Advancing Chiral Isoindolinone Production with Novel Rhodium Catalysis for Commercial Scale
Advancing Chiral Isoindolinone Production with Novel Rhodium Catalysis for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those containing quaternary carbon centers which are prevalent in bioactive molecules. Patent CN113735756A introduces a groundbreaking methodology for the synthesis of chiral 3,3-disubstituted isoindolinone compounds, utilizing a specialized chiral cyclopentadienyl rhodium catalyst. This innovation represents a significant leap forward in transition metal-catalyzed C-H bond activation, enabling the direct assembly of these valuable heterocyclic skeletons from readily available N-methoxybenzamides and 1,3-enynes. By achieving high yields and exceptional enantioselectivity under remarkably mild conditions, this technology addresses critical bottlenecks in the manufacturing of high-purity pharmaceutical intermediates, offering a robust solution for reliable agrochemical intermediate supplier networks and drug development pipelines alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 3,3-disubstituted chiral isoindolinone skeletons has been a formidable challenge for organic synthesizers, often relying on strategies that suffer from poor atom and step economy. Conventional routes typically involve the enantioselective functionalization of pre-formed isoindolinone cores, a process that necessitates complex multi-step sequences and the preparation of difficult-to-synthesize starting materials. These legacy methods frequently encounter issues with limited substrate diversity, harsh reaction conditions that degrade sensitive functional groups, and inconsistent stereocontrol, leading to costly purification burdens. Furthermore, the reliance on pre-activated substrates increases the overall environmental footprint and production costs, making it difficult to achieve the cost reduction in pharmaceutical intermediate manufacturing required for competitive commercial scale-up. The inability to efficiently introduce diverse substituents at the quaternary center restricts the chemical space available for drug discovery, slowing down the optimization of lead compounds.
The Novel Approach
In stark contrast, the method disclosed in the patent leverages a tandem sequence involving C-H bond activation, enyne migration insertion, 1,4-rhodium migration, and nucleophilic cyclization to construct the target skeleton in a single operational step. This enantioselective [4+1] cyclization strategy bypasses the need for substrate pre-activation, directly coupling simple N-methoxybenzamides with 1,3-enynes to forge the challenging quaternary stereocenter with precision. The use of a chiral cyclopentadienyl rhodium catalyst allows the reaction to proceed at temperatures as low as 5°C to 15°C, preserving the integrity of sensitive functional groups that would otherwise decompose under thermal stress. This streamlined approach not only drastically simplifies the synthetic route but also expands the scope of accessible derivatives, enabling the rapid generation of diverse compound libraries for biological evaluation. 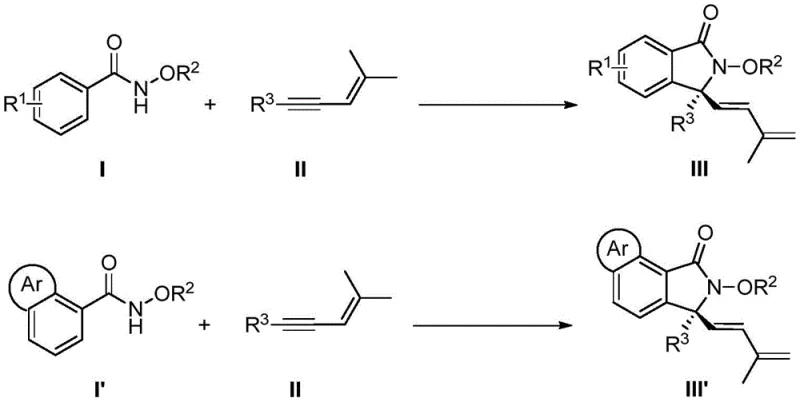
Mechanistic Insights into Rhodium-Catalyzed Enantioselective [4+1] Cyclization
The core of this technological advancement lies in the unique reactivity of the chiral cyclopentadienyl rhodium catalyst, which orchestrates a complex cascade of bond-forming events with high fidelity. The catalytic cycle initiates with the coordination of the rhodium species to the directing group of the N-methoxybenzamide, facilitating a concerted metalation-deprotonation (CMD) process that activates the ortho C-H bond. This key activation step generates a reactive rhodacycle intermediate, which subsequently undergoes migratory insertion with the alkyne moiety of the 1,3-enyne substrate. The resulting vinyl-rhodium species then participates in a rare 1,4-rhodium migration, effectively transposing the metal center to a position poised for intramolecular nucleophilic attack by the amide oxygen. This sequence culminates in the formation of the isoindolinone ring and the regeneration of the active catalyst, all while the chiral ligand environment strictly controls the facial selectivity of the bond formation to ensure high enantiomeric excess.
Understanding the catalyst structure is paramount for optimizing this transformation, as the steric and electronic properties of the cyclopentadienyl ligand directly influence the enantioselectivity and turnover number. The patent highlights specific catalyst variants, such as those featuring methoxy or isopropoxy substituents on the ligand framework, which create a well-defined chiral pocket around the rhodium center. 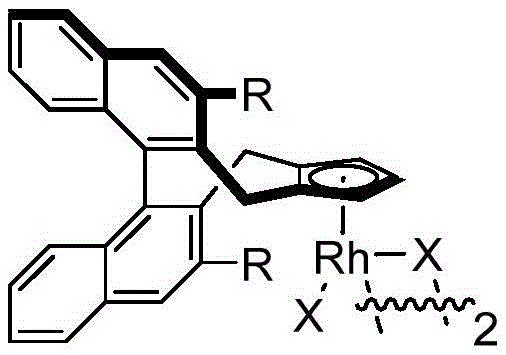 This precise architectural control minimizes non-productive side reactions and suppresses the formation of racemic byproducts, ensuring that the final product meets the stringent purity specifications demanded by regulatory bodies. The compatibility of this system with silver difluoride as a mild oxidant further enhances the reaction profile by facilitating the re-oxidation of the rhodium species without introducing aggressive conditions that could compromise the chiral integrity of the product.
This precise architectural control minimizes non-productive side reactions and suppresses the formation of racemic byproducts, ensuring that the final product meets the stringent purity specifications demanded by regulatory bodies. The compatibility of this system with silver difluoride as a mild oxidant further enhances the reaction profile by facilitating the re-oxidation of the rhodium species without introducing aggressive conditions that could compromise the chiral integrity of the product.
How to Synthesize Chiral 3,3-Disubstituted Isoindolinone Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and optical purity, leveraging the robust protocol established in the patent data. The process involves combining the N-methoxybenzamide substrate with a slight excess of the 1,3-enyne coupling partner in an alcohol solvent, such as 3-pentanol or ethanol, alongside the chiral rhodium catalyst and silver difluoride oxidant. A carboxylic acid additive, preferably acetic acid, is included to facilitate the C-H activation step, and the mixture is stirred at low temperatures for an extended period to ensure complete conversion. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining N-methoxybenzamide, 1,3-enyne compound, chiral cyclopentadienyl rhodium catalyst, silver difluoride oxidant, and carboxylic acid additive in an alcohol solvent.
- Maintain the reaction temperature between 5°C and 15°C and stir continuously for a duration of 60 to 80 hours to ensure complete conversion and high enantioselectivity.
- Quench the reaction with ethylenediamine, remove the solvent, and purify the crude product via silica gel column chromatography to isolate the target chiral isoindolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed methodology offers substantial strategic benefits that extend beyond mere technical novelty. The ability to utilize simple, stable, and commercially available starting materials like N-methoxybenzamides significantly de-risks the supply chain, reducing dependency on exotic or custom-synthesized precursors that often suffer from long lead times and price volatility. The mild reaction conditions translate directly into lower energy consumption and reduced safety hazards associated with high-temperature or high-pressure operations, contributing to a safer and more sustainable manufacturing environment. Furthermore, the high atom economy of the [4+1] cyclization minimizes waste generation, aligning with increasingly strict environmental compliance standards and reducing the costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The elimination of multi-step pre-functionalization sequences drastically reduces the overall material and labor costs associated with producing these complex chiral scaffolds. By consolidating multiple synthetic transformations into a single catalytic pot, manufacturers can achieve significant operational efficiencies and lower the cost of goods sold (COGS) for high-value intermediates. The low catalyst loading requirement of 3-5 mol% further optimizes the economic profile, making the use of precious metal catalysts financially viable even for large-scale production runs without compromising profitability.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction ensures that supply chains remain resilient even when specific building blocks face availability issues, as the chemistry tolerates a wide array of functional groups and structural variations. This flexibility allows for the rapid substitution of alternative starting materials without the need for extensive process re-validation, ensuring continuous production flow and minimizing downtime. The stability of the reagents and the robustness of the catalytic system under ambient storage conditions further simplify logistics and inventory management for global distribution networks.
- Scalability and Environmental Compliance: The protocol is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory benchtop to kiloliter reactors without loss of performance or selectivity. The use of alcohol solvents and mild oxidants simplifies the workup and purification processes, reducing the volume of hazardous organic solvents required and facilitating easier solvent recovery and recycling. This green chemistry approach not only lowers the environmental footprint but also streamlines the regulatory approval process for new drug applications by demonstrating a commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic methodology, providing clarity for stakeholders evaluating its potential integration into their existing workflows. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of this rhodium-catalyzed method over traditional synthesis?
A: This method utilizes direct C-H bond activation and [4+1] cyclization, eliminating the need for multi-step pre-functionalization. It operates under mild conditions (5-15°C) with high atom economy and achieves up to 96% ee, significantly simplifying the production of complex chiral scaffolds.
Q: What is the substrate scope for this catalytic system?
A: The process demonstrates exceptional versatility, tolerating a wide range of substituents on the benzamide ring including halogens, alkyl groups, esters, and nitro groups. It also accommodates various 1,3-enyne derivatives, allowing for the construction of diverse 3,3-disubstituted isoindolinone libraries.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the protocol uses stable, commercially available starting materials and low catalyst loading (3-5 mol%). The mild reaction temperatures and robust tolerance to functional groups make it highly scalable for industrial applications while maintaining stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,3-Disubstituted Isoindolinone Supplier
As the demand for complex chiral intermediates continues to surge in the pharmaceutical sector, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a distinct competitive advantage. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral isoindolinone delivered meets the highest international standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative rhodium-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this technology offers for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and accelerate your drug development timelines.
