Advanced Rhodium-Catalyzed Synthesis of Chiral 3,3-Disubstituted Isoindolinones for Pharmaceutical Applications
Introduction to Breakthrough Rhodium Catalysis Technology
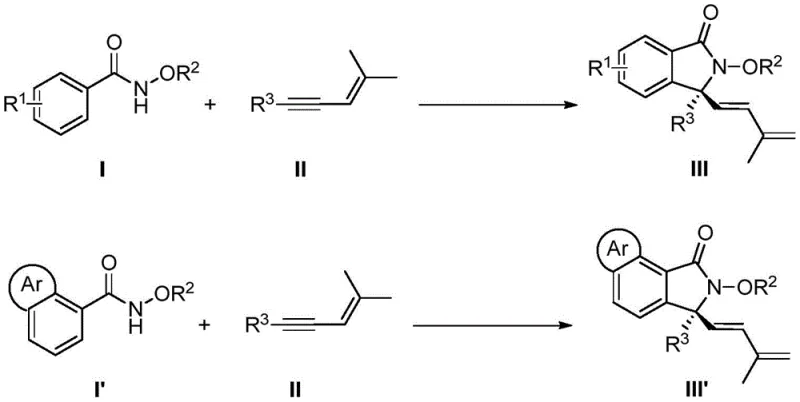
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive molecules. Patent CN113735756A introduces a transformative synthetic methodology for generating chiral 3,3-disubstituted isoindolinone skeletons, a structural motif prevalent in numerous therapeutic agents. This innovation leverages a sophisticated chiral cyclopentadienyl rhodium catalyst to orchestrate a tandem sequence involving C-H bond activation, enyne migration insertion, 1,4-rhodium migration, and nucleophilic cyclization. Unlike traditional approaches that often suffer from harsh conditions and poor stereocontrol, this protocol achieves high yields and exceptional enantioselectivity under remarkably mild temperatures ranging from 5°C to 15°C. The ability to utilize readily available N-methoxybenzamides and 1,3-enynes as starting materials represents a significant leap forward in step economy and operational simplicity for process chemists.
For procurement specialists and supply chain managers, the implications of this technology are profound. The reliance on stable, commercially accessible raw materials mitigates supply chain risks associated with exotic or unstable reagents. Furthermore, the robustness of the catalytic system across a wide array of substrates suggests a versatile platform technology capable of producing diverse libraries of intermediates without necessitating extensive process re-optimization for each new derivative. As a reliable pharmaceutical intermediate supplier, understanding such foundational patents allows us to offer clients superior route scouting and manufacturing capabilities that align with modern green chemistry principles and cost-efficiency targets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3,3-disubstituted chiral isoindolinone cores has been a formidable challenge in organic synthesis, often relying on strategies that are inherently inefficient and resource-intensive. Conventional literature methods typically involve the 3-enantioselective functionalization of pre-existing isoindolinone rings, a strategy that inevitably introduces significant bottlenecks. These traditional routes frequently require multi-step sequences to install the necessary functionality, leading to poor atom economy and reduced overall throughput. Moreover, the starting materials for these older methods are often difficult to synthesize, requiring specialized precursors that drive up raw material costs and extend lead times. The cumulative effect of these inefficiencies is a manufacturing process that is not only expensive but also environmentally burdensome due to the generation of excessive waste streams from protecting group manipulations and purification steps.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct C-H functionalization strategy that bypasses the need for pre-activated substrates entirely. By employing a chiral rhodium catalyst, the method enables a direct [4+1] cyclization between simple N-methoxybenzamides and 1,3-enynes. This paradigm shift eliminates several synthetic steps, drastically simplifying the process flow and enhancing the overall yield of the final product. The reaction proceeds with high regio- and stereoselectivity, constructing the quaternary carbon center with precision that is difficult to achieve via classical nucleophilic substitutions. This streamlined approach not only accelerates the timeline from discovery to production but also aligns perfectly with the industry's demand for cost reduction in pharmaceutical intermediate manufacturing by minimizing reagent consumption and waste disposal costs.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
At the heart of this transformation lies the unique capability of the chiral cyclopentadienyl rhodium catalyst to activate inert C-H bonds with high fidelity. The catalytic cycle initiates with the coordination of the rhodium species to the directing group of the N-methoxybenzamide, facilitating the cleavage of the ortho C-H bond to form a rhodacycle intermediate. This step is critical as it sets the stage for the subsequent insertion of the 1,3-enyne moiety. The chiral environment provided by the pentamethylcyclopentadienyl ligand, specifically modified with chiral appendages as seen in the catalyst structures, exerts precise steric control over the orientation of the incoming enyne. This ensures that the migration insertion occurs with the desired facial selectivity, which is paramount for establishing the correct absolute configuration at the newly formed stereocenter.
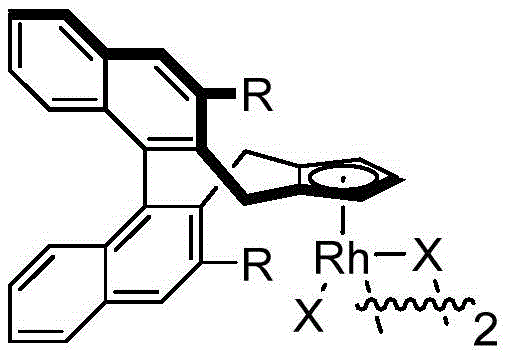
Following the insertion event, the mechanism proceeds through a rare 1,4-rhodium migration, a key feature that distinguishes this chemistry from standard alkyne insertions. This migration allows the metal center to traverse the carbon framework, positioning itself for the final nucleophilic cyclization that closes the isoindolinone ring. The use of silver difluoride as an oxidant is crucial for regenerating the active Rh(III) species from the reduced Rh(I) state, thereby sustaining the catalytic turnover. Understanding these mechanistic nuances is vital for R&D directors aiming to optimize reaction parameters or adapt the chemistry to novel substrates. The high enantiomeric excess values reported, reaching up to 96% ee in certain examples, underscore the effectiveness of the ligand design in transmitting chiral information throughout the catalytic cycle, ensuring the production of high-purity chiral intermediates essential for drug safety.
How to Synthesize Chiral 3,3-Disubstituted Isoindolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction conditions to maximize the benefits of the catalytic system. The protocol is designed to be operationally simple, utilizing common alcohol solvents such as 3-pentanol or ethanol, which are easy to handle and recycle. The reaction typically runs for 60 to 80 hours at low temperatures, a condition that, while requiring time, ensures the high selectivity observed in the patent examples. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that have been validated to produce consistent results across a broad substrate scope.
- Prepare the reaction mixture by combining N-methoxybenzamide, 1,3-enyne, chiral cyclopentadienyl rhodium catalyst, silver difluoride oxidant, and carboxylic acid additive in an alcohol solvent.
- Cool the reaction system to a temperature range of 5-15°C and maintain stirring for 60-80 hours to ensure complete conversion and high enantioselectivity.
- Quench the reaction with ethylenediamine, remove solvents, and purify the crude product via silica gel column chromatography to isolate the target chiral isoindolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed methodology offers tangible benefits that directly impact the bottom line and supply chain resilience. The primary advantage lies in the drastic simplification of the synthetic route, which translates to significant cost reduction in manufacturing. By eliminating the need for multiple protection and deprotection steps, as well as the synthesis of complex starting materials, the overall material cost is substantially lowered. Furthermore, the use of a low loading of catalyst (3-5 mol%) combined with the potential for catalyst recovery or the use of earth-abundant metals in future iterations suggests a pathway towards even greater economic efficiency. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a smaller carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot transformation significantly reduces the number of unit operations required. Fewer isolation and purification steps mean less solvent usage, lower labor costs, and reduced equipment occupancy time. Additionally, the high atom economy of the [4+1] cyclization ensures that a larger proportion of the raw material mass ends up in the final product, minimizing waste disposal fees which are a major component of chemical manufacturing expenses.
- Enhanced Supply Chain Reliability: The substrates employed, such as N-methoxybenzamides and simple enynes, are derived from commodity chemicals that are widely available from multiple global suppliers. This diversity in sourcing options mitigates the risk of supply disruptions that can plague processes reliant on bespoke or single-source reagents. The stability of these starting materials also allows for bulk purchasing and long-term storage, enabling better inventory management and buffering against market volatility.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer for scale-up due to the absence of highly reactive or pyrophoric reagents often found in organometallic chemistry. The use of alcohol solvents and moderate temperatures facilitates easier heat management in large reactors. Moreover, the high selectivity reduces the formation of difficult-to-remove impurities, simplifying the downstream processing and ensuring that the final product meets stringent regulatory purity specifications with less environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific rhodium-catalyzed technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this Rh-catalyzed method over traditional synthesis?
A: This method utilizes direct C-H bond activation, eliminating the need for pre-functionalized substrates and reducing step count. It operates under mild conditions (5-15°C) with high enantioselectivity (up to 96% ee) and yield (up to 91%), significantly improving atom economy compared to conventional multi-step routes.
Q: What is the substrate scope for this catalytic system?
A: The protocol demonstrates broad substrate tolerance, accommodating N-methoxybenzamides with electron-donating (alkyl, alkoxy) and electron-withdrawing (halogen, nitro, trifluoromethyl) groups, as well as various 1,3-enyne derivatives including cyclopropyl and alkyl-substituted variants.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process features simple operation, low catalyst loading (3-5 mol%), and stable starting materials. The mild reaction conditions and use of common alcohol solvents facilitate safe scale-up from laboratory to industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,3-Disubstituted Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the one described in CN113735756A for accelerating drug development pipelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into robust manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the most rigorous quality standards, supported by our state-of-the-art analytical infrastructure and stringent purity specifications. Our rigorous QC labs are equipped to verify the enantiomeric excess and chemical purity of every batch, guaranteeing consistency and reliability for your critical projects.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your specific API needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your project volume and timeline. We encourage you to reach out today to obtain specific COA data for similar compounds and discuss route feasibility assessments that could optimize your supply chain and reduce time-to-market for your next-generation therapeutics.
