Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
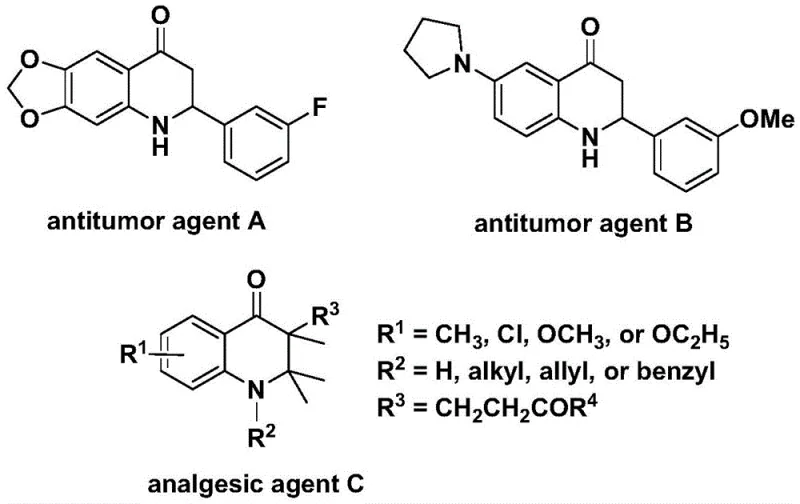
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those serving as privileged scaffolds in drug discovery. Patent CN112239456B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds. These structural motifs are not merely academic curiosities; they are foundational elements in numerous bioactive molecules, including potent antitumor agents and analgesics. As illustrated in the biological context, compounds such as antitumor agent A and agent B exhibit significant activity against human cancer cells, while agent C demonstrates potent analgesic properties. The ability to access these complex cores efficiently is paramount for accelerating drug development pipelines.
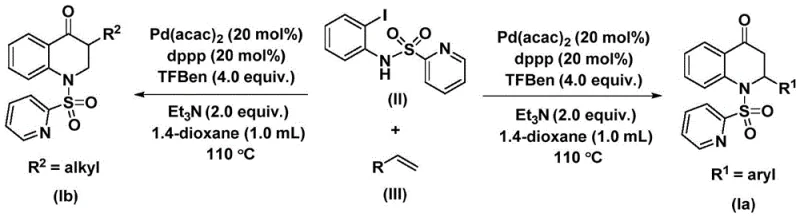
This patented technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. Unlike traditional approaches that often rely on hazardous gaseous carbon monoxide, this method employs a solid carbon monoxide substitute, specifically 1,3,5-trimesic acid phenol ester (TFBen). This strategic substitution fundamentally alters the safety profile and operational feasibility of the synthesis, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to minimize risk while maximizing output. The process is characterized by its simplicity, high reaction efficiency, and exceptional substrate compatibility, enabling the synthesis of both 2-aryl and 3-alkyl substituted derivatives through rational substrate design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has presented significant challenges in organic synthesis. Conventional carbonylation reactions typically require the use of carbon monoxide gas, which poses severe safety hazards due to its toxicity and flammability. Handling CO gas necessitates specialized high-pressure equipment and rigorous safety protocols, which drastically increases capital expenditure and operational complexity for manufacturing facilities. Furthermore, traditional methods often suffer from limited substrate tolerance, where sensitive functional groups on the aromatic ring or the alkene chain may degrade under harsh reaction conditions. This lack of chemoselectivity frequently leads to complex impurity profiles, requiring extensive and costly downstream purification processes that erode overall process efficiency. Additionally, many existing routes struggle to achieve high yields consistently across a broad range of substrates, limiting their utility in the rapid generation of diverse compound libraries for structure-activity relationship (SAR) studies.
The Novel Approach
The methodology described in patent CN112239456B represents a paradigm shift by replacing gaseous CO with a safe, solid surrogate. By utilizing 1,3,5-trimesic acid phenol ester, the reaction releases carbon monoxide in situ under controlled thermal conditions, effectively mitigating the risks associated with high-pressure gas handling. This innovation allows the reaction to proceed in standard laboratory glassware or conventional reactors without the need for autoclaves rated for toxic gases. The use of a palladium catalyst system, specifically bis(acetylacetone)palladium paired with a dppp ligand, ensures high catalytic activity and selectivity. This novel approach not only simplifies the operational procedure but also expands the scope of accessible chemical space. It enables the efficient coupling of diverse olefins, including sterically hindered aryl alkenes and aliphatic chains, with high conversion rates. The result is a streamlined synthetic route that offers substantial cost savings in pharmaceutical intermediate manufacturing by reducing equipment costs and simplifying waste management protocols.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The success of this synthesis relies on a sophisticated catalytic cycle driven by palladium. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the cornerstone of the subsequent transformations. Following this activation, the carbon monoxide molecule, released thermally from the TFBen surrogate, inserts into the palladium-carbon bond. This insertion forms a crucial acyl-palladium intermediate, effectively building the carbonyl functionality directly into the growing molecular framework. The precision of this insertion step is critical for maintaining the integrity of the final quinolone structure and avoiding side reactions such as homocoupling.
Subsequently, the olefin substrate coordinates with the palladium center and undergoes migratory insertion into the acyl-palladium bond. This step creates a new carbon-carbon bond and generates a palladium-alkyl intermediate, setting the stage for ring closure. The final step involves reductive elimination, which releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst to continue the cycle. This elegant mechanism, depicted in the reaction scheme below, highlights the atom economy and efficiency of the process. The presence of the pyridine sulfonyl group on the nitrogen atom plays a dual role: it acts as a directing group to facilitate the initial oxidative addition and serves as a protecting group that can be manipulated in downstream synthetic steps, adding further value to the intermediate for complex API synthesis.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The experimental protocol outlined in the patent provides a clear roadmap for executing this transformation with high reproducibility. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent such as dioxane. The mixture is then heated to temperatures between 100-120 °C, typically around 110 °C, for a duration of 24 to 48 hours. This thermal window is optimized to ensure complete decomposition of the CO surrogate while maintaining catalyst stability. For R&D teams looking to implement this chemistry, the detailed standardized synthesis steps are provided in the guide below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, TFBen (CO surrogate), N-pyridine sulfonyl-o-iodoaniline, and olefin in an organic solvent like dioxane.
- Heat the reaction mixture to 110 °C and stir for 48 hours under inert atmosphere to facilitate the carbonylation cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process addresses several critical pain points in the supply chain for heterocyclic intermediates. The shift away from gaseous carbon monoxide to a solid surrogate fundamentally changes the logistics and safety infrastructure required for production. This transition allows for manufacturing in facilities that may not be equipped for high-pressure toxic gas handling, thereby expanding the pool of potential contract manufacturing organizations (CMOs) and reducing dependency on specialized, high-cost vendors. Furthermore, the use of commercially available starting materials, such as various substituted styrenes and simple alkenes, ensures a stable and resilient supply chain. The robustness of the reaction conditions means that raw material quality fluctuations are less likely to cause batch failures, enhancing overall supply reliability.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas equipment results in significant capital expenditure savings. Additionally, the use of inexpensive and readily available palladium catalysts and ligands, combined with the high atom efficiency of the carbonylation, drives down the cost of goods sold (COGS). The simplified post-treatment process, which involves basic filtration and chromatography, reduces labor and solvent consumption compared to multi-step traditional syntheses. By avoiding complex protection-deprotection sequences often required in other routes, the overall material throughput is improved, leading to substantial cost optimization in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like TFBen and N-pyridine sulfonyl-o-iodoaniline minimizes the risks associated with the storage and transport of hazardous gases. This stability translates to fewer disruptions in production schedules and more predictable lead times for customers. The broad substrate scope allows for the rapid adaptation of the process to synthesize various analogues without retooling the entire production line, providing agility in responding to changing market demands for different drug candidates. This flexibility is a key asset for maintaining continuity in the supply of critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be scaled from gram to kilogram and eventually to tonnage levels without encountering the exponential safety risks associated with pressurized gas reactions. The use of dioxane as a solvent, while requiring careful management, is a well-understood industrial solvent with established recovery and recycling protocols. The high selectivity of the palladium catalyst minimizes the formation of heavy metal waste and organic byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This green chemistry aspect enhances the long-term sustainability of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation. They are intended to clarify the operational parameters and potential applications of this technology for prospective partners and technical stakeholders.
Q: What are the key advantages of using TFBen as a carbon monoxide surrogate in this synthesis?
A: Using 1,3,5-trimethoxyphenyl formate (TFBen) eliminates the need for handling toxic, high-pressure carbon monoxide gas, significantly improving operational safety and simplifying reactor requirements for industrial scale-up.
Q: What is the substrate scope for the olefin component in this palladium-catalyzed reaction?
A: The method demonstrates excellent compatibility with various olefins, including substituted styrenes (aryl), linear and branched alkenes (alkyl), and even silyl-substituted alkenes, allowing for diverse functionalization of the quinolone scaffold.
Q: How does this method compare to traditional carbonylation routes in terms of yield and purity?
A: This protocol achieves high isolated yields ranging from 59% to 88% across diverse substrates, with robust impurity control facilitated by the specific palladium catalytic cycle and straightforward chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
The technological breakthroughs detailed in patent CN112239456B underscore the immense potential of palladium-catalyzed carbonylation in modern medicinal chemistry. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways in delivering high-quality intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. We are committed to translating complex laboratory discoveries into robust, GMP-compliant manufacturing processes that ensure consistent quality and supply.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthetic route for your specific project needs. Whether you require custom synthesis of novel analogues or large-scale production of established intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, cost-effective, and scalable chemical solutions.
