Advanced Pd-Catalyzed Synthesis of 2-Pyridyloxydiaryl Ketones for Commercial Scale-up
Introduction to Novel Synthetic Methodology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable routes for constructing complex biaryl ketone scaffolds, which serve as critical building blocks for active pharmaceutical ingredients (APIs) and agrochemicals. Patent CN103242223A introduces a highly efficient preparation method for 2-pyridyloxydiaryl ketone derivatives, utilizing a palladium-catalyzed decarboxylative coupling strategy. This technology represents a significant advancement over traditional Friedel-Crafts acylations or cross-coupling reactions that often require pre-functionalized halides and generate stoichiometric amounts of toxic waste. By leveraging arylformylformic acids as coupling partners, this process achieves direct C-H functionalization ortho to the pyridyloxy group, streamlining the synthetic pathway.
The core innovation lies in the use of a divalent palladium catalyst system combined with a monovalent silver oxidant, facilitating a redox-neutral cycle that minimizes environmental impact. The reaction operates under relatively mild thermal conditions, typically between 90°C and 120°C, ensuring compatibility with a wide range of functional groups including halogens, alkyls, and trifluoromethyl substituents. For procurement managers and supply chain directors, this methodology offers a compelling value proposition: it utilizes commercially available starting materials and generates benign by-products (CO2 and H2O), thereby simplifying waste management and reducing the overall cost of goods sold (COGS) in large-scale manufacturing scenarios.
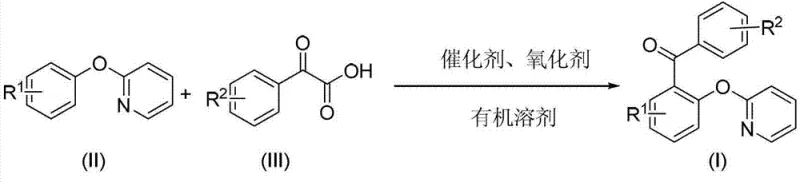
Furthermore, the versatility of this synthetic route allows for the rapid generation of diverse chemical libraries, enabling R&D teams to explore structure-activity relationships (SAR) efficiently. The resulting 2-pyridyloxydiaryl ketones are not only valuable intermediates in their own right but also serve as protected precursors to 2-hydroxydiaryl ketones, a class of compounds with known biological activities including anti-colon cancer and HIV-1 inhibition properties. As a reliable pharmaceutical intermediate supplier, understanding and implementing such atom-economical processes is essential for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of diaryl ketones often relies on Friedel-Crafts acylation, which suffers from poor regioselectivity, especially when dealing with deactivated aromatic rings or sensitive heterocycles. These classical methods frequently require harsh Lewis acids like aluminum chloride, generating substantial amounts of corrosive waste that necessitates expensive disposal protocols. Additionally, achieving ortho-substitution specifically adjacent to an ether linkage often requires multi-step protection and deprotection sequences, drastically increasing the step count and reducing overall yield. In the context of industrial scale-up, these inefficiencies translate to higher energy consumption, longer lead times, and increased safety risks associated with handling hazardous reagents.
Moreover, conventional cross-coupling strategies typically demand the use of aryl halides and organometallic reagents (such as Grignard or organolithium species), which are moisture-sensitive and pose significant safety challenges in large reactors. The requirement for cryogenic conditions or strict anhydrous environments further complicates the manufacturing process, limiting the ability to achieve cost reduction in API manufacturing. The accumulation of metal salts and organic by-products from these traditional routes creates a heavy burden on downstream purification processes, often requiring extensive chromatography or recrystallization steps that erode profit margins.
The Novel Approach
In stark contrast, the methodology disclosed in CN103242223A employs a direct C-H activation strategy that bypasses the need for pre-halogenated substrates. By utilizing arylformylformic acids, the reaction proceeds through a decarboxylative mechanism that inherently drives the equilibrium forward through the release of carbon dioxide gas. This thermodynamic driving force allows the reaction to proceed to completion with high conversion rates, often exceeding 70-80% yield across various substrates as demonstrated in the patent examples. The use of simple organic solvents like dioxane, acetic acid, or DMSO further enhances the practicality of the process, allowing for easy solvent recovery and recycling.
The operational simplicity of this novel approach cannot be overstated; the reaction mixture can be monitored via thin-layer chromatography (TLC) and typically completes within 6 to 12 hours. Post-reaction workup involves straightforward filtration to remove silver salts followed by standard silica gel chromatography, eliminating the need for complex aqueous quenches or extraction procedures associated with Lewis acid mediated reactions. This streamlined workflow significantly reduces the operational complexity for plant operators and enhances the commercial scale-up of complex pharmaceutical intermediates. The ability to tolerate diverse substituents (R1 and R2 groups) on both aromatic rings ensures that this platform technology can be adapted for the synthesis of a broad spectrum of high-purity organic intermediates.
Mechanistic Insights into Pd-Catalyzed Decarboxylative Coupling
The catalytic cycle proposed in the patent provides a fascinating glimpse into the mechanistic elegance of transition metal-catalyzed C-H functionalization. The process initiates with the coordination of the divalent palladium catalyst to the nitrogen atom of the pyridine ring in the 2-pyridyloxyaryl compound. This coordination event directs the palladium center to the ortho-position of the phenoxy ring, facilitating the activation of the proximal C-H bond to form a cyclopalladated intermediate (Intermediate A). This directed C-H activation is crucial for ensuring regioselectivity, preventing random substitution on the aromatic ring which would lead to difficult-to-separate isomers.
Subsequently, Intermediate A undergoes a transmetallation or ligand exchange with the arylformylformic acid substrate to generate Intermediate B. This step is followed by a critical decarboxylation event, where the loss of CO2 generates a new organopalladium species (Intermediate C). The final step involves reductive elimination, which forms the desired C-C bond to yield the 2-pyridyloxydiaryl ketone product while releasing a zero-valent palladium species. To close the catalytic cycle, the monovalent silver oxidant re-oxidizes the Pd(0) back to the active Pd(II) state, ready to enter another turnover. This redox economy ensures that only catalytic amounts of palladium are required, minimizing the residual metal content in the final product, a key parameter for regulatory compliance in drug substance manufacturing.
From an impurity control perspective, the mild reaction temperatures (optimized around 110-115°C) play a vital role in suppressing side reactions such as homocoupling or over-oxidation. The choice of silver oxidant (e.g., Ag2O, AgOAc) influences the rate of re-oxidation and the solubility of silver salts, which can impact the filtration efficiency during workup. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as the molar ratio of oxidant to catalyst (typically 1:1 to 3:1), to maximize yield and minimize the formation of palladium black or other inactive species. This level of control is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can affect downstream biological activity or material performance.
How to Synthesize 2-Pyridyloxydiaryl Ketone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring. The general procedure involves charging a reaction vessel with the 2-pyridyloxyaryl compound, arylformylformic acid, palladium catalyst, and silver oxidant in a suitable organic solvent. The mixture is then heated under stirring until the starting materials are consumed. Detailed standardized operating procedures (SOPs) regarding addition rates, temperature ramping, and safety precautions for handling silver salts are critical for consistent results. For a comprehensive guide on the specific stoichiometry and workup protocols derived from the patent examples, please refer to the technical steps outlined below.
- Combine 2-pyridyloxyaryl compound, arylformylformic acid, Pd catalyst (e.g., Pd(OAc)2), and Ag oxidant (e.g., Ag2O) in an organic solvent like dioxane.
- Heat the reaction mixture to 90-120°C and stir for 6-12 hours until TLC indicates completion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography to obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the significant cost optimization potential driven by the use of readily available, commodity-grade starting materials. Arylformylformic acids and substituted phenols are produced on a multi-ton scale globally, ensuring a stable supply base that is less susceptible to the volatility often seen with specialized organometallic reagents. This stability translates directly into enhanced supply chain reliability, allowing manufacturers to secure long-term contracts and mitigate the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated precursors and the reduction in step count directly lower the variable costs associated with production. Furthermore, the generation of benign by-products (water and carbon dioxide) drastically simplifies waste treatment protocols, reducing the environmental fees and disposal costs that often burden chemical manufacturing budgets. By avoiding the use of stoichiometric amounts of toxic Lewis acids, the facility also saves on corrosion-resistant equipment maintenance and personal protective equipment (PPE) requirements, contributing to substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent purity, which is a common cause of batch failures in more sensitive catalytic systems. This reliability ensures consistent output quality and volume, enabling suppliers to meet tight delivery schedules for downstream clients. The ability to source catalysts and oxidants from multiple vendors further diversifies the supply chain, reducing dependency on single-source suppliers and enhancing overall resilience against market disruptions.
- Scalability and Environmental Compliance: As regulatory pressures regarding green chemistry intensify, this method positions manufacturers favorably by adhering to principles of atom economy and waste prevention. The scalability of the process is supported by the use of common organic solvents and standard heating equipment, meaning that technology transfer from lab to kilo-lab to plant scale can be achieved with minimal engineering modifications. This ease of scale-up accelerates time-to-market for new drug candidates relying on this scaffold, providing a competitive edge in the fast-paced pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and descriptions provided in the patent documentation, aiming to clarify the operational feasibility and scope of the method for potential partners and licensees.
Q: What is the primary environmental benefit of this synthesis method?
A: The reaction produces only carbon dioxide and water as by-products during the decarboxylation step, significantly reducing hazardous waste generation compared to traditional acylation methods.
Q: Which catalysts are compatible with this protocol?
A: The patent specifies divalent palladium catalysts such as palladium acetate, Pd(CH3CN)2Cl2, or PdCl2, paired with monovalent silver oxidants like silver oxide or silver carbonate.
Q: Can this method be used to synthesize 2-hydroxybenzophenones?
A: Yes, the resulting 2-pyridyloxydiaryl ketones serve as excellent precursors that can be hydrolyzed under mild conditions to yield high-purity 2-hydroxybenzophenones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridyloxydiaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the development of next-generation therapeutics and functional materials. Our team of expert process chemists has extensively evaluated the methodology described in CN103242223A and possesses the technical capability to adapt and optimize this Pd-catalyzed coupling for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless and compliant with stringent purity specifications.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis can enhance your supply chain efficiency. Whether you require custom synthesis of specific derivatives or a full feasibility assessment for a new API intermediate, we are prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules, and let us help you accelerate your project timelines with our world-class CDMO capabilities.
