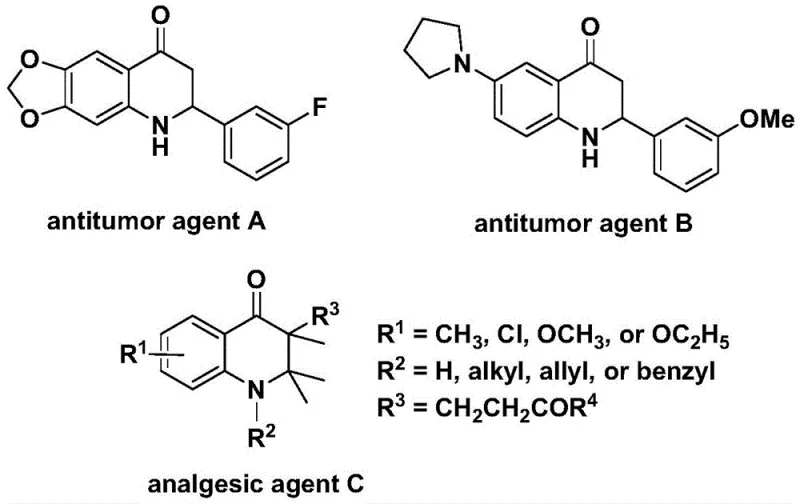
Scalable Synthesis of Substituted 2,3-Dihydroquinolones for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B introduces a significant advancement in the preparation of substituted 2,3-dihydroquinolone compounds, a core scaffold found in numerous therapeutic agents ranging from antitumor drugs to potent analgesics. This technology leverages a transition metal palladium-catalyzed carbonylation strategy that fundamentally shifts the paradigm from hazardous gas-phase reactions to safer, solid-source methodologies. By utilizing N-pyridine sulfonyl-o-iodoaniline and versatile olefin substrates, this process achieves high reaction efficiency and exceptional substrate compatibility. The ability to synthesize complex 2-aryl and 3-alkyl substituted derivatives under relatively mild conditions addresses critical bottlenecks in intermediate manufacturing. For R&D teams and procurement specialists, understanding the mechanistic depth and commercial viability of this patent is essential for securing a reliable supply chain of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the 2,3-dihydroquinolone skeleton often rely on classical cyclization reactions that suffer from significant operational drawbacks. Many conventional methods require harsh reaction conditions, such as extremely high temperatures or the use of corrosive acids, which can degrade sensitive functional groups and limit the scope of applicable substrates. Furthermore, historical approaches frequently involve the direct use of carbon monoxide gas, posing severe safety risks and necessitating specialized high-pressure equipment that increases capital expenditure and operational complexity. The purification processes associated with these older methods are often tedious, involving multiple recrystallization steps that reduce overall yield and generate substantial chemical waste. Additionally, the regioselectivity in traditional Friedel-Crafts or radical cyclization approaches can be difficult to control, leading to complex impurity profiles that require extensive downstream processing to meet stringent pharmaceutical purity standards.
The Novel Approach
The methodology disclosed in CN112239456B represents a transformative shift by employing a palladium-catalyzed carbonylation reaction using a solid carbon monoxide substitute. This innovative approach eliminates the need for handling toxic CO gas, thereby drastically simplifying the reactor setup and enhancing workplace safety for manufacturing personnel. The use of 1,3,5-mesitylene phenol ester as a CO source allows for a controlled release of carbon monoxide in situ, ensuring a steady concentration that drives the reaction forward without the risks associated with gas cylinders. This method operates effectively in common organic solvents like dioxane at moderate temperatures between 100°C and 120°C, making it energy-efficient and compatible with standard glass-lined reactors. The reaction demonstrates remarkable versatility, accommodating a wide range of olefin substrates including aryl, alkyl, and silyl groups, which enables the rapid generation of diverse chemical libraries for drug discovery programs without the need for process re-optimization.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The catalytic cycle begins with the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, forming a reactive aryl-palladium intermediate. This step is facilitated by the bidentate phosphine ligand, 1,3-bis(diphenylphosphino)propane (dppp), which stabilizes the palladium center and prevents the formation of inactive palladium black precipitates. Subsequently, the carbon monoxide released from the solid surrogate inserts into the palladium-carbon bond, generating an acyl-palladium species that is crucial for the subsequent ring-closing step. The coordination and migratory insertion of the olefin into this acyl-palladium intermediate proceed with high stereocontrol, dictated by the steric and electronic properties of the olefin substituent. Finally, a reductive elimination step releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, completing the cycle. This well-defined mechanism ensures high turnover numbers and minimizes the formation of side products, resulting in a cleaner reaction profile compared to non-catalytic thermal methods.
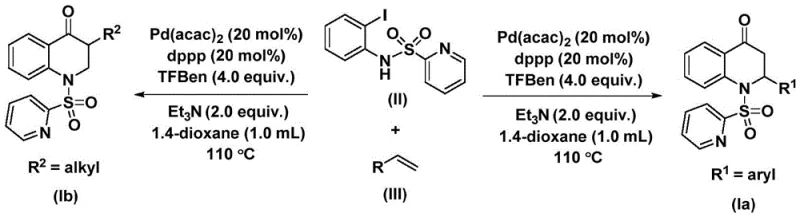
Impurity control in this synthesis is inherently managed through the specificity of the palladium catalytic cycle. Unlike radical-mediated processes that often generate polymeric byproducts or isomeric mixtures, this ionic organometallic pathway follows a discrete sequence of elementary steps that limits off-cycle reactivity. The choice of the pyridine sulfonyl protecting group plays a pivotal role in directing the cyclization and preventing unwanted N-alkylation or polymerization of the olefin. Furthermore, the use of a solid CO source prevents local excesses of carbon monoxide that could lead to double carbonylation or other insertion errors. Post-reaction workup is streamlined, typically involving simple filtration to remove palladium residues followed by standard silica gel chromatography. This predictability in impurity formation allows quality control teams to establish robust specifications and ensures that the final API intermediate meets the rigorous purity requirements demanded by global regulatory agencies.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis route requires precise attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The process involves charging a reactor with the iodoaniline derivative, the selected olefin, the palladium catalyst system, and the solid CO source in an anhydrous aprotic solvent. Maintaining an inert atmosphere is critical to prevent oxidation of the phosphine ligand and deactivation of the catalyst. The reaction mixture is then heated to the optimal temperature range and stirred for a duration sufficient to ensure complete consumption of the starting material, typically monitored by HPLC or TLC. While the general procedure is robust, specific substrate combinations may require minor adjustments to the ligand-to-metal ratio or reaction time to achieve optimal results. For detailed standard operating procedures and specific parameter optimization for your target molecule, please refer to the technical guide below.
- Prepare the reaction mixture by combining Pd(acac)2 catalyst, dppp ligand, solid CO source, and substrates in dioxane solvent.
- Heat the reaction system to 100-120°C and maintain stirring for 24-48 hours to ensure complete conversion.
- Perform post-treatment via filtration and silica gel column chromatography to isolate the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The transition from hazardous gas reagents to stable solid surrogates significantly reduces the regulatory burden and insurance costs associated with chemical manufacturing facilities. By simplifying the reaction setup, companies can utilize existing general-purpose reactors rather than investing in specialized high-pressure infrastructure, leading to immediate capital expenditure savings. The high substrate compatibility means that a single production line can be adapted to manufacture a wide variety of analogues, increasing asset utilization rates and reducing changeover times. Furthermore, the use of commercially available and inexpensive starting materials ensures that the supply chain remains robust against raw material shortages, providing a strategic advantage in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide gas removes the need for expensive safety monitoring systems and specialized containment equipment, drastically lowering operational overheads. The high efficiency of the palladium catalyst system ensures that reagent consumption is minimized, reducing the cost of goods sold per kilogram of product. Additionally, the simplified post-treatment process reduces solvent usage and waste disposal costs, contributing to a more sustainable and economically viable manufacturing model. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as dioxane, triethylamine, and common olefins mitigates the risk of supply disruptions caused by niche reagent shortages. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuations in demand. The stability of the solid CO source also simplifies logistics and storage requirements, removing the complexities associated with transporting and storing compressed gases. This reliability ensures consistent delivery timelines for downstream API manufacturers, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The process is inherently scalable from gram-level laboratory synthesis to multi-ton commercial production without significant re-engineering of the reaction parameters. The reduced generation of hazardous waste and the avoidance of toxic gas emissions align with increasingly stringent environmental regulations and corporate sustainability goals. The high atom economy of the carbonylation reaction ensures that raw materials are efficiently converted into product, minimizing the environmental footprint of the manufacturing process. This compliance readiness facilitates faster regulatory approvals and market entry for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. For further customization or specific technical data sheets, our team is available to provide deeper insights tailored to your project requirements.
Q: What are the key advantages of this carbonylation method over traditional synthesis?
A: This method utilizes a solid carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas equipment, thereby enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What is the substrate compatibility of this Pd-catalyzed protocol?
A: The protocol demonstrates excellent tolerance for various functional groups, including aryl, alkyl, and silyl substituents, allowing for the diverse synthesis of 2-aryl and 3-alkyl substituted derivatives without significant yield loss.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of commercially available starting materials, mild reaction conditions, and straightforward post-treatment procedures makes this method highly adaptable for kilogram-to-ton scale production in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patented methodologies like CN112239456B into commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the highest global standards. Our commitment to quality and consistency makes us a preferred partner for multinational pharmaceutical companies seeking to secure their supply chains for critical oncology and analgesic drug candidates.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
