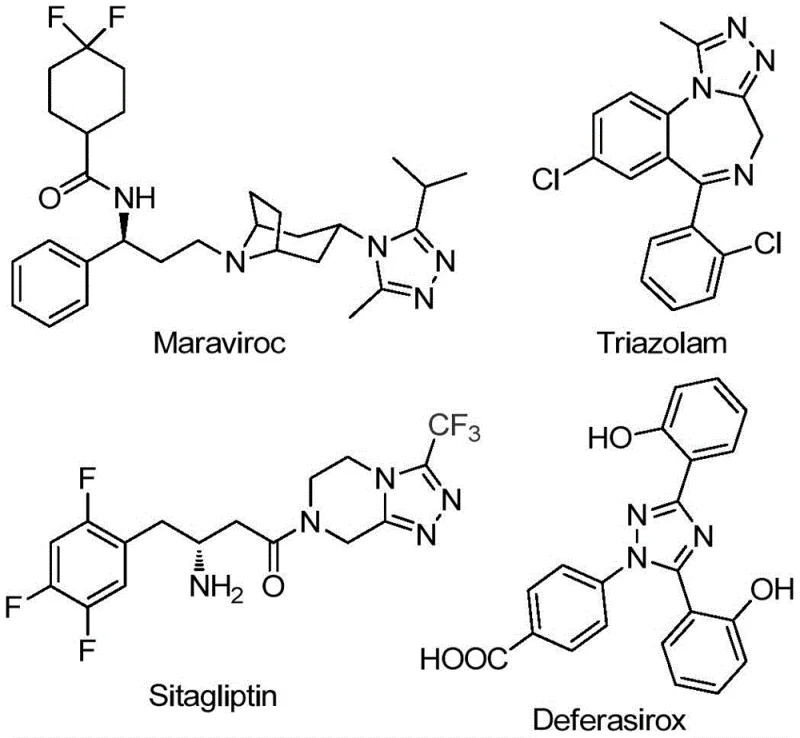
For R&D directors and process chemists, the ability to access these privileged structures efficiently is paramount. The disclosed method not only simplifies the synthetic route but also expands the chemical space accessible to medicinal chemists by tolerating a wide range of functional groups. By utilizing cheap and readily available acyl hydrazides and trifluoroethylimidoyl chlorides, this innovation represents a significant leap forward in the cost reduction in API manufacturing, offering a reliable pathway for the commercial scale-up of complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by substantial operational challenges that hinder efficient production. Traditional routes often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines, processes that frequently suffer from harsh reaction conditions and lengthy multi-step sequences. Furthermore, earlier methodologies exhibited a narrow substrate scope, particularly failing to accommodate alkyl hydrazones, which severely restricted the diversity of analogues available for drug discovery campaigns. These inefficiencies result in lower overall yields and increased waste generation, creating bottlenecks for supply chain heads who require consistent and scalable sources of high-purity intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a tandem cyclization strategy promoted by inexpensive ferric chloride, enabling the direct conversion of trifluoroethylimidoyl chlorides and hydrazides into the desired triazole framework. This method is distinguished by its operational simplicity, proceeding effectively in common organic solvents like 1,4-dioxane without the need for inert atmosphere techniques. The versatility of this route is exemplified by its ability to synthesize a diverse array of derivatives, including those with alkyl, alkenyl, and various substituted aryl groups at the 3-position, as illustrated by the successful preparation of compounds I-1 through I-5.
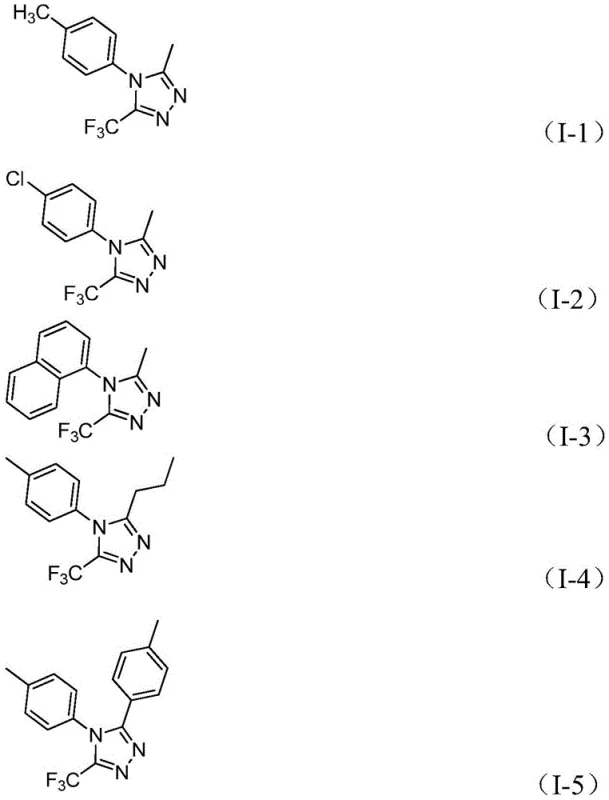
This expanded scope allows for the rapid generation of structural analogues essential for structure-activity relationship (SAR) studies. For procurement managers, the shift towards using commercially available acyl chlorides and hydrazine hydrate as precursors translates to significant supply chain reliability, as these raw materials are widely sourced and cost-effective. The elimination of exotic reagents and the tolerance for ambient air conditions further streamline the manufacturing process, reducing lead time for high-purity pharmaceutical intermediates and ensuring a more resilient supply chain.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and Lewis acid-catalyzed dehydration. Initially, sodium bicarbonate facilitates the intermolecular formation of a carbon-nitrogen bond between the hydrazide and the imidoyl chloride, generating a trifluoroacetamidine intermediate. Subsequently, the addition of ferric chloride acts as a potent Lewis acid promoter, driving the intramolecular dehydration condensation that closes the triazole ring. This dual-activation strategy ensures high conversion rates while minimizing the formation of side products that typically arise from uncontrolled reactivity in harsher acidic or basic environments.
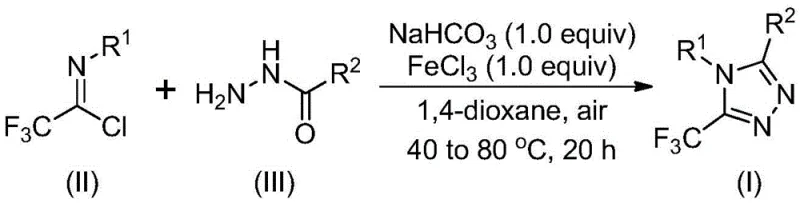
From an impurity control perspective, the mild nature of the FeCl3 catalysis is crucial. Unlike strong mineral acids which might degrade sensitive functional groups or promote polymerization, ferric chloride provides a controlled acidic environment that favors the specific cyclization pathway. This selectivity is vital for maintaining the integrity of the final product, ensuring that the resulting 5-trifluoromethyl-1,2,4-triazole derivatives meet the stringent purity specifications required for pharmaceutical applications. The ability to fine-tune the reaction temperature between 40°C and 80°C further allows process engineers to optimize the balance between reaction rate and selectivity, maximizing yield while suppressing potential degradation pathways.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly attractive for both laboratory scale-up and industrial production. The protocol begins with the mixing of sodium bicarbonate, trifluoroethylimide chloride, and the chosen hydrazide in an organic solvent, followed by a moderate heating phase to establish the initial adduct. The subsequent addition of the iron catalyst triggers the cyclization, after which standard workup procedures involving filtration and silica gel purification yield the target compound. Detailed standardized synthesis steps see the guide below.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an organic solvent like 1,4-dioxane and stir at 30-50°C for 8-16 hours.
- Add ferric chloride (FeCl3) to the reaction mixture and increase the temperature to 70-90°C to continue the reaction for 6-10 hours.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, this patented methodology offers compelling economic and logistical benefits. The transition from complex, multi-step traditional syntheses to this streamlined one-pot style process drastically reduces the consumption of solvents, energy, and labor hours. By leveraging earth-abundant iron catalysis instead of precious metals, the process inherently lowers the cost of goods sold (COGS) and simplifies the regulatory burden associated with heavy metal residue limits in final drug substances.
- Cost Reduction in Manufacturing: The utilization of ferric chloride and sodium bicarbonate represents a massive cost advantage over expensive transition metal catalysts or specialized reagents often required in heterocycle synthesis. Since the reaction does not require rigorous exclusion of moisture or oxygen, the capital expenditure for specialized reactor equipment is significantly minimized, allowing for production in standard glass-lined or stainless steel vessels. This simplification of infrastructure directly translates to substantial cost savings in both CAPEX and OPEX for manufacturing facilities.
- Enhanced Supply Chain Reliability: The starting materials, specifically acyl chlorides and hydrazides, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by environmental factors or minor variations in raw material quality. This reliability ensures a steady flow of high-purity intermediates, critical for maintaining uninterrupted API production lines and meeting tight delivery windows for downstream customers.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from milligram to gram levels with consistent yields, indicating strong potential for ton-scale production. The use of 1,4-dioxane, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols. Furthermore, the avoidance of stoichiometric amounts of hazardous oxidizing or reducing agents reduces the generation of toxic waste streams, aligning with modern green chemistry principles and facilitating easier environmental compliance permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy for technical decision-makers. Understanding these nuances is essential for evaluating the fit of this technology within your specific development pipeline.
Q: What are the key advantages of this FeCl3-catalyzed method over traditional synthesis?
A: This method eliminates the need for harsh anhydrous or oxygen-free conditions, utilizes cheap and readily available starting materials like hydrazides, and offers a broader substrate scope including alkyl hydrazones which previous methods failed to convert efficiently.
Q: What represents the optimal reaction conditions for high yield?
A: The optimal protocol involves using 1,4-dioxane as the solvent with a molar ratio of trifluoroethylimide chloride to hydrazide of approximately 1:1.5, utilizing sodium bicarbonate as the base and ferric chloride as the promoter at temperatures ranging from 40°C to 80°C.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable as it avoids sensitive reaction conditions, uses inexpensive catalysts, and has been demonstrated to work effectively on gram scales with straightforward post-processing via filtration and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocycle synthesis in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole derivatives delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced FeCl3-catalyzed platform can serve as a cornerstone for your next successful commercial campaign.
