Advanced Catalytic Synthesis of Chloramphenicol for Industrial Scale-up
The pharmaceutical industry continuously seeks more efficient and sustainable pathways for producing essential antibiotics, and the methodology detailed in patent CN102285896B represents a significant leap forward in the synthesis of the broad-spectrum antibiotic chloramphenicol. This innovative approach fundamentally shifts the synthetic strategy away from the traditional reliance on p-nitroacetophenone, opting instead for a more atom-economical route starting from benzaldehyde and 2-nitroethanol. By leveraging a sophisticated chiral catalytic system, the process achieves high stereoselectivity in the initial bond-forming step, thereby circumventing the notorious yield limitations associated with classical resolution techniques. The integration of catalytic hydrogenation and streamlined acylation steps further refines the workflow, resulting in a robust four-step sequence that delivers the final active pharmaceutical ingredient with purity exceeding 98.0%. For global supply chain leaders, this technology offers a compelling value proposition by reducing the number of unit operations and minimizing the generation of hazardous aluminum-containing waste streams. Ultimately, this patent provides a blueprint for a greener, more cost-effective manufacturing paradigm that aligns perfectly with modern regulatory and economic demands for antibiotic production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chloramphenicol has been plagued by inherent inefficiencies stemming from the use of p-nitroacetophenone as the primary starting material. The conventional synthetic route typically involves a lengthy sequence including bromination, ammonification, acetylation, and aldol condensation, culminating in a Meerwein-Ponndorf-Verley reduction using aluminum isopropoxide. A critical bottleneck in this traditional pathway is the subsequent chiral resolution step, which theoretically caps the maximum yield at merely 50%, effectively discarding half of the synthesized material as the unwanted enantiomer. Furthermore, the use of aluminum isopropoxide generates substantial quantities of difficult-to-treat industrial waste, creating significant environmental compliance burdens and disposal costs for manufacturers. The combination of low theoretical yield, extensive waste generation, and the use of relatively expensive starting materials renders the classical method increasingly uncompetitive in a market driven by cost efficiency and sustainability metrics. These structural deficiencies in the legacy process have long necessitated the development of alternative synthetic strategies that can bypass these thermodynamic and environmental hurdles.
The Novel Approach
In stark contrast to the legacy methods, the novel approach described in the patent utilizes a direct asymmetric Henry reaction between benzaldehyde and 2-nitroethanol to establish the critical chiral centers with high fidelity. This strategy eliminates the need for chiral resolution entirely, as the desired stereochemistry is induced catalytically during the initial carbon-carbon bond formation, potentially doubling the theoretical yield compared to resolution-based routes. The subsequent transformation of the nitro group to an amine via catalytic hydrogenation is a clean and high-yielding process that avoids the use of stoichiometric metal reductants. Following acylation with methyl dichloroacetate, the final nitration step installs the requisite nitro group on the aromatic ring to complete the chloramphenicol structure. This streamlined four-step sequence not only simplifies the operational complexity but also utilizes benzaldehyde, a commodity chemical that is significantly more inexpensive and readily available than p-nitroacetophenone. 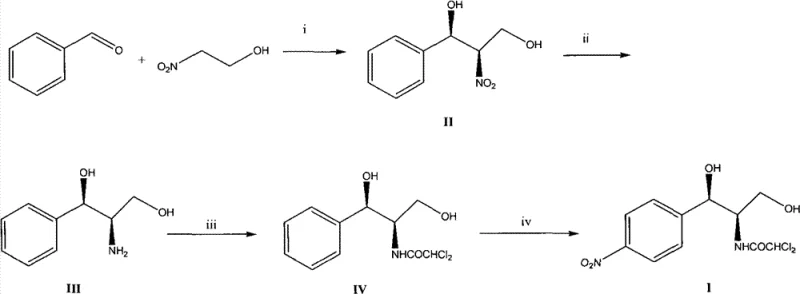
Mechanistic Insights into Cu-Catalyzed Asymmetric Henry Reaction
The cornerstone of this synthetic innovation lies in the employment of a chiral copper catalyst system that facilitates the enantioselective addition of 2-nitroethanol to benzaldehyde. The catalyst is generated in situ from copper(II) triflate and a specialized chiral ligand, specifically a 2,6-bis[(S)-4-isopropyl-1-phenyl-4,5-dihydro-1H-2-imidazolyl]pyridine derivative. This ligand architecture creates a rigid chiral environment around the copper center, effectively differentiating the prochiral faces of the aldehyde substrate during the nucleophilic attack. The coordination of the nitroalkane and the aldehyde to the metal center activates both species, lowering the activation energy for the C-C bond formation while strictly controlling the spatial orientation of the incoming groups. Experimental data from the patent indicates that this system achieves an enantiomeric excess of 93%, demonstrating the high level of stereocontrol exerted by the catalyst. Such high selectivity is crucial for pharmaceutical applications, as it minimizes the formation of diastereomeric impurities that would otherwise require complex and yield-losing purification steps downstream. 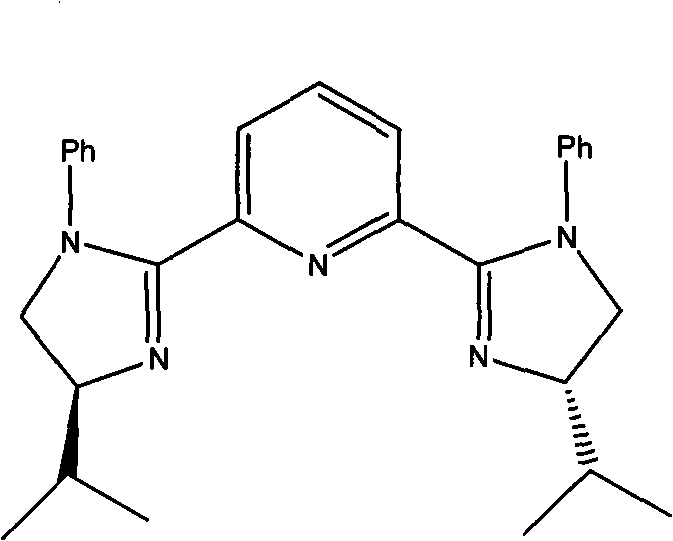
Beyond stereocontrol, the mechanistic design of this process inherently supports superior impurity management throughout the synthesis. By avoiding the harsh conditions and stoichiometric reagents associated with the traditional aluminum isopropoxide reduction, the new route significantly reduces the profile of metal-containing byproducts. The catalytic hydrogenation step used to convert the nitro intermediate to the amine is highly chemoselective, leaving the benzylic hydroxyl groups intact while cleanly reducing the nitro functionality. Furthermore, the final nitration is performed under controlled low-temperature conditions using a mixture of sulfuric and nitric acid, which helps to minimize over-nitration or oxidative degradation of the sensitive dichloroacetamide side chain. The cumulative effect of these mechanistic choices is a crude product profile that is much cleaner than that obtained from conventional methods, facilitating the attainment of greater than 98.0% purity through simple recrystallization. This high level of purity is essential for meeting the stringent quality standards required for antibiotic APIs intended for human therapeutic use.
How to Synthesize Chloramphenicol Efficiently
The synthesis of chloramphenicol via this patented route offers a practical and scalable solution for manufacturers seeking to optimize their production capabilities. The process begins with the preparation of the chiral catalyst in a dry solvent such as dioxane, followed by the addition of benzaldehyde and 2-nitroethanol under inert atmosphere to ensure consistent catalytic activity. The subsequent steps involving hydrogenation, acylation, and nitration are all standard unit operations that can be easily integrated into existing pharmaceutical manufacturing infrastructure without requiring exotic equipment. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Perform an asymmetric Henry reaction between benzaldehyde and 2-nitroethanol using a copper-chiral ligand complex to obtain (1R, 2R)-2-nitro-1-phenyl-1,3-propanediol.
- Subject the nitro-diol intermediate to catalytic hydrogenation using palladium on carbon to reduce the nitro group to an amine.
- React the resulting amino-diol with methyl dichloroacetate to form the dichloroacetamide derivative, followed by nitration to yield chloramphenicol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages regarding cost stability and supply security. The shift from p-nitroacetophenone to benzaldehyde as the starting material leverages a commodity chemical with a robust global supply chain, thereby mitigating the risk of raw material shortages and price volatility. Additionally, the elimination of the chiral resolution step removes the inherent 50% yield penalty associated with separating enantiomers, effectively doubling the mass efficiency of the process relative to the input materials. This improvement in atom economy directly correlates to a reduction in the cost of goods sold, as less raw material is required to produce the same amount of final API. Furthermore, the avoidance of aluminum isopropoxide not only lowers reagent costs but also drastically simplifies waste treatment protocols, leading to significant operational savings in environmental compliance and disposal fees.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive specialty reagents with commodity chemicals and the removal of yield-limiting purification steps. By bypassing the chiral resolution stage, the process avoids the massive material loss that traditionally inflates the cost of chiral antibiotics. The use of catalytic amounts of copper and ligand, rather than stoichiometric amounts of metal reductants, further decreases the direct material costs per kilogram of product. Moreover, the simplified workup procedures reduce the consumption of solvents and energy, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: Relying on benzaldehyde, a widely produced industrial chemical, ensures a stable and continuous supply of starting materials, unlike more specialized intermediates that may be subject to supply disruptions. The shortened synthetic route, comprising only four steps compared to the longer traditional sequence, reduces the lead time required for production batches. This agility allows manufacturers to respond more quickly to fluctuations in market demand for chloramphenicol, ensuring that inventory levels can be maintained without excessive safety stock. The robustness of the catalytic system also implies consistent batch-to-batch quality, reducing the risk of production delays caused by failed quality control tests.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily managed in large-scale reactors without requiring extreme temperatures or pressures. The significant reduction in hazardous waste, particularly the elimination of aluminum sludge, aligns with increasingly strict environmental regulations and corporate sustainability goals. This 'greener' profile facilitates easier permitting for new production facilities and reduces the long-term liability associated with waste storage and treatment. Consequently, the technology supports sustainable growth and expansion of production capacity to meet global health needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced chloramphenicol synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent documentation. They are intended to provide clarity on the feasibility and benefits of adopting this route for commercial manufacturing purposes.
Q: How does this new synthesis route improve upon traditional chloramphenicol manufacturing?
A: The novel route replaces the traditional p-nitroacetophenone pathway, eliminating the need for chiral resolution which theoretically limits yield to 50%, and avoids the use of aluminum isopropoxide, significantly reducing hazardous waste generation.
Q: What represents the key technological breakthrough in this patent?
A: The core breakthrough is the application of a copper-based chiral catalyst system for the asymmetric Henry reaction, achieving high enantiomeric excess (93% ee) directly from achiral starting materials like benzaldehyde.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials, operates under manageable conditions, and simplifies the purification steps, making it highly adaptable for commercial scale-up with reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic asymmetric synthesis for the global antibiotic market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international pharmacopeial standards. We are uniquely positioned to leverage this patented technology to deliver high-purity chloramphenicol intermediates and APIs that offer superior value to our partners.
We invite forward-thinking pharmaceutical companies to collaborate with us to explore the full commercial potential of this efficient synthesis route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can optimize your supply chain and reduce your overall manufacturing costs.
