Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral scaffolds that serve as critical building blocks in modern drug discovery and catalysis. Patent CN110452150B introduces a groundbreaking methodology for the preparation of axial chiral indole-naphthalene compounds, a class of molecules renowned for their utility as organocatalysts and ligands. This innovation addresses a significant gap in the prior art by providing a robust, one-step synthetic route that constructs the complex axial chiral skeleton directly from racemic starting materials. Unlike traditional methods that often struggle with low efficiency or require harsh conditions, this approach leverages the power of chiral phosphoric acid catalysis to achieve exceptional stereocontrol. The resulting compounds, characterized by the general structure shown below, exhibit high optical purity and structural diversity, making them indispensable assets for the development of next-generation asymmetric transformations.
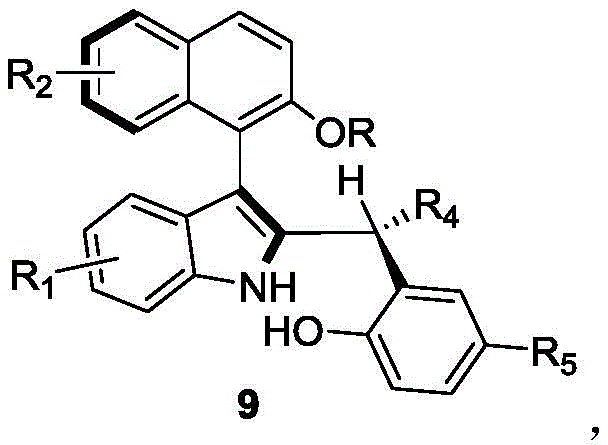
For research and development teams focused on process optimization, the ability to access these complex biaryl systems through a streamlined pathway represents a substantial technical advancement. The patent details a versatile substrate scope, accommodating various substituents on both the indole and naphthalene rings, which allows for the fine-tuning of steric and electronic properties essential for catalyst design. By utilizing a dynamic kinetic resolution mechanism, the process effectively converts racemic mixtures into single enantiomers, maximizing atom economy and reducing waste. This capability is particularly valuable for a reliable pharmaceutical intermediate supplier aiming to deliver high-value chiral intermediates with consistent quality and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral indole-naphthalene frameworks has been fraught with challenges that hinder large-scale adoption and cost-effectiveness. Conventional strategies predominantly rely on cross-coupling reactions between pre-functionalized indole and naphthalene units, which often necessitate expensive transition metal catalysts and rigorous exclusion of air and moisture. These methods frequently suffer from limited substrate tolerance, where bulky substituents can severely impede the coupling efficiency, leading to poor yields and difficult purification processes. Furthermore, achieving high enantioselectivity in these coupling reactions typically requires intricate chiral ligands and multiple synthetic steps to install the axial chirality, significantly increasing the overall production cost and lead time. The reliance on heavy metals also introduces downstream complications regarding residual metal removal, a critical quality attribute for pharmaceutical applications that adds further complexity and expense to the manufacturing workflow.
The Novel Approach
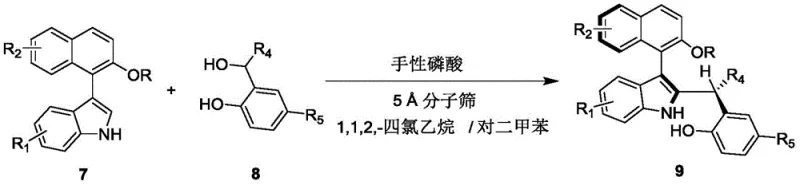
In stark contrast, the methodology disclosed in the patent offers a paradigm shift by employing an asymmetric addition reaction catalyzed by small organic molecules. As illustrated in the reaction scheme below, the process combines a compound of formula 7 with a compound of formula 8 in the presence of a chiral phosphoric acid catalyst. This direct approach bypasses the need for pre-functionalized coupling partners and transition metals, operating under remarkably mild conditions ranging from 20 to 30°C. The use of a mixed solvent system comprising 1,1,2,2-tetrachloroethane and p-xylene, along with 5 Angstrom molecular sieves, creates an optimal environment for the dynamic kinetic resolution to proceed efficiently. This novel route not only simplifies the operational procedure but also dramatically enhances the enantioselectivity, delivering products with er values as high as 98:2, thereby establishing a new standard for cost reduction in chiral catalyst manufacturing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The success of this synthesis hinges on the precise interaction between the chiral phosphoric acid catalyst and the substrates, which facilitates a highly stereoselective transformation. The catalyst, typically a binaphthyl derivative such as the compound of formula 6 featuring a 9-anthracenyl group, acts as a bifunctional activator. It simultaneously activates the electrophilic species through hydrogen bonding interactions while organizing the nucleophile within a well-defined chiral pocket. This dual activation mode lowers the energy barrier for the desired reaction pathway while sterically blocking the formation of the undesired enantiomer. The rigidity of the catalyst's backbone, reinforced by the bulky anthracenyl substituents, ensures that the axial chirality is induced with high fidelity during the bond-forming event. Understanding this mechanistic nuance is crucial for scaling up the process, as it highlights the importance of maintaining strict control over reaction parameters such as temperature and solvent composition to preserve the integrity of the chiral environment.
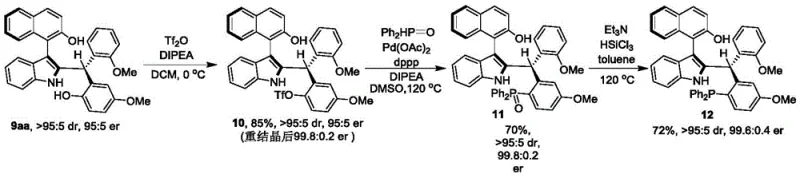
Beyond the primary synthesis, the utility of these compounds extends to their application as precursors for other potent catalysts. The patent demonstrates that the resulting axial chiral indole-naphthalene compound 9aa can be chemically modified to generate chiral phosphine catalysts, such as compound 12. This derivatization involves converting the hydroxyl groups into phosphine moieties, which retain the axial chirality of the parent scaffold. As shown in the subsequent transformation, these derived phosphine catalysts are capable of promoting complex asymmetric reactions, such as the [4+1] cyclization of o-hydroxy-p-benzoquinone with Morita-Baylis-Hillman esters. This downstream applicability underscores the strategic value of the initial synthesis, providing a versatile platform for generating a library of chiral ligands tailored for specific asymmetric transformations in fine chemical synthesis.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The experimental protocol outlined in the patent provides a clear and reproducible pathway for producing these high-value intermediates. The procedure emphasizes the use of readily available reagents and standard laboratory equipment, making it highly accessible for process development teams. Key to the success is the activation of the molecular sieves and the precise control of the solvent ratio, which ensures the removal of water generated during the reaction and drives the equilibrium towards the product. The reaction is monitored via thin-layer chromatography (TLC) to determine the endpoint, ensuring maximum conversion before workup. Following the reaction, a straightforward filtration and concentration sequence followed by silica gel column chromatography yields the pure product. For detailed operational parameters and safety considerations, the standardized synthesis steps are provided below.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene (volume ratio 1: 4).
- Add 5 Angstrom molecular sieves and a chiral phosphoric acid catalyst (such as compound of formula 6) to the reaction vessel.
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that directly address the pain points of sourcing complex chiral intermediates. The shift from transition metal catalysis to organocatalysis eliminates the dependency on scarce and volatile precious metals, thereby stabilizing raw material costs and mitigating supply chain risks associated with geopolitical fluctuations in metal availability. Moreover, the simplified workup procedure, which avoids extensive heavy metal scavenging steps, significantly reduces the consumption of auxiliary materials and shortens the overall production cycle time. This streamlining of the manufacturing process translates into substantial cost savings and enhanced throughput, allowing suppliers to offer more competitive pricing without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification steps drastically lowers the bill of materials for production. By utilizing organocatalysts that can be recovered or used in low loadings, the process minimizes the input cost per kilogram of the final product. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, further contributing to a leaner manufacturing cost structure that benefits the entire value chain.
- Enhanced Supply Chain Reliability: The starting materials, compounds of formula 7 and 8, are synthesized from common industrial feedstocks, ensuring a stable and continuous supply base. The robustness of the reaction against minor variations in conditions means that batch-to-batch consistency is easier to maintain, reducing the risk of production delays due to failed batches. This reliability is critical for long-term supply agreements where uninterrupted delivery of high-purity intermediates is paramount for downstream drug manufacturing schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard solvents and ambient temperatures, which simplifies the engineering requirements for scale-up from pilot to commercial plant. The absence of toxic heavy metals in the final product simplifies regulatory compliance and waste disposal, aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint but also lowers the costs associated with waste treatment and regulatory reporting.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the feasibility of integrating this technology into their supply chains, we have compiled answers to common technical inquiries based on the patent specifications. These questions address the core aspects of catalyst selection, reaction scope, and product application, providing a clear understanding of the method's capabilities. The responses are derived directly from the experimental data and examples provided in the intellectual property documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: This method utilizes a dynamic kinetic resolution strategy via chiral phosphoric acid catalysis, allowing for the one-step construction of the axial chiral skeleton from racemic raw materials with high enantioselectivity (up to 98:2 er) and excellent yields, avoiding complex multi-step coupling sequences.
Q: What type of catalyst is employed in this process?
A: The process employs a chiral phosphoric acid catalyst, specifically derivatives with binaphthyl, octahydrobinaphthyl, or spiro skeletons, with compound of formula 6 (containing a 9-anthracenyl group) being particularly effective for high stereocontrol.
Q: Can the resulting compounds be used directly as catalysts?
A: Yes, the synthesized axial chiral indole-naphthalene compounds serve as valuable precursors. They can be derivatized into chiral phosphine catalysts (such as compound 12) which effectively catalyze asymmetric reactions like the [4+1] cyclization of o-hydroxy-p-benzoquinone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the production of advanced chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including high enantiomeric ratios essential for chiral catalyst applications. We are committed to leveraging this innovative organocatalytic technology to deliver high-quality axial chiral indole-naphthalene derivatives that meet the exacting standards of the global pharmaceutical and fine chemical industries.
We invite you to collaborate with us to explore how this efficient synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our available batches and to discuss route feasibility assessments for your upcoming projects. Together, we can accelerate the development of your chiral programs with a reliable and cost-effective supply solution.
