Revolutionizing Chloramphenicol Production: A High-Yield Asymmetric Catalytic Route for Global API Manufacturers
The global demand for broad-spectrum antibiotics remains a critical pillar of modern healthcare infrastructure, yet the manufacturing processes for key agents like Chloramphenicol have historically been plagued by inefficiency and environmental burden. A pivotal technological breakthrough detailed in patent CN102399163B offers a transformative approach to synthesizing this vital antibiotic, shifting the paradigm from laborious multi-step sequences to a streamlined, high-yield catalytic process. This innovation centers on the strategic utilization of 4-chlorobenzaldehyde as a primary feedstock, leveraging advanced asymmetric catalysis to establish chirality early in the synthetic sequence rather than relying on wasteful resolution techniques. By integrating a novel palladium-catalyzed nitro substitution mechanism, this methodology not only simplifies the operational workflow but also significantly enhances the atom economy of the entire production line. For pharmaceutical manufacturers and procurement strategists, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-purity antibiotic intermediates. The following analysis dissects the technical merits and commercial implications of this route, providing a roadmap for adopting more sustainable and cost-effective manufacturing standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Chloramphenicol has relied heavily on ethylbenzene as the starting raw material, a pathway characterized by excessive length and inherent thermodynamic inefficiencies. The traditional synthetic trajectory involves a cumbersome series of transformations including oxidation, nitration, bromination, ammonification, acetylation, aldol condensation, and critically, a chiral resolution step that fundamentally caps the theoretical yield. As illustrated in the conventional pathway below, the reliance on chiral resolution means that nearly half of the produced material is discarded as the unwanted enantiomer, driving the actual industrial yield down to approximately 30% when calculated from the initial ethylbenzene feedstock. Furthermore, the legacy process necessitates the use of aluminum isopropoxide for reduction steps, a reagent known for generating substantial quantities of aluminum-containing waste that is notoriously difficult and costly to treat. These factors combine to create a production model with a heavy environmental footprint and inflated operational costs, rendering it increasingly unsustainable in a regulatory environment that demands greener chemistry.
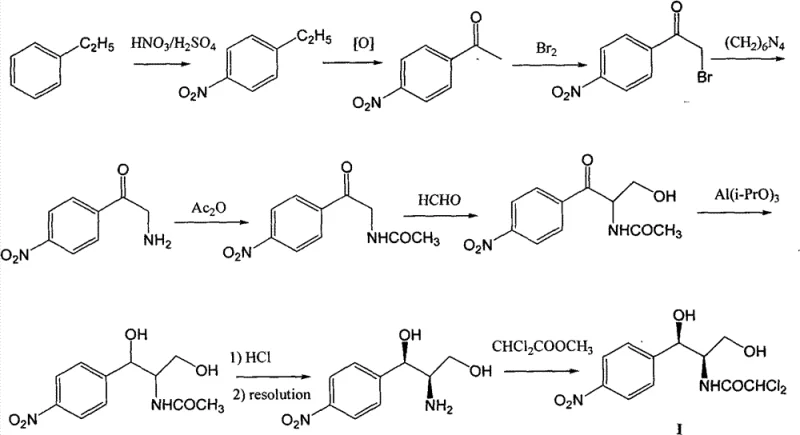
The Novel Approach
In stark contrast to the archaic ethylbenzene route, the methodology disclosed in CN102399163B introduces a concise four-step synthesis that begins with the readily available and inexpensive 4-chlorobenzaldehyde. This novel approach ingeniously bypasses the need for chiral resolution by employing an asymmetric Henry reaction to directly install the required stereochemistry with high enantiomeric excess, effectively doubling the potential yield compared to resolution-based strategies. The subsequent steps utilize catalytic hydrogenation and a unique palladium-mediated nitro substitution to construct the final molecular architecture, eliminating the need for hazardous aluminum reagents and complex protection-deprotection sequences. As depicted in the reaction scheme below, the transition from a linear, waste-generating process to this convergent catalytic route represents a significant leap in process chemistry, offering a total recovery rate of approximately 60% to 66%. This dramatic improvement in efficiency not only lowers the cost of goods sold but also aligns perfectly with modern principles of green chemistry by minimizing waste generation and energy consumption throughout the manufacturing lifecycle.
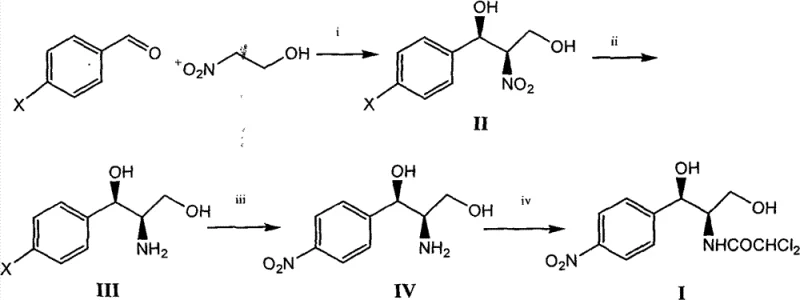
Mechanistic Insights into Asymmetric Henry Reaction and Pd-Catalyzed Substitution
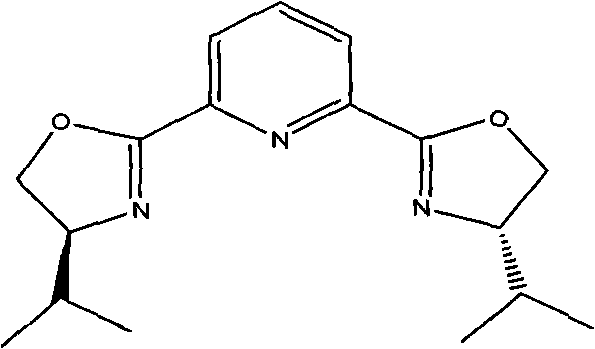
The cornerstone of this innovative synthesis is the initial asymmetric Henry reaction, where 4-chlorobenzaldehyde reacts with 2-nitroethanol in the presence of a sophisticated chiral catalyst system. The patent specifies the use of copper triflate (Cu(OTf)2) coordinated with a chiral bis-imidazoline pyridine ligand, a complex that creates a highly defined chiral environment around the metal center. This coordination geometry directs the nucleophilic attack of the nitroalkane onto the aldehyde with exceptional stereoselectivity, yielding the (1R,2R)-2-nitro-1-(4-chlorophenyl)-1,3-propanediol intermediate with an enantiomeric excess (e.e.) exceeding 93%. The structure of this critical chiral ligand, shown below, features bulky sec-butyl groups that provide the necessary steric hindrance to differentiate between the prochiral faces of the aldehyde substrate. This level of control at the very first step ensures that downstream processing does not require purification of diastereomers, thereby simplifying the entire workflow and preserving the optical purity of the final antibiotic product without the need for recrystallization of salts.
Following the establishment of chirality, the process employs a remarkable palladium-catalyzed nitro substitution to convert the chloro-intermediate into the nitro-precursor, a transformation that defies conventional nucleophilic aromatic substitution limitations. Traditionally, introducing a nitro group onto an aromatic ring with such precision requires harsh nitration conditions that often lead to mixtures of isomers and over-nitrated byproducts. However, this patent utilizes a catalytic system comprising tris(dibenzylideneacetone)dipalladium (Pd2(dba)3) and specialized phosphine ligands like t-BuXPhos in conjunction with sodium nitrite. This catalytic cycle facilitates the precise replacement of the chlorine atom with a nitro group under relatively mild thermal conditions (105-110°C), ensuring that the sensitive amino and hydroxyl functionalities on the side chain remain intact. The mechanistic elegance of this step lies in its ability to achieve regioselective functionalization without compromising the stereochemical integrity established in the first step, ultimately delivering the key intermediate with high purity and minimal impurity profiles.
How to Synthesize Chloramphenicol Efficiently
The practical implementation of this synthesis requires careful attention to catalyst loading and reaction conditions to maximize the benefits of the patented route. The process begins with the preparation of the chiral nitro-diol, followed by reduction and substitution, culminating in the final acylation. While the patent provides specific molar ratios and solvent systems, scaling this chemistry requires robust process control to maintain the high enantiomeric purity and yield observed in the laboratory examples. The detailed standardized synthesis steps for implementing this high-efficiency route are outlined in the guide below.
- Perform an asymmetric Henry reaction between 4-chlorobenzaldehyde and 2-nitroethanol using a chiral copper catalyst to obtain (1R,2R)-2-nitro-1-(4-chlorophenyl)-1,3-propanediol.
- Execute catalytic hydrogenation using Pd/C to reduce the nitro group to an amine, yielding (1R,2R)-2-amino-1-(4-chlorophenyl)-1,3-propanediol.
- Conduct a palladium-catalyzed nitro substitution reaction using Sodium Nitrite to replace the chloro group with a nitro group, forming the key nitro-intermediate.
- Finalize the synthesis through dichloroacetylation with methyl dichloroacetate to produce high-purity Chloramphenicol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages that extend far beyond simple chemical curiosity. The shift from a low-yield, waste-intensive process to a high-efficiency catalytic route fundamentally alters the cost structure of Chloramphenicol production, offering significant opportunities for margin improvement and supply security. By eliminating the theoretical 50% loss associated with chiral resolution, manufacturers can effectively double their output from the same quantity of raw materials, creating a buffer against feedstock price volatility. Furthermore, the removal of aluminum isopropoxide from the process inventory reduces the logistical burden of handling hazardous reagents and mitigates the costs associated with waste disposal and environmental compliance. These operational efficiencies collectively contribute to a more resilient supply chain capable of meeting global demand with greater reliability and lower total cost of ownership.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology stems from the drastic increase in overall yield, which rises from a historical average of 30% to over 60%. This doubling of efficiency means that the consumption of starting materials like 4-chlorobenzaldehyde is effectively halved per unit of final product, leading to substantial savings in raw material procurement costs. Additionally, the elimination of the resolution step removes the need for resolving agents and the associated solvent volumes required for separating enantiomers, further reducing utility and material expenses. The simplified four-step sequence also shortens the production cycle time, allowing for faster turnover of manufacturing assets and reduced work-in-progress inventory costs. Consequently, the cost of goods sold (COGS) is significantly optimized, providing a competitive pricing advantage in the global antibiotic market.
- Enhanced Supply Chain Reliability: Relying on 4-chlorobenzaldehyde as a starting material offers superior supply chain stability compared to the multi-step derivation from ethylbenzene. 4-Chlorobenzaldehyde is a commodity chemical produced at massive scales for various industries, ensuring consistent availability and shielding manufacturers from the bottlenecks often seen in specialized intermediate supply chains. The robustness of the catalytic steps, particularly the use of stable palladium and copper systems, ensures that production campaigns can run with high reproducibility and minimal batch failures. This reliability is crucial for maintaining continuous supply to downstream formulators and preventing stockouts that can disrupt the availability of essential medicines. Moreover, the shorter synthesis route reduces the number of intermediate storage and transportation requirements, streamlining the logistics network and minimizing the risk of quality degradation during transit.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this process offers a clear path to scalable, eco-friendly manufacturing. The avoidance of aluminum isopropoxide eliminates the generation of large volumes of aluminum sludge, a persistent waste management challenge in the pharmaceutical industry. The catalytic nature of the key transformations means that reagent usage is minimized, aligning with green chemistry principles and facilitating easier compliance with increasingly stringent environmental regulations. The high purity of the crude product (>98.0%) achieved before final crystallization reduces the load on purification units and minimizes solvent waste from extensive recrystallization processes. This environmental profile not only reduces disposal costs but also enhances the corporate sustainability credentials of the manufacturer, a factor that is becoming increasingly important in supplier selection criteria for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Chloramphenicol synthesis route. These answers are derived directly from the experimental data and claims presented in patent CN102399163B, providing clarity on yield expectations, catalyst performance, and regulatory considerations. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term viability of this supply source.
Q: How does the new asymmetric route improve yield compared to traditional ethylbenzene methods?
A: Traditional routes starting from ethylbenzene involve chiral resolution, which theoretically limits yield to 50% at that step, resulting in an overall industrial yield of only about 30%. The new asymmetric catalytic route bypasses resolution entirely, achieving a total recovery of approximately 60-66%.
Q: What are the environmental benefits of avoiding aluminum isopropoxide reduction?
A: Conventional methods utilize aluminum isopropoxide for reduction, which generates significant amounts of difficult-to-handle aluminum waste. The new method employs catalytic hydrogenation and palladium-catalyzed substitution, drastically reducing the three wastes (wastewater, waste gas, solid waste) associated with production.
Q: Is the chiral catalyst used in this process commercially viable for scale-up?
A: Yes, the process utilizes accessible raw materials like 4-chlorobenzaldehyde and robust catalyst systems (Cu(OTf)2 with chiral ligands and Pd2(dba)3). The patent explicitly states the method is suitable for industrial production with high purity (>98.0%) achievable via simple recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes like the one described in CN102399163B requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent can be reliably replicated on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped to monitor critical parameters such as enantiomeric excess and residual metal content, guaranteeing that every batch of Chloramphenicol intermediate meets the highest global pharmacopeial standards. Our commitment to quality assurance ensures that the theoretical advantages of this asymmetric route are fully realized in the final commercial product delivered to your facility.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their antibiotic supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this efficient route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that secure your supply of high-quality Chloramphenicol while driving down manufacturing costs.
