Advancing Benzyl Cyanide Production: A Safe Copper-Catalyzed Route for Commercial Scale-Up
The chemical industry is constantly seeking safer and more economically viable pathways for producing essential nitrile intermediates, which serve as critical building blocks for a vast array of bioactive molecules. Patent CN102381918A introduces a transformative methodology for synthesizing benzyl cyanide compounds by utilizing benzyl chloride precursors in the presence of a copper salt catalyst and potassium ferrocyanide. This innovation addresses the long-standing dichotomy between process safety and economic efficiency, offering a robust alternative to traditional cyanation methods that rely on hazardous reagents. By leveraging the stability of potassium ferrocyanide, this technique mitigates the severe occupational health risks associated with free cyanide ions while maintaining high reaction efficiency. For R&D directors and procurement strategists alike, this patent represents a pivotal shift towards sustainable manufacturing practices that do not compromise on yield or purity standards. The ability to produce high-purity pharmaceutical intermediates without the burden of extreme toxicity protocols opens new avenues for cost-effective supply chain management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylacetonitrile derivatives has relied heavily on nucleophilic substitution reactions using alkali metal cyanides such as sodium cyanide or potassium cyanide, which present formidable challenges in an industrial setting. These reagents are acutely toxic, requiring rigorous safety measures, specialized containment infrastructure, and complex waste treatment protocols to prevent environmental contamination and ensure worker safety. Furthermore, alternative methods employing trimethylsilyl cyanide (TMSCN) introduce different sets of complications, primarily due to the high cost of the reagent and its tendency to release toxic hydrogen cyanide gas upon exposure to moisture. These factors collectively inflate the operational expenditure (OpEx) and capital expenditure (CapEx) for manufacturers, creating a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing. The reliance on such hazardous materials also complicates regulatory compliance and extends the lead time for high-purity intermediates due to the stringent auditing and handling requirements imposed by global safety standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN102381918A utilizes potassium ferrocyanide, a coordination complex that exhibits significantly lower toxicity compared to free cyanide salts, thereby fundamentally altering the safety profile of the production process. This novel approach employs inexpensive copper salts as catalysts to facilitate the transfer of the cyanide group from the ferrocyanide complex to the benzyl chloride substrate in an organic solvent medium. The reaction proceeds smoothly under thermal conditions, typically ranging from 140°C to 190°C, effectively bypassing the need for expensive palladium catalysts or moisture-sensitive silyl reagents. By substituting hazardous inputs with stable, low-cost alternatives, this method achieves a substantial reduction in raw material costs while simultaneously simplifying the engineering controls required for safe operation. The result is a streamlined synthetic route that enhances the reliability of the supply chain for critical agrochemical and pharmaceutical precursors.
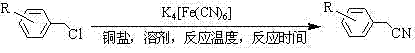
Mechanistic Insights into Copper-Catalyzed Cyanation
The core of this synthetic advancement lies in the copper-catalyzed nucleophilic substitution mechanism, where the copper species acts as a mediator to activate the cyanide source and facilitate its attack on the electrophilic benzylic carbon. The catalytic cycle likely involves the formation of a transient copper-cyanide complex in situ, which possesses enhanced nucleophilicity compared to the free ferrocyanide ion, allowing it to displace the chloride leaving group efficiently. This mechanistic pathway is particularly advantageous because it operates effectively with a variety of copper salts, including cuprous iodide, cuprous chloride, and cuprous bromide, providing flexibility in catalyst selection based on availability and cost. The use of polar aprotic or aromatic solvents such as toluene, DMF, or NMP further stabilizes the transition state, ensuring that the reaction proceeds to completion with minimal side reactions. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize conversion rates and minimize the formation of byproducts like dibenzyl ethers or hydrolysis products.
From an impurity control perspective, the choice of potassium ferrocyanide as the cyanide donor offers a distinct advantage by preventing the generation of free hydrogen cyanide gas, which is a common contaminant in traditional cyanation processes. The stability of the ferrocyanide complex ensures that cyanide is released only in the presence of the copper catalyst and under controlled thermal conditions, thereby reducing the risk of uncontrolled exotherms or toxic gas evolution. This inherent safety feature simplifies the downstream purification process, as there is no need for extensive scrubbing systems to remove volatile cyanide species from the off-gas stream. Consequently, the final product exhibits a cleaner impurity profile, which is critical for meeting the stringent quality specifications required for active pharmaceutical ingredients (APIs) and high-value agrochemical intermediates. The ability to achieve yields of up to 85% with such a clean profile underscores the robustness of this catalytic system for commercial applications.
How to Synthesize Benzyl Cyanide Efficiently
Implementing this synthesis requires careful attention to the stoichiometric ratios and thermal profiles outlined in the patent to ensure optimal performance and reproducibility. The process begins with the preparation of the catalytic mixture, followed by the sequential addition of reagents to maintain control over the reaction kinetics. Detailed standard operating procedures regarding the specific order of addition, stirring rates, and cooling protocols are essential for scaling this chemistry from the laboratory bench to pilot plant reactors. The following guide summarizes the critical operational steps derived from the patent examples to assist technical teams in replicating these results.
- Prepare the reaction vessel by adding the copper salt catalyst (preferably cuprous iodide) and the organic solvent (preferably toluene), followed by brief stirring.
- Introduce the potassium ferrocyanide cyanide source and the benzyl chloride substrate into the mixture, ensuring the molar ratio aligns with the optimized 1: 0.5:0.3 stoichiometry.
- Seal the reaction container and heat the system to approximately 180°C for a duration of 20 hours to facilitate the nucleophilic substitution and obtain the target nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed ferrocyanide route offers compelling economic and logistical benefits that extend far beyond simple raw material substitution. By eliminating the need for highly toxic cyanide salts, manufacturers can significantly reduce the costs associated with safety training, personal protective equipment (PPE), and specialized waste disposal services, leading to a leaner operational budget. The use of abundant and inexpensive copper salts instead of precious metal catalysts like palladium further drives down the direct material costs, making the final product more competitive in the global market. Additionally, the stability of the reagents allows for more flexible inventory management and reduces the risks associated with the storage and transportation of hazardous chemicals, thereby enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with low-cost alternatives such as potassium ferrocyanide and copper iodide directly impacts the bottom line by lowering the bill of materials. Unlike palladium-catalyzed systems which require expensive metal recovery processes to meet residual metal specifications, this copper-based system utilizes base metals that are easier to remove and less costly to replace. Furthermore, the avoidance of moisture-sensitive reagents like TMSCN eliminates the need for strictly anhydrous conditions and specialized drying equipment, reducing energy consumption and infrastructure maintenance costs. These cumulative savings contribute to a more sustainable economic model for the production of benzyl cyanide derivatives.
- Enhanced Supply Chain Reliability: Sourcing potassium ferrocyanide and common copper salts is generally more straightforward and less regulated than procuring controlled substances like sodium cyanide, which are subject to strict international monitoring and trade restrictions. This ease of sourcing ensures a more stable supply of raw materials, reducing the likelihood of production delays caused by regulatory bottlenecks or supplier shortages. Moreover, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling, as the process is less susceptible to minor variations in environmental humidity or temperature compared to sensitive organometallic transformations. This reliability is crucial for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than conventional methods, primarily due to the absence of highly toxic waste streams and the use of recyclable organic solvents like toluene. Scaling this reaction to multi-ton quantities does not require exponential increases in safety infrastructure, as the inherent low toxicity of the reagents mitigates the risk of catastrophic accidents. This facilitates smoother regulatory approvals for new manufacturing sites and expansions, enabling faster time-to-market for new products. The alignment with green chemistry principles also enhances the corporate sustainability profile, which is increasingly becoming a key criterion for supplier selection by major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the practical implications of adopting this technology for industrial production and to assist decision-makers in evaluating its fit within their existing manufacturing portfolios.
Q: Why is potassium ferrocyanide preferred over sodium cyanide in this synthesis?
A: Potassium ferrocyanide is significantly less toxic than traditional cyanide reagents like sodium cyanide or potassium cyanide, drastically reducing safety hazards and the associated costs of specialized containment and waste treatment facilities.
Q: What represents the optimal catalyst for this transformation?
A: While various copper salts such as cuprous chloride and cuprous bromide are effective, cuprous iodide (CuI) has been identified as the preferred catalyst, delivering superior yields up to 85% under optimized conditions.
Q: Can this method be scaled for industrial production of agrochemical intermediates?
A: Yes, the use of inexpensive copper salts and stable solvents like toluene, combined with the elimination of highly toxic gases, makes this process highly suitable for the commercial scale-up of complex agrochemical and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Cyanide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance efficiency, safety, and cost-effectiveness in the production of fine chemical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. We are committed to delivering high-purity benzyl cyanide derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging technologies such as the copper-catalyzed cyanation described in CN102381918A, we can offer our partners a secure and sustainable supply of essential building blocks for their drug discovery and development programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are ready to provide the support you need. Contact us today to explore how our expertise in scalable catalytic processes can drive value and innovation in your organization.
