Advanced Chiral Hydroxyl Amide Catalysts: Technical Breakthroughs and Commercial Scalability
Advanced Chiral Hydroxyl Amide Catalysts: Technical Breakthroughs and Commercial Scalability
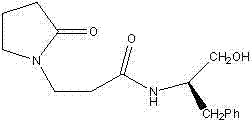
The landscape of asymmetric synthesis is continually evolving, driven by the demand for highly efficient and selective chiral catalysts that can streamline the production of complex pharmaceutical intermediates. A significant advancement in this domain is detailed in patent CN102898342A, which discloses a novel chiral hydroxyl amide compound, specifically N-2-hydroxyethyl-1-(R)-benzyl-1-pyrrole propanamide-2-ketone. This invention represents a pivotal shift in how chiral ligands are constructed, moving away from traditional multi-step protections towards a more direct and robust synthetic pathway. The disclosed methodology not only achieves high stereochemical fidelity but also demonstrates remarkable versatility in catalytic applications, such as the cyanosilylation of aldehydes. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for evaluating its potential integration into existing supply chains for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral hydroxyl amides has relied heavily on the direct condensation of carboxylic acids with chiral amino alcohols or the acid-catalyzed ring-opening of oxazolines. These conventional routes often suffer from significant drawbacks, including the requirement for harsh acidic conditions that can lead to racemization of the chiral center, thereby compromising the optical purity of the final product. Furthermore, the purification of intermediates in these traditional pathways frequently necessitates complex chromatographic separations or multiple recrystallization steps, which drastically reduces overall yield and increases waste generation. The reliance on sensitive protecting groups also adds unnecessary complexity and cost to the manufacturing process, making scale-up economically challenging for industrial applications where cost reduction in fine chemical manufacturing is a primary objective.
The Novel Approach
In contrast, the novel approach outlined in the patent utilizes a unique strategy involving the reaction of 2-ketopyrrolidine benzene propane nitrile with D-phenylalaninol under the catalysis of anhydrous zinc chloride. This method operates under anhydrous and oxygen-free conditions in a chlorobenzene solvent, facilitating a highly selective transformation that bypasses many of the pitfalls associated with traditional acid catalysis. The subsequent step involves an unexpected yet beneficial ring-opening reaction mediated by cupric acetate, which converts the oxazoline intermediate directly into the target chiral hydroxyl amide. This streamlined process eliminates the need for aggressive acidic reagents and simplifies the downstream purification workflow, offering a clear pathway for the commercial scale-up of complex chiral catalysts with improved economic viability.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization and Lewis Acid Activation
The core of this technological breakthrough lies in the precise mechanistic control exerted by the anhydrous zinc chloride catalyst during the initial cyclization phase. Zinc chloride acts as a potent Lewis acid, coordinating with the nitrile group of the precursor to activate it towards nucleophilic attack by the amino alcohol. This coordination lowers the activation energy barrier for the formation of the oxazoline ring, ensuring that the reaction proceeds efficiently at reflux temperatures without degrading the sensitive chiral information encoded in the D-phenylalaninol substrate. The strict exclusion of moisture is critical here, as water would compete for coordination sites on the zinc center, potentially leading to hydrolysis of the nitrile or the formation of unwanted byproducts, thus underscoring the importance of rigorous process control in maintaining high selectivity.
Following the formation of the oxazoline intermediate, the mechanism shifts to a copper-mediated transformation that is both surprising and highly effective. The interaction between the oxazoline nitrogen and the copper(II) center in cupric acetate facilitates the cleavage of the C-O bond within the oxazoline ring, effectively unlocking the structure to reveal the free hydroxyl and amide functionalities characteristic of the final product. This ring-opening event is not merely a structural modification but serves to generate a potent Lewis acid catalyst capable of activating carbonyl compounds for nucleophilic addition. As demonstrated in the patent data, this resulting chiral complex exhibits superior catalytic performance, driving the cyanosilylation of 2-tolualdehyde to near-quantitative conversion, which validates the efficacy of this specific structural motif in asymmetric induction.
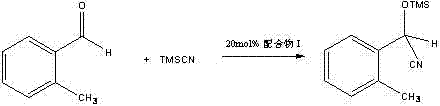
Furthermore, the versatility of this catalytic system extends beyond simple cyanosilylation, as evidenced by its application in Henry reactions (nitroaldol reactions). The chiral environment provided by the hydroxyl amide backbone allows for the differentiation of enantiotopic faces of the aldehyde substrate, guiding the approach of the nitromethane nucleophile with high precision. This dual functionality suggests that the catalyst operates through a well-defined transition state where both the Lewis acidic metal center and the hydrogen-bonding capability of the hydroxyl group work in concert to stabilize the developing charge during the bond-forming event. Such mechanistic robustness is a key indicator of a reliable technology platform that can be adapted for the synthesis of a wide array of chiral building blocks required in the pharmaceutical industry.
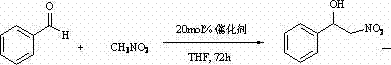
How to Synthesize N-2-hydroxyethyl-1-(R)-benzyl-1-pyrrole propanamide-2-ketone Efficiently
The synthesis of this high-performance chiral catalyst is designed to be operationally straightforward while maintaining the rigorous standards required for fine chemical production. The process begins with the preparation of the oxazoline intermediate, which serves as the crucial scaffold for the final active species. By adhering to the specific stoichiometric ratios and solvent systems defined in the patent, manufacturers can ensure consistent batch-to-batch reproducibility. The detailed standardized synthesis steps provided below outline the precise conditions necessary to achieve the reported yields and purity levels, serving as a foundational guide for process chemists looking to implement this technology.
- Reflux 2-ketopyrrolidine benzene propane nitrile with D-phenylalaninol in chlorobenzene using anhydrous ZnCl2 catalyst for 24 hours under inert atmosphere.
- Purify the intermediate 1-[2-[(4R)-4,5-dihydro-4-benzyl-2-oxazolinyl]ethyl]-2-ketopyrrolidine via water dissolution, dichloromethane extraction, and column chromatography.
- React the purified intermediate with cupric acetate monohydrate in ethanol mixed solvent for 48 hours, followed by recrystallization using ethanol and normal hexane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers substantial benefits regarding cost structure and supply chain resilience. The elimination of expensive transition metal catalysts typically used in asymmetric synthesis, replacing them with more abundant and cost-effective zinc and copper salts, directly contributes to significant cost savings in raw material expenditure. Moreover, the use of common solvents like chlorobenzene and ethanol simplifies the solvent recovery and recycling processes, further reducing the operational overhead associated with waste management and environmental compliance. These factors combine to create a manufacturing profile that is not only economically attractive but also aligned with modern green chemistry principles, making it a compelling choice for long-term supply agreements.
- Cost Reduction in Manufacturing: The process leverages inexpensive and readily available starting materials such as D-phenylalaninol and simple nitrile derivatives, avoiding the need for proprietary or exotic reagents that often drive up costs in chiral synthesis. By utilizing a catalytic amount of zinc chloride rather than stoichiometric quantities of expensive chiral auxiliaries, the overall material cost per kilogram of the final product is drastically reduced. Additionally, the high conversion rates observed in the downstream catalytic applications mean that less catalyst is required to achieve the desired transformation, amplifying the economic efficiency of the entire value chain.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized precursors. Since the reaction conditions utilize standard industrial equipment capable of handling reflux and inert atmospheres, there are no significant barriers to scaling production from laboratory to pilot plant and eventually to commercial tonnage. This scalability guarantees a steady and continuous supply of the chiral catalyst, allowing downstream users to plan their production schedules with greater confidence and reduced risk of stockouts.
- Scalability and Environmental Compliance: The synthetic pathway generates minimal hazardous waste compared to traditional methods that might require heavy metal scavengers or aggressive acidic workups. The ability to purify the final product through simple recrystallization techniques rather than extensive column chromatography makes the process inherently more scalable and environmentally friendly. This alignment with stringent environmental regulations reduces the liability and permitting hurdles often associated with chemical manufacturing, facilitating smoother operations and faster time-to-market for new products derived from this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this chiral hydroxyl amide technology. These answers are derived directly from the experimental data and specifications provided in the underlying patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for assessing the feasibility of integrating this catalyst into your specific synthetic workflows.
Q: What is the catalytic efficiency of this chiral hydroxyl amide compound?
A: According to patent CN102898342A, the compound demonstrates exceptional catalytic performance in the nitrile siliconization reaction of 2-tolualdehydes, achieving a conversion rate of up to 99 percent.
Q: What are the critical reaction conditions for synthesizing the intermediate?
A: The synthesis requires strictly anhydrous and oxygen-free conditions. The reaction involves refluxing the precursors in chlorobenzene solvent for 24 hours in the presence of anhydrous ZnCl2 as a catalyst.
Q: How is the final chiral compound purified to ensure high quality?
A: The final product is obtained by reacting the intermediate with cupric acetate dihydrate in an ethanol mixed solvent for 48 hours, followed by recrystallization using a mixture of ethanol and normal hexane to obtain complex single crystals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Hydroxyl Amide Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral catalysts play in accelerating drug discovery and optimizing fine chemical production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our dedication to quality assurance means that you can rely on us to provide the consistent, high-performance materials necessary for your most demanding synthetic challenges.
We invite you to explore how our expertise in chiral synthesis can drive value for your organization. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Partner with us to leverage cutting-edge chemical technologies that enhance your competitive edge in the global marketplace.
