Advanced Chiral Zinc-Nitrogen Complexes for High-Precision Asymmetric Synthesis
Advanced Chiral Zinc-Nitrogen Complexes for High-Precision Asymmetric Synthesis
The landscape of modern organic synthesis is continually evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal development in this domain is documented in patent CN102887912A, which introduces a novel chiral zinc-nitrogen complex designed to serve as a robust catalyst for asymmetric transformations. This specific metal-organic coordination compound represents a significant leap forward in ligand design, moving beyond traditional oxazoline systems to incorporate a hybrid pyridine-oxazoline architecture. For R&D directors and procurement specialists seeking a reliable chiral catalysts supplier, understanding the structural nuances and synthetic accessibility of this complex is paramount. The patent details a rigorous preparation method involving the coordination of zinc chloride with a specifically designed chiral ligand derived from L-leucinol, resulting in a stable crystalline material with defined stereochemistry. This innovation addresses the critical need for catalysts that offer both high enantioselectivity and operational stability under varied reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has relied heavily on simple oxazoline ligands coordinated with transition metals. While effective in many scenarios, these conventional systems often suffer from limitations regarding thermal stability and substrate scope. Traditional zinc-based catalysts frequently exhibit sensitivity to moisture and air, necessitating stringent handling protocols that increase operational costs and complexity in a manufacturing setting. Furthermore, the rigidity of the chiral pocket in older generation ligands may not provide sufficient steric differentiation for bulky substrates, leading to suboptimal enantiomeric excess (ee) values. In large-scale pharmaceutical intermediate manufacturing, these inefficiencies translate to higher waste generation and increased purification burdens. The reliance on expensive noble metals in some alternative catalytic systems also poses a significant cost barrier, prompting the industry to seek earth-abundant metal alternatives like zinc that do not compromise on performance.
The Novel Approach
The approach detailed in the patent data offers a sophisticated solution by engineering a tetranuclear or polynuclear zinc structure stabilized by bulky isobutyl groups on the oxazoline ring. This novel architecture enhances the steric environment around the active zinc center, potentially improving substrate discrimination during catalytic cycles. The synthesis utilizes 6-methyl-2-cyano-3-hydroxypyridine and L-leucinol, reacting them in the presence of anhydrous zinc chloride in chlorobenzene. This method ensures the formation of a robust Zn-N and Zn-O coordination network that is less susceptible to decomposition compared to simpler analogues. 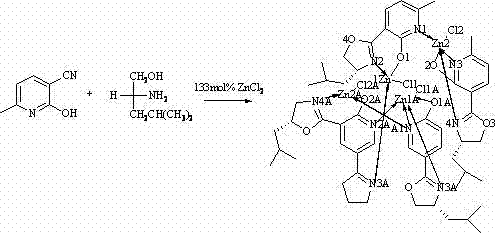 By leveraging this specific ligand framework, the new complex provides a versatile platform for Lewis acid catalysis, particularly in reactions requiring precise spatial control such as cyanosilylation. This represents a strategic advancement for cost reduction in chiral catalysts manufacturing, as it utilizes readily available amino alcohol derivatives and avoids the need for exotic metal precursors.
By leveraging this specific ligand framework, the new complex provides a versatile platform for Lewis acid catalysis, particularly in reactions requiring precise spatial control such as cyanosilylation. This represents a strategic advancement for cost reduction in chiral catalysts manufacturing, as it utilizes readily available amino alcohol derivatives and avoids the need for exotic metal precursors.
Mechanistic Insights into Zn-N Coordination and Catalytic Activity
To fully appreciate the utility of this material for high-purity pharmaceutical intermediates, one must examine the crystallographic data provided in the patent. The single-crystal X-ray diffraction analysis reveals a monoclinic crystal system with space group C2, indicating a highly ordered and chiral arrangement of the molecules within the lattice. The zinc centers exhibit a distorted tetrahedral or trigonal bipyramidal geometry, coordinated by nitrogen atoms from the pyridine and oxazoline rings, as well as oxygen atoms from the phenolic hydroxyl groups. 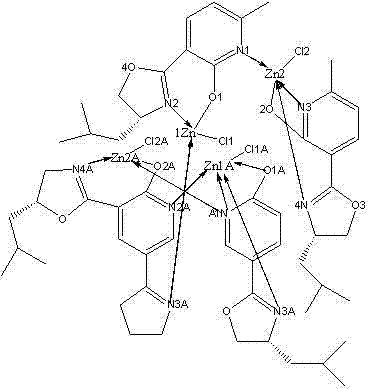 Key bond lengths, such as Zn(1)-N(1) at 2.050 Angstroms and Zn(1)-O(2) at 1.953 Angstroms, confirm strong covalent character in the metal-ligand interactions, which is crucial for maintaining catalyst integrity during reflux conditions. The presence of chlorine atoms in the coordination sphere, as indicated by Zn-Cl bond lengths around 2.2 Angstroms, suggests that the complex may act as a Lewis acid by dissociating these labile ligands to create open coordination sites for substrate binding. This mechanistic understanding allows chemists to predict reactivity patterns and optimize reaction parameters for specific synthetic targets.
Key bond lengths, such as Zn(1)-N(1) at 2.050 Angstroms and Zn(1)-O(2) at 1.953 Angstroms, confirm strong covalent character in the metal-ligand interactions, which is crucial for maintaining catalyst integrity during reflux conditions. The presence of chlorine atoms in the coordination sphere, as indicated by Zn-Cl bond lengths around 2.2 Angstroms, suggests that the complex may act as a Lewis acid by dissociating these labile ligands to create open coordination sites for substrate binding. This mechanistic understanding allows chemists to predict reactivity patterns and optimize reaction parameters for specific synthetic targets.
Furthermore, the impurity profile of the synthesized complex is tightly controlled through the use of anhydrous and anaerobic conditions during the 48-hour reflux period. The rigorous purification protocol involving column chromatography with petroleum ether and dichloromethane ensures that unreacted starting materials and side products are effectively removed. This level of purity is essential for R&D teams focusing on impurity control mechanisms, as trace metal contaminants or free ligands can interfere with downstream reactions or complicate regulatory filings for drug substances. The specific rotation value of +30.0 degrees (c=0.08, THF) serves as a critical quality attribute (CQA) for batch-to-batch consistency, ensuring that the chiral information is faithfully transmitted from the L-leucinol starting material to the final metal complex. Such detailed characterization data provides the confidence needed for scaling up complex organometallic syntheses in a GMP environment.
How to Synthesize Chiral Zinc Complex Efficiently
The preparation of this advanced catalytic material requires strict adherence to moisture-free protocols to prevent hydrolysis of the zinc-chloride bonds. The process begins with the precise weighing of anhydrous zinc chloride and the chiral ligand precursors in a glovebox or under a nitrogen blanket. Solvent selection is critical; chlorobenzene is chosen for its high boiling point, which facilitates the prolonged heating necessary for complete complexation without solvent loss. Following the reaction, the workup procedure involves careful phase separation and drying to isolate the white crystalline product. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps below.
- Combine anhydrous ZnCl2, 6-methyl-2-cyano-3-hydroxypyridine, and L-leucinol in chlorobenzene solvent under strict anhydrous and anaerobic conditions.
- Reflux the mixture at high temperature for 48 hours to ensure complete coordination and complex formation.
- Remove solvent under reduced pressure, extract the residue with chloroform, and purify the crude product via column chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this zinc-based technology offers several compelling advantages over traditional noble metal catalysts. The primary benefit lies in the raw material availability; L-leucinol and substituted pyridines are commodity chemicals with stable global supply chains, reducing the risk of procurement bottlenecks. This stability is crucial for ensuring continuous production schedules for high-value fine chemicals. Moreover, the elimination of expensive metals like palladium or rhodium from the catalyst formulation leads to substantial cost savings in the overall bill of materials. These savings can be passed down the value chain, making the final pharmaceutical intermediates more price-competitive in the global market. The robustness of the complex also implies a longer shelf life and reduced waste due to degradation, further enhancing the economic efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The shift to a zinc-based system significantly lowers the raw material costs associated with catalyst procurement. Unlike precious metal catalysts that require expensive recovery processes to meet environmental regulations, zinc residues are easier to manage and dispose of, reducing the overhead for waste treatment facilities. The synthetic route described avoids the use of cryogenic conditions or ultra-high vacuum equipment, allowing the reaction to be performed in standard glass-lined reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure minimizes capital expenditure requirements for technology transfer. Additionally, the high atom economy of the ligand synthesis ensures that minimal material is wasted during the preparation of the catalyst itself, contributing to a leaner and more sustainable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing chiral building blocks can often be a challenge due to limited suppliers of enantiopure materials. However, the reliance on L-leucinol, a derivative of a common natural amino acid, ensures a diverse and competitive supplier base. This diversity mitigates the risk of single-source dependency, a critical factor for supply chain heads managing long-term contracts. The synthetic pathway is straightforward and does not rely on proprietary reagents that might be subject to export controls or trade restrictions. Consequently, manufacturers can maintain higher safety stocks of the finished catalyst without worrying about rapid degradation, providing a buffer against market volatility. This reliability is essential for meeting the just-in-time delivery expectations of major pharmaceutical clients who cannot afford production delays.
- Scalability and Environmental Compliance: The process described operates at atmospheric pressure and uses solvents that are well-understood in terms of recovery and recycling. Chlorobenzene, while requiring careful handling, has established protocols for distillation and reuse, aligning with green chemistry principles. The absence of toxic heavy metals simplifies the environmental impact assessment and regulatory compliance procedures for new drug applications. Scaling this reaction from the laboratory gram scale to multi-kilogram production is feasible with standard agitation and heating systems, as the reaction kinetics are not dependent on specialized mixing technologies. This scalability ensures that as demand for the downstream chiral intermediates grows, the supply of the catalyst can be ramped up seamlessly without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the handling and application of this chiral zinc complex. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these details is vital for process chemists integrating this catalyst into their synthetic routes. We encourage technical teams to review these points to ensure optimal usage and safety.
Q: What is the primary structural feature of this chiral zinc complex?
A: The complex features a unique coordination environment where zinc centers are bound to nitrogen and oxygen atoms from the oxazoline-pyridine hybrid ligands, creating a rigid chiral pocket essential for enantioselective catalysis.
Q: What are the storage requirements for this organometallic catalyst?
A: Due to its sensitivity to moisture and oxygen during synthesis, the final complex should be stored in a cool, dry place, preferably under an inert atmosphere, to maintain its structural integrity and catalytic activity.
Q: Can this complex be scaled for industrial pharmaceutical production?
A: Yes, the synthesis utilizes commercially available starting materials like L-leucinol and standard solvents like chlorobenzene, making the pathway amenable to scale-up with appropriate engineering controls for solvent recovery and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic systems play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our chiral intermediates meets the highest international standards. Our commitment to quality assurance means that you receive materials with consistent optical rotation and metal content, minimizing the risk of batch failures in your critical synthesis steps.
We invite you to contact our technical procurement team to discuss how this chiral zinc technology can be integrated into your current pipeline. We are prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Reach out today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and innovation in your chemical manufacturing operations.
