Advanced Pd-Catalyzed Synthesis of 2-Pyridyloxydiaryl Ketones for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Benzophenone Synthesis
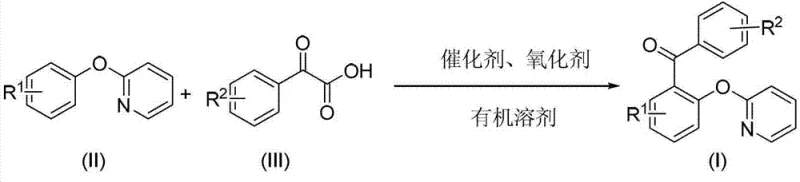
The landscape of pharmaceutical intermediate manufacturing is constantly evolving towards greener, more atom-economical processes. A significant breakthrough in this domain is documented in Chinese Patent CN103242223A, which discloses a novel preparation method for 2-pyridyloxydiaryl ketone derivatives. These compounds serve as critical precursors for a wide array of bioactive molecules, including anti-colon cancer agents found in natural products like Garcinia xanthochymus and non-nucleoside reverse transcriptase inhibitors (NNRTIs) for HIV-1 treatment. The patented methodology leverages a palladium-catalyzed oxidative decarboxylative coupling strategy, representing a paradigm shift from traditional stoichiometric approaches. By utilizing readily available 2-pyridyloxyaryl compounds and arylformylformic acids, this route achieves high structural diversity while maintaining operational simplicity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and commercial viability of this transformation is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl ketones and their functionalized derivatives has relied heavily on Friedel-Crafts acylation or the coupling of organometallic reagents with acid chlorides. These classical pathways often suffer from significant drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Friedel-Crafts reactions typically require harsh Lewis acids in stoichiometric quantities, generating substantial amounts of corrosive waste and posing severe challenges for downstream purification. Furthermore, regioselectivity can be difficult to control, leading to complex impurity profiles that require extensive chromatographic separation. Alternatively, methods involving pre-functionalized organometallic species (such as Grignard or organolithium reagents) demand strictly anhydrous conditions and cryogenic temperatures, drastically increasing energy consumption and operational costs. The generation of stoichiometric metal salts as by-products further complicates waste management, creating a heavy burden on environmental compliance teams.
The Novel Approach
In stark contrast, the method described in CN103242223A introduces a catalytic cycle that bypasses the need for pre-halogenated substrates or stoichiometric organometallics. This approach utilizes the pyridine nitrogen atom as an intrinsic directing group to facilitate ortho-C-H activation, enabling direct functionalization of the aromatic ring. The use of arylformylformic acids as coupling partners is particularly innovative; these reagents undergo decarboxylation during the reaction, releasing carbon dioxide gas as a benign by-product. This feature not only drives the reaction equilibrium forward but also eliminates the formation of heavy metal waste salts associated with traditional cross-couplings. The reaction proceeds in common organic solvents like dioxane or acetic acid at moderate temperatures ranging from 90°C to 120°C. This mildness ensures compatibility with a broad range of functional groups, including halogens, alkyls, and alkoxy substituents, thereby offering superior substrate design flexibility for medicinal chemists.
Mechanistic Insights into Pd-Catalyzed Oxidative Decarboxylative Coupling
The core of this technology lies in a sophisticated palladium catalytic cycle mediated by a silver oxidant. The reaction initiates with the coordination of the divalent palladium catalyst to the nitrogen atom of the pyridine ring in the 2-pyridyloxyaryl substrate. This coordination directs the palladium center to activate the proximal ortho-C-H bond, forming a stable five-membered palladacycle intermediate. Subsequently, this organopalladium species undergoes transmetallation with the arylformylformic acid. The resulting intermediate then experiences a rapid decarboxylation event, extruding carbon dioxide and generating a diaryl-palladium complex. The final step involves reductive elimination, which releases the desired 2-pyridyloxydiaryl ketone product and regenerates a zero-valent palladium species. To close the catalytic loop, the monovalent silver oxidant re-oxidizes the zero-valent palladium back to its active divalent state, ready for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages for high-purity API intermediate manufacturing. Since the by-products are primarily gaseous carbon dioxide and water (from the oxidant reduction), the crude reaction mixture is remarkably clean compared to methods producing inorganic salts. The absence of halogenated leaving groups minimizes the risk of homocoupling side reactions often seen in Suzuki or Stille couplings. Furthermore, the directing group strategy ensures high regioselectivity, preventing the formation of meta- or para-substituted isomers that are difficult to separate. This inherent selectivity reduces the load on purification units, allowing for simpler workup procedures such as filtration and standard silica gel chromatography. For quality assurance teams, this translates to a more robust process capable of consistently meeting stringent purity specifications required for clinical trial materials.
How to Synthesize 2-Pyridyloxydiaryl Ketone Efficiently
Implementing this synthesis requires careful optimization of catalyst loading and oxidant ratios to maximize turnover numbers while minimizing precious metal usage. The patent outlines a straightforward protocol where the catalyst, oxidant, and substrates are combined in a Schlenk tube or reactor with an appropriate organic solvent. The reaction progress is conveniently monitored via thin-layer chromatography (TLC), typically reaching completion within 6 to 12 hours depending on the electronic nature of the substituents. Post-reaction processing is designed for efficiency; the mixture is filtered to remove reduced silver species, and the filtrate is concentrated. Final purification is achieved through column chromatography using petroleum ether as the eluent. For detailed standard operating procedures and specific molar ratios tailored to your specific substrate, please refer to the standardized synthesis guide below.
- Combine 2-pyridyloxyaryl compound, arylformylformic acid, Pd(II) catalyst, and Ag(I) oxidant in an organic solvent such as dioxane or acetic acid.
- Heat the reaction mixture to 90-120°C and stir for 6-12 hours under monitoring by TLC until completion.
- Filter the mixture, purify via silica gel column chromatography using petroleum ether to isolate the target 2-pyridyloxydiaryl ketone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic methodology represents a strategic opportunity for cost reduction in pharmaceutical intermediate manufacturing. The primary economic driver is the elimination of expensive and hazardous pre-functionalized starting materials. By utilizing simple arylformylformic acids and phenol-derived ethers, the raw material costs are significantly lowered compared to purchasing aryl halides or organoboron reagents. Additionally, the atom economy of the reaction is superior because the carboxyl group acts as a traceless activating moiety that departs as gas, meaning less mass needs to be processed and disposed of in the waste stream. This reduction in waste volume directly correlates to lower disposal fees and reduced environmental compliance overhead, contributing to substantial cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The use of earth-abundant silver oxidants and recyclable palladium catalysts optimizes the cost profile of the reaction. Unlike traditional methods that generate stoichiometric amounts of salt waste requiring expensive treatment, this process produces benign by-products. The simplified downstream processing, which avoids complex aqueous workups for salt removal, reduces solvent consumption and labor hours. Consequently, the overall cost of goods sold (COGS) for these intermediates is drastically improved, allowing for more competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted arylformylformic acids and pyridyl ethers, are commercially available from multiple global vendors. This diversity in sourcing mitigates the risk of supply disruptions that often plague single-source specialty reagents. Furthermore, the reaction conditions are robust and tolerant to minor variations in temperature and mixing, ensuring consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with strong Lewis acids. The generation of CO2 gas is easily managed with standard venting systems, posing no safety risks at large scales. From an environmental standpoint, the process aligns with green chemistry principles by minimizing E-factors (mass of waste per mass of product). This eco-friendly profile simplifies the permitting process for new manufacturing lines and enhances the corporate sustainability metrics of the supply chain, which is increasingly important for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for feasibility assessments. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing process workflows and quality frameworks.
Q: What are the primary environmental advantages of this synthesis method?
A: The process generates carbon dioxide and water as the sole by-products, significantly reducing hazardous waste treatment costs compared to traditional halogenated methods.
Q: Can this method be adapted for large-scale commercial production?
A: Yes, the reaction utilizes commercially available divalent palladium catalysts and silver oxidants under moderate temperatures (90-120°C), making it highly suitable for kilogram-to-ton scale-up.
Q: How is the final 2-hydroxybenzophenone obtained from these derivatives?
A: The 2-pyridyloxy group serves as a removable directing group; it can be efficiently hydrolyzed under mild acidic or basic conditions to yield the valuable 2-hydroxydiaryl ketone scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridyloxydiaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory routes like the one described in CN103242223A can be successfully translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch. Whether you require custom synthesis of specific derivatives or bulk supply of standard intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to engage with our technical procurement team to discuss how this innovative coupling strategy can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity 2-pyridyloxydiaryl ketones and their hydrolyzed 2-hydroxybenzophenone counterparts reliably. Partner with us to leverage cutting-edge chemistry for your next generation of therapeutic agents.
