Advanced Manufacturing of Remimazolam Key Intermediates via Boron-Catalyzed Condensation
Advanced Manufacturing of Remimazolam Key Intermediates via Boron-Catalyzed Condensation
The pharmaceutical landscape for ultrashort-acting anesthetics has been significantly reshaped by the introduction of remimazolam, a benzodiazepine derivative that offers rapid onset and offset due to metabolism by tissue esterases. However, the commercial viability of this critical medication relies heavily on the efficient and high-purity synthesis of its key intermediates, specifically the chiral benzodiazepine core. Patent CN115626913A discloses a groundbreaking preparation method for (3S)-7-bromo-2,3-dihydro-2-oxo-5-(2-pyridyl)-1H-1,4-benzodiazepine-3-methyl propionate, addressing long-standing challenges in yield and stereochemical integrity. This technical insight report analyzes the novel trityl-protection strategy coupled with boron-catalyzed condensation, providing a robust pathway for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios. By shifting away from traditional coupling agents and protecting groups, this methodology ensures enhanced supply chain reliability and cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of remimazolam intermediates has been plagued by inefficient protection strategies and difficult purification profiles. Early approaches, such as those disclosed in WO0069836A1 and WO2013029431A1, utilized Fmoc protecting groups which required harsh conditions for removal and resulted in a disappointing total yield of only 48.2%. As illustrated in the reaction scheme below, the reliance on coupling agents often introduced significant impurities that were difficult to separate, compromising the overall quality of the final active pharmaceutical ingredient.
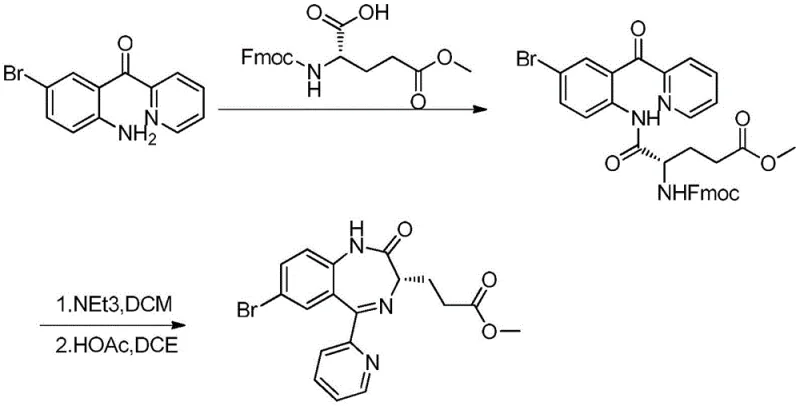
Furthermore, alternative routes described in WO2011032692A1 employed Boc protection strategies which, while an improvement, still suffered from critical drawbacks. In these processes, the intermediate compounds often existed as viscous oils rather than crystalline solids, making isolation and purification exceptionally challenging. The accumulation of impurities through multiple steps led to a final chemical purity of only 93.91% for the remimazolam precursor, necessitating extensive and yield-loss-inducing recrystallization steps to meet regulatory standards. The use of DCC as a coupling agent also generated dicyclohexylurea byproducts, creating substantial waste disposal issues and increasing the cost of goods sold.
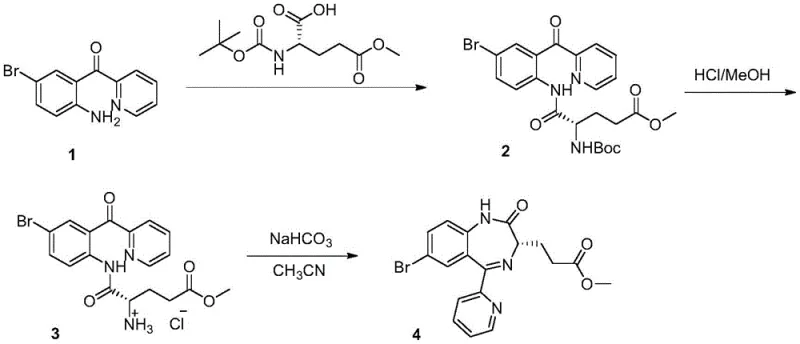
The Novel Approach
The methodology presented in CN115626913A represents a paradigm shift by introducing a trityl (Tr) protecting group combined with a boron-catalyzed condensation mechanism. This approach fundamentally alters the reaction landscape by ensuring that intermediates remain stable and easily separable throughout the synthesis. Unlike the Fmoc or Boc groups, the trityl group provides robust protection during the initial condensation phase but can be cleanly removed under acidic conditions without affecting the sensitive chiral center. This strategic choice allows for the direct formation of solid intermediates, facilitating simple filtration and washing procedures that drastically reduce solvent consumption and processing time.
Mechanistic Insights into Boron-Catalyzed Condensation and Cyclization
The core innovation of this process lies in the replacement of traditional carbodiimide coupling agents with boron-containing reagents such as tris(trifluoroethanol)borate or B(C6F5)3. These Lewis acid catalysts facilitate the dehydration condensation between 2-(2-amino-5-bromo-benzoyl)pyridine and N-Tr-glutamic acid-5-methyl ester with high efficiency. The mechanism avoids the formation of insoluble urea byproducts, thereby simplifying the workup procedure. Moreover, the boron catalyst maintains the enantiomeric integrity of the glutamic acid derivative during the reflux dehydration process, a critical factor where traditional thermal methods often induce racemization. This ensures that the optical purity is preserved from the very first step of the synthesis.
Following condensation and deprotection, the cyclization step is meticulously controlled to prevent epimerization at the chiral alpha-carbon. The patent specifies the use of organic bases such as morpholine or N-methylmorpholine at low temperatures ranging from -10°C to 0°C. This kinetic control is vital; experiments indicate that using common inorganic bases like sodium bicarbonate or raising the temperature leads to a gradual decrease in enantioselectivity. By maintaining these strict low-temperature alkaline conditions, the reaction achieves an enantiomeric excess (ee) of greater than 99%, significantly outperforming previous methods that struggled to exceed 96-97% ee without massive yield penalties.
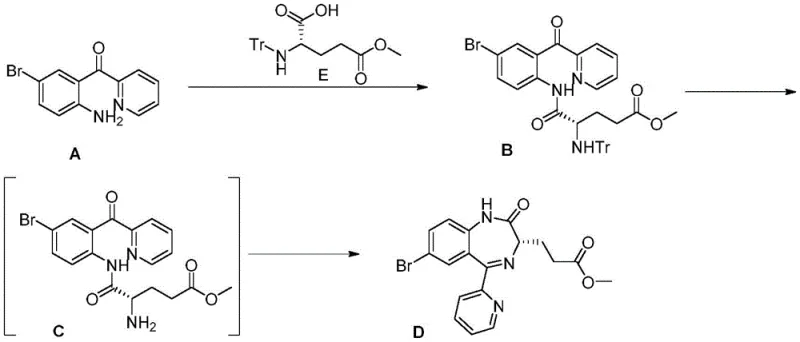
How to Synthesize Remimazolam Intermediate Efficiently
The synthesis protocol outlined in the patent offers a streamlined three-step sequence that is highly amenable to standard reactor setups found in modern fine chemical facilities. The process begins with the condensation of the pyridine ketone and the protected amino acid, followed by a one-pot deprotection and cyclization sequence. This telescoping of steps minimizes material handling and exposure to potential contaminants. For detailed operational parameters, including specific solvent ratios, catalyst loading, and temperature ramps, please refer to the standardized synthesis guide below which encapsulates the critical process parameters identified in the patent examples.
- Condense 2-(2-amino-5-bromo-benzoyl)pyridine with N-Tr-glutamic acid-5-methyl ester using a boron-containing reagent catalyst in an organic solvent.
- Perform acidic deprotection of the trityl group to generate the free amine intermediate without isolating the crude salt if possible.
- Execute ring closure under alkaline low-temperature conditions (-10°C to 0°C) using organic bases like morpholine to prevent racemization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits beyond mere chemical elegance. The elimination of difficult-to-remove byproducts and the generation of crystalline intermediates directly translate to reduced manufacturing cycles and lower operational expenditures. By adopting this technology, organizations can secure a more reliable pharmaceutical intermediate supplier partnership that guarantees consistent quality without the volatility associated with complex purification workflows.
- Cost Reduction in Manufacturing: The substitution of expensive and waste-generating coupling agents like DCC with catalytic amounts of boron reagents significantly lowers raw material costs. Additionally, the ability to isolate intermediates as filterable solids rather than viscous oils reduces the requirement for extensive chromatographic purification or repeated recrystallizations. This simplification of the downstream processing workflow leads to substantial cost savings in solvent usage, labor hours, and waste treatment, effectively optimizing the cost structure for high-purity pharmaceutical intermediate production.
- Enhanced Supply Chain Reliability: The robustness of the trityl protection strategy ensures high process reproducibility, minimizing the risk of batch failures that can disrupt supply continuity. The starting materials, including 2-(2-amino-5-bromo-benzoyl)pyridine and N-Tr-glutamic acid derivatives, are commercially available and stable, reducing the lead time for high-purity pharmaceutical intermediates. Furthermore, the mild reaction conditions and use of common organic solvents mitigate safety risks, ensuring uninterrupted production schedules even in stringent regulatory environments.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with examples demonstrating successful execution from gram to kilogram scales without loss of efficiency. The avoidance of heavy metal catalysts and the reduction of hazardous waste streams align with green chemistry principles, facilitating easier environmental compliance. The high yields reported in the patent examples indicate that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal material loss, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this method over legacy technologies.
Q: How does the Trityl protection strategy improve chiral purity compared to Fmoc or Boc methods?
A: The Trityl (Tr) group offers superior stability during the initial condensation phase compared to Fmoc, preventing premature cleavage. Furthermore, the subsequent cyclization utilizes specific organic bases at low temperatures (-10°C to 0°C), which kinetically suppresses racemization at the chiral center, achieving ee values exceeding 99% compared to the 93-96% often seen in older protocols.
Q: What are the specific advantages of using boron-containing reagents over traditional coupling agents like DCC?
A: Traditional coupling agents like DCC generate dicyclohexylurea (DCU) byproducts which are notoriously difficult to remove completely, leading to impurity accumulation. The novel method employs boron-containing reagents (e.g., tris(trifluoroethanol)borate or B(C6F5)3) which facilitate dehydration condensation cleanly. This results in easier downstream processing, higher chemical purity (>99%), and eliminates the need for complex filtration steps associated with urea removal.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. Unlike previous methods that yielded viscous oils requiring difficult crystallization, this route produces solid intermediates that are easily separated via filtration. The use of common organic solvents like THF, toluene, and dioxane, combined with robust temperature controls, ensures good process reproducibility and safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remimazolam Intermediate Supplier
As the global demand for remimazolam continues to grow, securing a supply chain partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the boron-catalyzed trityl protection route to deliver superior quality intermediates. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. Supported by rigorous QC labs and state-of-the-art analytical equipment, we guarantee that every batch meets the highest standards for chiral purity and chemical identity.
We invite you to collaborate with us to optimize your supply chain for remimazolam production. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this novel route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you navigate the complexities of modern anesthetic manufacturing with confidence and precision.
