Scalable Green Synthesis of Cinnamic Acid Derivatives via Supported Palladium Catalysis
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies that balance high efficiency with environmental responsibility. A pivotal advancement in this domain is detailed in patent CN101781189A, which discloses a novel method for synthesizing cinnamic acid and its derivatives using a Heck coupling reaction. This technology represents a significant departure from traditional protocols by utilizing pure water as the sole solvent and employing a specialized solid-supported double-layer ionic liquid-palladium chloride catalyst. For R&D directors and procurement managers alike, this innovation addresses critical pain points regarding solvent toxicity, catalyst recovery, and overall process economics. By shifting away from volatile organic compounds and difficult-to-separate homogeneous catalysts, this approach offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The ability to operate under mild conditions without inert gas protection further underscores its potential for reducing lead time and operational overhead in large-scale manufacturing environments.
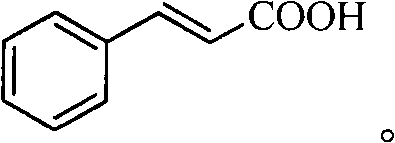
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinnamic acid derivatives via Heck coupling has relied heavily on polar aprotic organic solvents such as N,N-dimethylformamide (DMF), acetonitrile, or N-methylpyrrolidone. While these solvents facilitate the reaction, they pose severe environmental hazards and complicate waste management, directly conflicting with modern green chemistry principles. Furthermore, conventional processes typically utilize homogeneous palladium catalysts, which, despite their high activity, present substantial downstream challenges. Separating these soluble metal complexes from the reaction mixture is notoriously difficult, often requiring expensive purification steps to meet stringent purity specifications required for high-purity pharmaceutical intermediates. The inevitable loss of precious palladium during these separation phases not only inflates raw material costs but also introduces the risk of heavy metal contamination in the final product, necessitating additional quality control measures that delay supply chains.
The Novel Approach
The methodology outlined in the patent data introduces a transformative solution by replacing hazardous organic solvents with pure water, creating a non-polluting reaction medium that simplifies post-treatment procedures. Central to this innovation is the use of a solid-supported double-layer ionic liquid-palladium chloride catalyst, which combines the high activity of palladium with the ease of separation associated with heterogeneous catalysis. This catalyst system allows for the reaction to proceed under atmospheric pressure and without the need for inert gas protection, significantly lowering energy consumption and equipment requirements. The structural integrity of the supported catalyst ensures that it can be recovered through simple filtration and reused multiple times, thereby extending its service life and minimizing metal waste. This approach not only enhances the sustainability profile of the synthesis but also streamlines the workflow for cost reduction in fine chemical manufacturing by eliminating complex solvent recovery and metal scavenging steps.
Mechanistic Insights into Supported Ionic Liquid-Palladium Catalysis
The efficacy of this synthesis relies on the unique architecture of the solid-supported catalyst, which anchors palladium species within a dual-layer ionic liquid matrix on a silica surface. As illustrated in the catalyst structure below, the silica support provides a robust physical framework, while the ionic liquid layers create a microenvironment that stabilizes the active palladium centers. This configuration prevents the aggregation of palladium nanoparticles, a common deactivation pathway in heterogeneous catalysis, ensuring sustained high catalytic activity throughout the reaction cycle. The ionic liquid component also enhances the solubility of organic substrates within the aqueous phase, effectively acting as a phase transfer mediator without the need for additional additives. This synergistic interaction between the support, the ionic liquid, and the metal center facilitates the oxidative addition and reductive elimination steps critical to the Heck coupling mechanism, resulting in superior conversion rates even under mild thermal conditions.
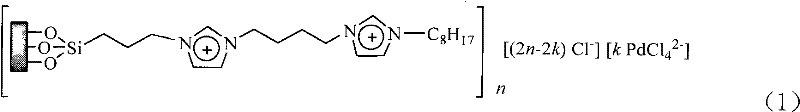
From an impurity control perspective, the heterogeneous nature of the catalyst plays a pivotal role in maintaining product purity. Unlike homogeneous systems where metal leaching is pervasive, the strong coordination between the palladium and the ionic liquid ligands minimizes metal detachment into the bulk solution. This retention capability means that the final filtrate contains negligible amounts of palladium, reducing the burden on downstream purification units. Furthermore, the use of water as a solvent inherently limits the formation of certain organic byproducts that might arise from solvent degradation or participation in side reactions. The reaction conditions, specifically the absence of harsh bases or extreme temperatures, further preserve the integrity of sensitive functional groups on the aromatic ring. For R&D teams focused on impurity profiling, this translates to a cleaner crude product profile, simplifying the crystallization process and ensuring that the final high-purity cinnamic acid derivatives meet rigorous regulatory standards with minimal processing.
How to Synthesize Cinnamic Acid Efficiently
The synthesis protocol described in the patent offers a straightforward and reproducible route for producing cinnamic acid and its various substituted analogues. The process begins with the preparation of the reaction mixture, where sodium carbonate and the specialized catalyst are suspended in pure water, followed by the addition of acrylic acid and the chosen iodoarene substrate. The simplicity of the reagent setup, combined with the absence of inert gas requirements, makes this method highly accessible for both laboratory optimization and industrial implementation. Detailed standard operating procedures regarding specific molar ratios, temperature controls, and workup techniques are essential for maximizing yield and catalyst longevity. The following guide outlines the generalized steps derived from the patent examples to assist technical teams in replicating this efficient synthetic pathway.
- Prepare the reaction mixture by suspending the solid-supported double-layer ionic liquid-palladium chloride catalyst and sodium carbonate in pure water within a reaction vessel.
- Add acrylic acid and the specific iodoarene substrate to the mixture, maintaining a molar ratio of approximately 1: 1.2:2.4:200:0.005 for substrate, acrylic acid, base, water, and catalyst respectively.
- Stir and reflux the mixture for 12 to 36 hours under normal pressure without inert gas protection, then filter to recover the catalyst and acidify the filtrate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this water-based Heck coupling technology presents compelling economic and logistical benefits. The elimination of expensive and toxic organic solvents drastically reduces raw material expenditures and waste disposal fees, contributing to substantial cost savings over the lifecycle of the product. Additionally, the recyclability of the supported catalyst means that the effective cost per kilogram of product decreases with each reuse cycle, optimizing the overall cost of goods sold. The simplified operational requirements, such as running reactions at atmospheric pressure without inert gas, lower the barrier for entry for contract manufacturing organizations and reduce the capital expenditure needed for specialized reactor setups. These factors collectively enhance the reliability of the supply chain by minimizing process variability and dependency on complex utility systems.
- Cost Reduction in Manufacturing: The transition to a water-based solvent system removes the need for costly solvent recovery distillation columns and hazardous waste incineration services. By utilizing a catalyst that can be filtered and reused, the consumption of precious palladium metals is significantly minimized, leading to direct material cost optimization. The mild reaction conditions also translate to lower energy consumption for heating and cooling, further driving down operational expenses. These cumulative efficiencies allow for a more competitive pricing structure for reliable pharmaceutical intermediate suppliers offering these derivatives to the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which do not require strict exclusion of oxygen or moisture, reduces the risk of batch failures due to environmental fluctuations. The availability of water as a solvent ensures that supply chain disruptions related to specialty solvent shortages are mitigated. Furthermore, the simplified workup procedure, involving basic filtration and acidification, shortens the production cycle time, enabling faster turnaround for customer orders. This operational stability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery expectations of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard stainless steel reactors without the need for exotic lining materials required for corrosive organic solvents. The aqueous nature of the waste stream simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The ability to recycle the catalyst reduces the generation of heavy metal-containing solid waste, aligning with corporate sustainability goals. This eco-friendly profile enhances the marketability of the final product to environmentally conscious partners in the fine chemical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on substrate compatibility, catalyst performance, and process parameters. Understanding these details is vital for technical teams evaluating the feasibility of integrating this green chemistry approach into their existing manufacturing portfolios. The insights provided here aim to bridge the gap between academic innovation and practical industrial application.
Q: What are the primary advantages of using water as a solvent in this Heck coupling reaction?
A: Using pure water eliminates the need for toxic polar aprotic solvents like DMF or acetonitrile, significantly reducing environmental pollution and waste treatment costs while simplifying the post-reaction separation process.
Q: How does the supported ionic liquid catalyst improve upon traditional homogeneous palladium catalysts?
A: The solid-supported double-layer ionic liquid-palladium chloride catalyst allows for easy separation via filtration and can be recycled multiple times, preventing palladium loss and reducing contamination of the final pharmaceutical intermediate.
Q: Does this process require specialized equipment like inert gas protection?
A: No, the patented method operates effectively under normal atmospheric pressure without the need for inert gas protection, which drastically lowers operational complexity and equipment costs for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinnamic Acid Supplier
The technological advancements described in patent CN101781189A highlight the immense potential for greener, more efficient synthesis of valuable chemical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methods into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this water-based Heck coupling can be realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cinnamic acid derivative meets the exacting standards required by the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific supply chain needs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this sustainable manufacturing route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials against your current benchmarks. Together, we can drive forward the adoption of green chemistry solutions that deliver both commercial value and environmental stewardship.
