Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharma
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharma
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable manufacturing processes for bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are not merely academic curiosities; they serve as critical building blocks in the development of advanced pharmaceutical agents and functional materials, including ligands for organic light-emitting diodes (OLEDs). The innovation lies in shifting away from multi-step, low-yield traditional syntheses toward a direct, oxidative cyclization strategy that leverages cheap, commercially available starting materials. By utilizing a metal-free catalytic system, this technology addresses key pain points in modern chemical manufacturing, offering a pathway to high-purity intermediates with drastically simplified operational protocols.
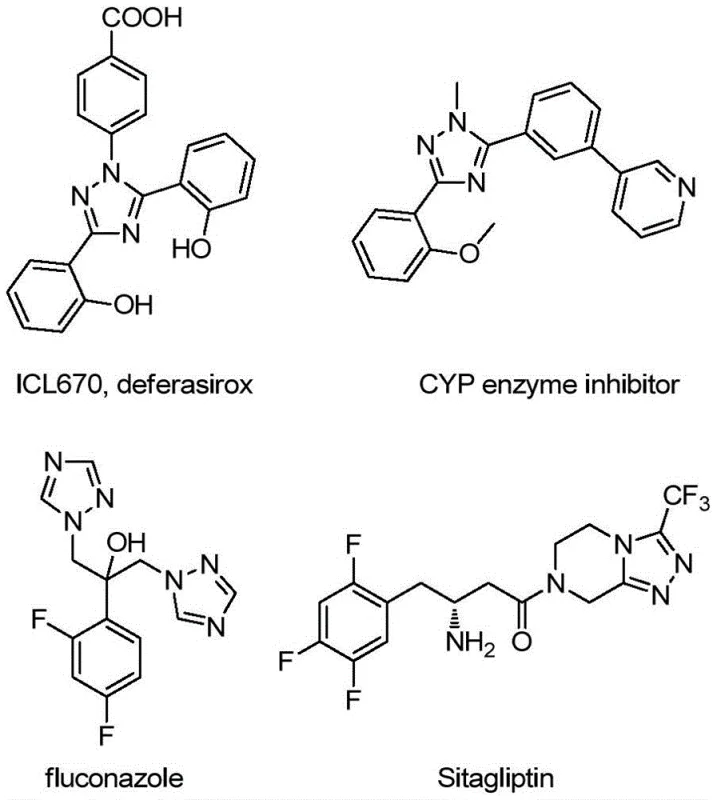
The versatility of the 1,2,4-triazole ring system is evident in its presence across a diverse array of high-value applications, ranging from iron chelators like deferasirox to antifungal agents such as fluconazole and DPP-4 inhibitors like sitagliptin. The ability to efficiently construct these rings, particularly when fused or linked with quinoline systems, opens new avenues for drug discovery and material science. The methodology described in the patent provides a reliable foundation for generating these complex architectures, ensuring that research and development teams have access to the necessary chemical diversity without being bottlenecked by inefficient synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale production. Traditional protocols often rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This multi-step approach is not only time-consuming but also suffers from poor atom economy and cumulative yield losses, with reported total yields hovering around a mere 17%. Furthermore, these conventional routes frequently demand severe reaction conditions, including the use of harsh reagents and strict environmental controls, which escalate operational costs and safety risks. For procurement managers and supply chain heads, such inefficiencies translate into higher raw material costs, extended lead times, and increased waste generation, making the traditional pathway economically unsustainable for commercial-scale manufacturing of these valuable intermediates.
The Novel Approach
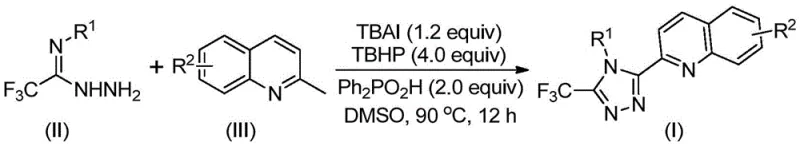
In stark contrast, the novel method disclosed in patent CN113307790B revolutionizes the synthesis by employing a direct oxidative cyclization strategy. This approach utilizes 2-methylquinoline and trifluoroacetimidohydrazide as readily accessible starting materials, bypassing the need for pre-functionalized carboxylic acid derivatives. The reaction is promoted by a synergistic catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), facilitated by diphenylphosphoric acid as an additive. This one-pot transformation proceeds smoothly at temperatures between 80°C and 100°C, eliminating the need for complex multi-step sequences. The result is a dramatic improvement in efficiency, with isolated yields reaching as high as 97% for certain substrates, representing a paradigm shift in how these heterocyclic compounds are manufactured. This streamlined process not only enhances throughput but also significantly reduces the environmental footprint associated with chemical synthesis.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway facilitated by the TBAI/TBHP system. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it in situ into a reactive aldehyde equivalent. This activated species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination triggers an intramolecular electrophilic substitution, leading to ring closure. The final step involves aromatization to yield the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole structure. This cascade of events occurs within a single reaction vessel, showcasing the power of modern catalytic design to simplify complex molecular constructions. The involvement of diphenylphosphoric acid further stabilizes the reaction environment, ensuring high conversion rates and minimizing the formation of unwanted by-products.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. By avoiding the use of transition metal catalysts, the process eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates. The absence of metal residues simplifies the downstream purification process, often removing the need for expensive scavenging resins or complex extraction protocols. Furthermore, the mild reaction conditions and the specificity of the oxidative cyclization reduce the likelihood of side reactions such as over-oxidation or polymerization. This results in a cleaner crude reaction profile, allowing for easier isolation of the target compound via standard column chromatography or crystallization techniques, thereby ensuring the delivery of high-purity products suitable for sensitive downstream applications.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be robust, tolerating a wide range of substituents on both the quinoline and the hydrazide components, which allows for the generation of a diverse library of analogues. The use of dimethyl sulfoxide (DMSO) as the preferred solvent ensures excellent solubility of all reactants and promotes the oxidative cycle effectively. Operators should note that while the reaction does not require strict anhydrous or anaerobic conditions, maintaining consistent temperature control between 80°C and 100°C is vital for optimal performance. The following guide outlines the standardized procedure derived from the patent examples, providing a clear roadmap for technical teams to replicate these successful outcomes.
- Combine tetrabutylammonium iodide, tert-butyl peroxide, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, this patented methodology offers compelling economic and operational benefits. The shift from a five-step sequence to a single-pot reaction fundamentally alters the cost structure of manufacturing these intermediates. By reducing the number of unit operations, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. Moreover, the elimination of expensive transition metal catalysts removes a major cost driver and simplifies the supply chain for raw materials. The ability to operate without stringent moisture or oxygen exclusion further reduces infrastructure requirements, allowing for production in standard reactor setups rather than specialized glovebox environments. These factors combine to create a highly competitive manufacturing process that aligns with the goals of cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic reduction in synthetic steps and the use of commodity chemicals. Traditional routes involving quinoline-2-carboxylic acid are inherently expensive due to the cost of the starting material and the cumulative losses over five steps. In contrast, the new method utilizes 2-methylquinoline, a bulk chemical available at a fraction of the cost. Additionally, the catalyst system relies on tetrabutylammonium iodide and tert-butyl peroxide, which are inexpensive and widely sourced. The removal of heavy metal catalysts also negates the need for costly purification steps to meet regulatory limits on residual metals, further driving down the overall cost of goods sold (COGS) and improving margin potential for commercial production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by reliance on specialized or scarce reagents. This synthesis mitigates such risks by relying on starting materials that are commercially available in large quantities from multiple global suppliers. 2-methylquinoline and the requisite hydrazides are established industrial chemicals with stable supply lines, reducing the risk of procurement bottlenecks. Furthermore, the robustness of the reaction conditions means that production is less susceptible to delays caused by environmental control failures or equipment limitations. This reliability ensures consistent delivery schedules for downstream customers, fostering stronger partnerships and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this method is inherently scalable due to its simplicity and safety profile. The reaction operates at moderate temperatures and does not involve hazardous pyrophoric reagents or high-pressure hydrogenation, making it safer to run in large-scale reactors. From an environmental standpoint, the reduction in waste generation—stemming from fewer reaction steps and higher atom economy—aligns with green chemistry principles. The absence of toxic heavy metals simplifies waste treatment and disposal, lowering compliance costs and minimizing the environmental footprint of the manufacturing facility, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthesis method is crucial for making informed sourcing decisions. The following questions address common inquiries regarding the practical application and scope of this technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a transparent view of what can be expected during technology transfer and commercial adoption. Whether you are concerned about substrate scope or purification requirements, these insights aim to clarify the operational realities of producing 3-quinolyl-5-trifluoromethyl-1,2,4-triazole derivatives.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes a metal-free catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), eliminating the need for costly heavy metals and subsequent removal steps.
Q: What are the typical reaction conditions for this oxidative cyclization?
A: The reaction operates under mild thermal conditions, typically between 80°C and 100°C, in an organic solvent like DMSO, without requiring strict anhydrous or anaerobic environments.
Q: How does the yield compare to traditional quinoline-2-carboxylic acid routes?
A: Traditional methods often suffer from low total yields around 17% over five steps, whereas this novel one-pot approach achieves significantly higher isolated yields, reaching up to 97% for specific substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free oxidative cyclization technology for the pharmaceutical and fine chemical industries. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to seamlessly adopt such innovative pathways, ensuring that our clients benefit from the latest advancements in synthetic chemistry. Our facilities are equipped to handle complex heterocyclic synthesis with precision, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole intermediate meets the highest standards required for drug substance manufacturing and advanced material applications.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this efficient synthesis route for their projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing capabilities.
