Advanced Palladium-Catalyzed Carbonylation for Scalable Production of Bioactive 2,3-Dihydroquinolone Scaffolds
Advanced Palladium-Catalyzed Carbonylation for Scalable Production of Bioactive 2,3-Dihydroquinolone Scaffolds
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles, particularly those serving as core scaffolds for potent bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN112239456B, which discloses a novel preparation method for substituted 2,3-dihydroquinolone compounds. These heterocyclic structures are not merely academic curiosities; they form the backbone of numerous therapeutic agents, including established antitumor agents and analgesics that have demonstrated efficacy in clinical settings. The patent outlines a transition metal palladium-catalyzed carbonylation reaction that utilizes N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. This approach represents a paradigm shift from traditional methods by employing a solid carbon monoxide substitute, thereby enhancing operational safety and simplifying the reaction setup for industrial applications.
The biological relevance of the 2,3-dihydroquinolone skeleton cannot be overstated, as evidenced by its presence in diverse pharmacophores. As illustrated in the structural examples provided in the patent documentation, derivatives of this core structure exhibit significant anti-cancer activity against human cell lines and possess potent analgesic properties. 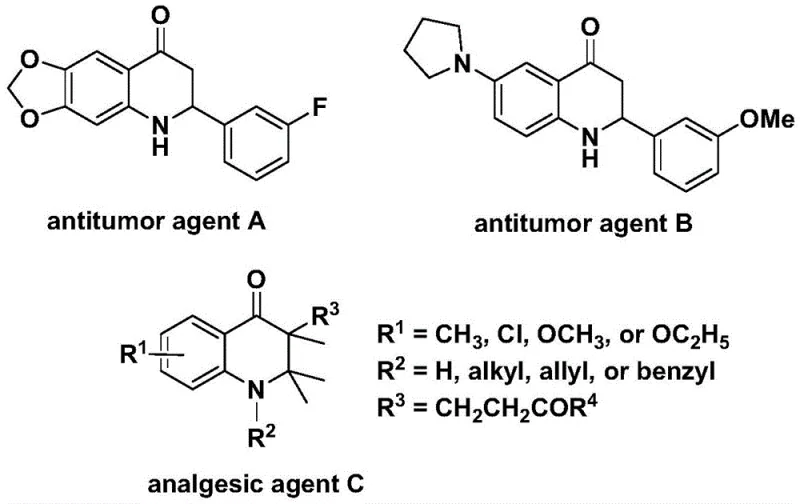 . The ability to efficiently construct this ring system with high regioselectivity and functional group tolerance is therefore of paramount importance to medicinal chemists and process development teams aiming to bring new drug candidates to market. The method described in CN112239456B addresses these needs by offering a versatile pathway that can be adapted to synthesize both 2-aryl and 3-alkyl substituted variants through simple substrate design.
. The ability to efficiently construct this ring system with high regioselectivity and functional group tolerance is therefore of paramount importance to medicinal chemists and process development teams aiming to bring new drug candidates to market. The method described in CN112239456B addresses these needs by offering a versatile pathway that can be adapted to synthesize both 2-aryl and 3-alkyl substituted variants through simple substrate design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has relied heavily on direct carbonylation reactions using carbon monoxide gas. While effective in principle, the use of gaseous CO presents severe logistical and safety challenges, particularly when scaling up to commercial production levels. The requirement for high-pressure reactors and specialized gas handling infrastructure significantly increases capital expenditure and operational complexity. Furthermore, the toxicity of carbon monoxide necessitates rigorous safety protocols and monitoring systems, which can slow down process throughput and increase the risk profile of the manufacturing site. Alternative methods often involve multi-step sequences with poor atom economy or require harsh reaction conditions that are incompatible with sensitive functional groups, leading to lower overall yields and difficult purification processes.
The Novel Approach
The methodology presented in the patent overcomes these hurdles by introducing a homogeneous palladium catalytic system that operates under much milder and safer conditions. Instead of pressurized CO gas, the reaction utilizes 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide surrogate. This substitution eliminates the need for high-pressure equipment and drastically reduces safety risks associated with toxic gas leaks. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at temperatures around 110 °C. 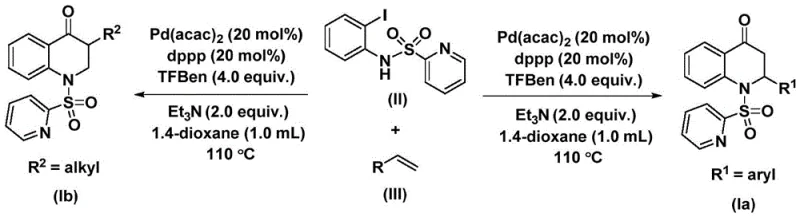 . This general reaction scheme highlights the convergence of the iodoaniline derivative and the olefin substrate in the presence of the palladium catalyst and ligand system. The result is a streamlined, one-pot transformation that delivers the target 2,3-dihydroquinolone cores with high efficiency and excellent substrate compatibility, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.
. This general reaction scheme highlights the convergence of the iodoaniline derivative and the olefin substrate in the presence of the palladium catalyst and ligand system. The result is a streamlined, one-pot transformation that delivers the target 2,3-dihydroquinolone cores with high efficiency and excellent substrate compatibility, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for complex molecule synthesis. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the key entry point for the subsequent carbonylation event. The carbon monoxide, released in situ from the thermal decomposition of the TFBen additive, then inserts into the palladium-carbon bond to form an acyl-palladium species. This insertion step is critical as it constructs the carbonyl functionality directly within the growing molecular framework without requiring external gas feeds.
Following the formation of the acyl-palladium intermediate, the olefin substrate coordinates to the metal center and undergoes migratory insertion. This step establishes the new carbon-carbon bond at the 3-position of the quinolone ring, determining the substitution pattern of the final product. The cycle concludes with an intramolecular reductive elimination or nucleophilic attack that closes the six-membered ring, regenerating the active palladium catalyst and releasing the substituted 2,3-dihydroquinolone product.  . This mechanistic pathway ensures high purity profiles by minimizing side reactions often associated with radical pathways or uncontrolled polymerization of olefins. The use of the dppp ligand further stabilizes the palladium center, preventing catalyst deactivation and ensuring consistent turnover numbers throughout the reaction duration.
. This mechanistic pathway ensures high purity profiles by minimizing side reactions often associated with radical pathways or uncontrolled polymerization of olefins. The use of the dppp ligand further stabilizes the palladium center, preventing catalyst deactivation and ensuring consistent turnover numbers throughout the reaction duration.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and thermal parameters to maximize yield and minimize impurity formation. The patent specifies a molar ratio of catalyst to ligand to CO source that optimizes the reaction kinetics, typically employing 20 mol% of Pd(acac)2 and dppp relative to the substrate. The reaction is conducted in 1,4-dioxane with triethylamine as a base to scavenge acidic byproducts. Detailed standardized operating procedures regarding work-up and purification are essential for maintaining batch-to-batch consistency. For the complete step-by-step experimental protocol, please refer to the structured guide below.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, TFBen, N-pyridine sulfonyl-o-iodoaniline, and the desired olefin substrate in 1,4-dioxane.
- Heat the reaction mixture to 110 °C and maintain stirring for 24 to 48 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers distinct advantages that translate directly into cost reduction in pharmaceutical intermediate manufacturing and enhanced supply security. The shift from gaseous reagents to solid surrogates fundamentally alters the risk profile and infrastructure requirements of the production facility. By eliminating the need for specialized high-pressure carbon monoxide cylinders and the associated regulatory compliance costs, manufacturers can achieve substantial cost savings in both capital investment and ongoing operational expenditures. Furthermore, the use of commercially available starting materials such as olefins and iodoanilines ensures a stable supply chain, reducing the risk of raw material shortages that often plague specialty chemical production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the reaction setup and the high efficiency of the catalytic system. Because the reaction utilizes a solid CO source, there is no need for expensive gas scrubbing systems or high-pressure autoclaves, which significantly lowers the barrier to entry for production. Additionally, the high yields reported (often exceeding 80% for various substrates) mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The ability to use standard glass-lined reactors rather than specialized pressure vessels further contributes to significant cost savings in facility maintenance and operation.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the chemical transformation and the availability of reagents. The tolerance of the catalytic system to various functional groups allows for the use of diverse olefin feedstocks, providing flexibility in sourcing strategies. If a specific olefin becomes unavailable, alternative substrates can often be substituted without requiring a complete re-optimization of the process conditions. This flexibility ensures that production schedules can be maintained even in the face of raw material volatility, securing the delivery of high-purity pharmaceutical intermediates to downstream customers without interruption.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is straightforward due to the homogeneous nature of the reaction and the absence of hazardous gases. The environmental footprint is also reduced, as the solid byproducts from the TFBen decomposition are easier to manage and dispose of compared to gaseous effluents. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The simplified post-treatment process, involving filtration and standard chromatography, facilitates rapid turnaround times, enabling faster response to market demands for complex organic compounds.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent literature, ensuring accuracy and relevance for process development discussions. Understanding these nuances is key to successfully integrating this methodology into existing production workflows.
Q: What is the primary safety advantage of this carbonylation method?
A: Unlike traditional carbonylation which utilizes toxic carbon monoxide gas, this protocol employs 1,3,5-trimesic acid phenol ester (TFBen) as a solid, safe, and easy-to-handle carbon monoxide surrogate, significantly reducing operational hazards.
Q: What types of substrates are compatible with this palladium catalytic system?
A: The method exhibits excellent substrate compatibility, successfully accommodating various aryl olefins with electron-donating or withdrawing groups (such as methyl, chloro, fluoro) as well as alkyl and silyl-substituted olefins.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available catalysts and ligands, operates under relatively mild thermal conditions (110 °C), and avoids high-pressure gas equipment, making it highly amenable to scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results can be seamlessly translated into industrial reality. We are committed to delivering high-purity 2,3-dihydroquinolone intermediates that meet the most stringent purity specifications required by global regulatory bodies. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing consistency and reliability for our partners.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed technology for your next project. Whether you require custom synthesis of novel analogs or large-scale manufacturing of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and cost-effectiveness.
