Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Introduction to Patent CN115353511A: A Breakthrough in Heterocyclic Chemistry
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. Patent CN115353511A introduces a highly efficient, palladium-catalyzed multicomponent reaction strategy for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the valuable indolinone-imidazole hybrid structure. This innovation addresses critical challenges in modern organic synthesis by replacing hazardous gaseous carbon monoxide with a safe, liquid-phase surrogate system while maintaining high atom economy and reaction efficiency. The disclosed method enables the one-pot assembly of three distinct building blocks—trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—under remarkably mild conditions, typically around 30°C. For R&D directors and process chemists, this represents a significant leap forward in accessing diversified chemical space without the logistical burdens associated with high-pressure gas handling. The ability to incorporate trifluoromethyl groups directly into the scaffold further enhances the metabolic stability and lipophilicity of the resulting intermediates, making them prime candidates for next-generation drug development programs.
To fully appreciate the structural complexity achieved through this single synthetic operation, it is essential to examine the specific molecular architectures involved. The starting materials are readily accessible commercial commodities or easily prepared precursors, which lowers the barrier to entry for adoption. The trifluoroethylimidoyl chloride acts as both a nitrogen source and a trifluoromethyl donor, while the propargylamine provides the alkyne functionality necessary for the initial cyclization events. The acrylamide component serves as the electrophilic partner that eventually forms the indolinone core. By integrating these components through a sophisticated catalytic cycle, the process constructs multiple carbon-carbon and carbon-heteroatom bonds simultaneously. This level of convergent synthesis is rare in industrial settings, where step-count reduction is directly correlated with cost savings and yield improvement. The following structural representations illustrate the precise connectivity and functional group distribution that defines this novel class of compounds.


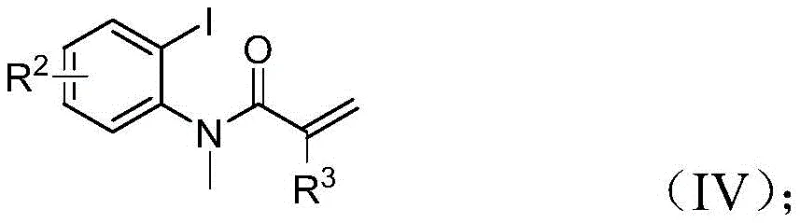
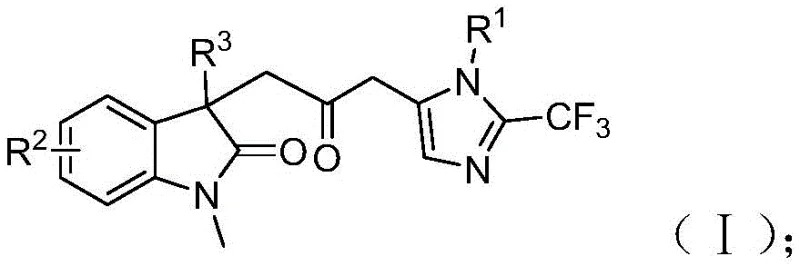
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has relied heavily on traditional carbonylation reactions that necessitate the use of carbon monoxide gas. This approach presents severe safety hazards and engineering challenges, requiring specialized high-pressure reactors and rigorous leak detection systems to protect personnel and the environment. Furthermore, conventional methods often suffer from poor regioselectivity and limited substrate scope, particularly when dealing with sensitive functional groups that might degrade under harsh thermal conditions or in the presence of strong acids and bases. Another significant drawback is the stepwise nature of many legacy syntheses, where the indolinone and imidazole rings are constructed separately and then coupled, leading to lower overall yields and increased waste generation. These inefficiencies translate directly into higher production costs and longer lead times, which are unacceptable in the fast-paced landscape of generic drug manufacturing and custom synthesis. Additionally, the reliance on stoichiometric amounts of toxic reagents or expensive noble metal catalysts without efficient recycling protocols further exacerbates the environmental footprint of these older processes.
The Novel Approach
In stark contrast, the methodology described in CN115353511A utilizes a transition metal-catalyzed cascade reaction that operates under atmospheric pressure and mild thermal conditions. By employing a formic acid and acetic anhydride mixture as an in situ source of carbon monoxide, the process completely eliminates the need for external CO gas cylinders, thereby drastically reducing safety risks and infrastructure costs. This multicomponent strategy allows for the simultaneous formation of the indolinone and imidazole rings in a single pot, significantly improving atom economy and reducing solvent consumption. The use of a palladium catalyst system paired with a trifurylphosphine ligand ensures high turnover numbers and excellent chemoselectivity, even in the presence of diverse substituents on the aromatic rings. This flexibility allows chemists to rapidly iterate on molecular designs, introducing halogens, alkyl groups, or electron-withdrawing moieties without compromising reaction efficiency. The result is a streamlined, scalable process that delivers high-purity products with minimal downstream purification requirements, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
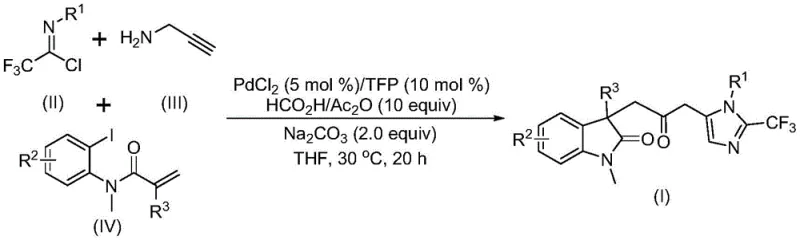
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this transformation hinges on a meticulously orchestrated catalytic cycle initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This key step generates an organopalladium intermediate that subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a five-membered palladacycle. The presence of the formic acid/acetic anhydride system is crucial at this stage, as it decomposes to release carbon monoxide, which then inserts into the palladium-carbon bond to form an acyl-palladium species. This carbonylation event is the defining feature that installs the bridging ketone functionality, linking the two heterocyclic domains. Following this, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted intermolecular condensation to generate a trifluoroacetamidine intermediate, which then undergoes isomerization. The final ring closure is facilitated by the nucleophilic attack of this amidine species onto the activated acyl-palladium complex, releasing the final carbonyl-bridged biheterocyclic product and regenerating the active palladium catalyst. Understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining strict stoichiometric balance and controlling the rate of CO generation to prevent catalyst poisoning or side reactions.
From an impurity control perspective, the mechanistic pathway offers inherent advantages due to its concerted nature. The rapid consumption of reactive intermediates minimizes the accumulation of unstable species that could lead to polymerization or decomposition byproducts. The choice of sodium carbonate as a base ensures a mildly alkaline environment that promotes the desired condensation steps without hydrolyzing the sensitive imidoyl chloride or the final product. Furthermore, the use of tetrahydrofuran (THF) as the solvent provides optimal solubility for all reactants and intermediates, ensuring homogeneous reaction conditions that favor the formation of the target molecule over oligomeric side products. For quality control teams, this means that the crude reaction mixture typically contains fewer structurally related impurities, simplifying the purification workflow. The robustness of the catalytic system also implies that trace amounts of water or oxygen do not catastrophically inhibit the reaction, providing a wider operating window for manufacturing teams. This resilience is a key factor in ensuring batch-to-batch consistency, which is a non-negotiable requirement for supplying regulated pharmaceutical intermediates.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, and base, followed by the sequential addition of the three organic substrates dissolved in dry THF. The CO surrogate mixture is added last to initiate the carbonylation phase once the initial palladium cycle is established. Maintaining the temperature at 30°C is critical; higher temperatures may accelerate catalyst decomposition, while lower temperatures could stall the carbonylation step. The reaction progress should be monitored via TLC or HPLC to determine the optimal endpoint, typically between 12 to 20 hours depending on the specific electronic nature of the substituents. Upon completion, the workup involves simple filtration to remove inorganic salts, followed by concentration and purification via silica gel column chromatography. This straightforward isolation procedure underscores the practicality of the method for both laboratory discovery and pilot plant operations.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride CO surrogate system in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction mixture under inert atmosphere.
- Stir the reaction at mild temperatures (30°C) for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers substantial strategic benefits beyond mere chemical novelty. The elimination of high-pressure carbon monoxide gas removes a major regulatory and safety bottleneck, allowing production to occur in standard glass-lined or stainless steel reactors without the need for specialized autoclaves. This flexibility significantly reduces capital expenditure requirements for new production lines and lowers insurance premiums associated with hazardous gas storage. Moreover, the starting materials—trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides—are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. The ability to source these raw materials competitively drives down the overall cost of goods sold (COGS), enabling more aggressive pricing strategies in the marketplace. Additionally, the high atom economy of the multicomponent reaction means less raw material is wasted as byproduct, further enhancing the economic viability of the process at scale.
- Cost Reduction in Manufacturing: The replacement of toxic gas with liquid reagents drastically simplifies the engineering controls required for production, leading to significant operational cost savings. By avoiding the need for specialized gas handling infrastructure and reducing the number of synthetic steps through a one-pot cascade, the overall processing time and energy consumption are minimized. The use of inexpensive palladium chloride and recyclable ligands ensures that catalyst costs remain a minor fraction of the total production budget. Furthermore, the high yields reported across a broad range of substrates mean that less starting material is needed to produce a given amount of product, directly improving the material cost efficiency. These factors combine to create a highly competitive cost structure that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on widely available, stable solid and liquid reagents mitigates the risk of supply disruptions often associated with compressed gases or exotic reagents. Since the reaction tolerates a wide variety of functional groups, manufacturers can easily switch between different substituted analogs without requalifying the entire process, providing agility in responding to changing customer demands. The robustness of the reaction conditions also reduces the likelihood of batch failures due to minor deviations in temperature or mixing, ensuring consistent on-time delivery performance. This reliability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed supply continuity for their clinical and commercial programs. The scalability demonstrated in the patent up to gram levels suggests a clear path to multi-kilogram production without fundamental process changes.
- Scalability and Environmental Compliance: The process aligns with increasingly stringent environmental regulations by minimizing the generation of hazardous waste and avoiding the release of toxic gases. The use of THF as a solvent allows for efficient recovery and recycling, reducing the volume of organic waste requiring disposal. The mild reaction conditions reduce the energy load on heating and cooling systems, contributing to a lower carbon footprint for the manufacturing site. As regulatory bodies continue to tighten restrictions on volatile organic compounds and hazardous reagents, adopting this greener synthesis route future-proofs the production facility against compliance risks. The simplified workup and purification steps also reduce the consumption of silica gel and eluents, further decreasing the environmental impact of the downstream processing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus of the technical data provided in CN115353511A, ensuring accuracy and relevance for decision-makers.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods requiring high-pressure toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety and eliminating the need for specialized high-pressure equipment.
Q: How does the substrate compatibility impact the versatility of this route for drug discovery?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and electron-withdrawing groups on the aromatic rings, allowing medicinal chemists to rapidly generate diverse libraries of biologically active indolinone-imidazole hybrids.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly validates the scalability of the reaction to the gram level with high efficiency, and the use of commercially available, inexpensive catalysts and reagents supports a straightforward transition to kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multicomponent synthesis for the development of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific safety and engineering requirements of this chemistry, including the safe management of formic acid derivatives and palladium catalysts. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbonyl-bridged biheterocyclic compounds meets the highest international standards for identity, potency, and impurity profiles. Our commitment to quality assurance ensures that the complex molecular architectures generated by this method are delivered with the consistency required for regulatory submissions.
We invite you to leverage our technical expertise to optimize this synthesis for your specific application needs. Whether you require custom analogs for structure-activity relationship (SAR) studies or bulk quantities for clinical trials, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project goals. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain capable of delivering high-purity intermediates with reduced lead times, empowering your R&D efforts and accelerating your time to market.
