Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
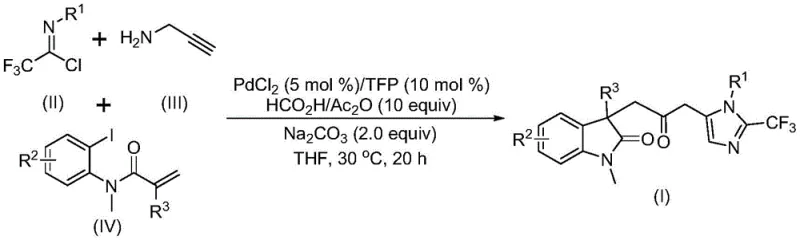
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as the core backbone for bioactive molecules. Patent CN115353511A discloses a groundbreaking multicomponent method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in process chemistry by addressing the longstanding challenges associated with carbonylation reactions. Traditionally, introducing a carbonyl bridge between two heterocyclic systems required hazardous carbon monoxide gas under high pressure, posing severe safety and logistical hurdles for manufacturing facilities. The disclosed invention circumvents these issues by employing a transition metal palladium-catalyzed cascade reaction that operates under mild conditions, utilizing safe and readily available starting materials. For R&D directors and process chemists, this methodology offers a robust platform for generating diverse libraries of fluorinated biheterocycles, which are increasingly valuable in modern drug discovery for their metabolic stability and binding affinity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks containing a carbonyl linker has been fraught with synthetic inefficiencies and safety concerns. Conventional strategies often rely on the direct coupling of two pre-formed heterocyclic substrates, which frequently suffers from low atom economy and requires harsh activation conditions that can degrade sensitive functional groups. Alternatively, oxidative cyclization methods involving dual nucleophiles often demand stoichiometric oxidants, generating substantial chemical waste and complicating downstream purification processes. Most critically, traditional carbonylation techniques necessitate the use of gaseous carbon monoxide, a highly toxic and flammable reagent that requires specialized high-pressure reactors and rigorous safety protocols. These factors collectively inflate the cost of goods sold (COGS) and limit the scalability of such processes, making them less attractive for the commercial production of high-purity pharmaceutical intermediates where supply chain reliability is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated multicomponent reaction (MCR) strategy that converges three distinct building blocks in a single operational step. By reacting trifluoroethylimidoyl chloride, propargylamine, and an acrylamide derivative in the presence of a palladium catalyst, the method efficiently assembles the complex carbonyl-bridged architecture. A key innovation is the use of a formic acid and acetic anhydride mixture as an ex situ source of carbon monoxide, thereby eliminating the need for external CO gas cylinders. This transformation proceeds at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating exceptional functional group tolerance. The reaction mechanism involves a cascade of palladium-catalyzed events, including C-I bond activation, intramolecular Heck cyclization, and carbonylation, followed by base-promoted amidine formation and final cyclization. This streamlined workflow not only enhances reaction efficiency but also drastically simplifies the operational complexity for manufacturing teams.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, offering deep insights for researchers aiming to optimize similar cascades. The cycle likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating an aryl-palladium(II) intermediate. This species subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a stable five-membered alkyl-palladium ring characteristic of the indolinone core. Crucially, the carbon monoxide required for the bridging carbonyl group is generated in situ from the decomposition of the formic acid/acetic anhydride adduct. This CO molecule inserts into the palladium-carbon bond to yield an acyl-palladium intermediate, setting the stage for the final ring closure. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine under basic conditions to form a trifluoroacetamidine intermediate, which then undergoes isomerization and cyclization activated by the acyl-palladium species to furnish the final imidazole ring fused to the system.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By telescoping multiple bond-forming events into a single pot, the exposure of reactive intermediates to the bulk environment is minimized, reducing the formation of hydrolysis byproducts or oligomeric impurities. The use of sodium carbonate as a mild base ensures that the sensitive imidoyl chloride moiety is consumed efficiently without promoting excessive degradation of the acrylamide component. Furthermore, the specific choice of the trifurylphosphine (TFP) ligand stabilizes the palladium center throughout the catalytic cycle, preventing premature catalyst deactivation which is a common failure mode in carbonylation reactions. For quality assurance teams, understanding this mechanism highlights the importance of strict moisture control during the CO surrogate generation phase, as water can compete with the amine nucleophile, though the patent indicates the system is robust enough to tolerate standard laboratory conditions while maintaining high purity specifications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
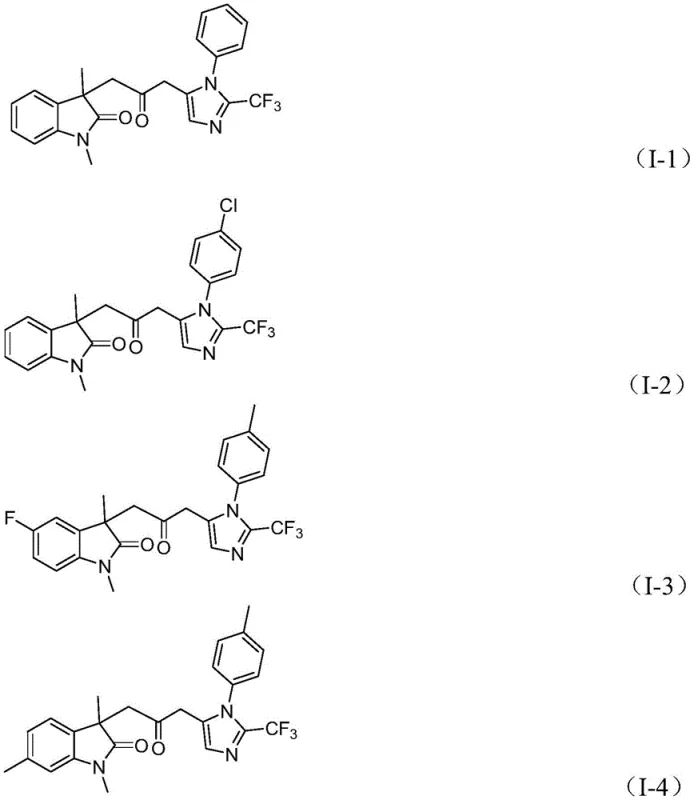
Implementing this synthesis in a pilot or production plant requires careful attention to reagent addition sequences and temperature control to maximize yield and safety. The protocol dictates dissolving the palladium catalyst, ligand, and base in the organic solvent before introducing the substrates to ensure homogeneous catalysis initiation. The reaction is typically run for 12 to 20 hours at 30°C, a timeframe that balances complete conversion with operational throughput. Post-reaction workup is notably straightforward, involving simple filtration to remove inorganic salts followed by silica gel treatment and column chromatography, which aligns well with standard purification trains in fine chemical facilities. For detailed standard operating procedures and specific molar ratios optimized for different substrate classes, please refer to the technical guide below.
- Mix palladium chloride, trifurylphosphine ligand, sodium carbonate, and the CO surrogate system (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction mixture.
- Stir the reaction at 30°C for 12 to 20 hours, then filter and purify the crude product via column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and vendor reliability. The shift away from hazardous gaseous reagents to liquid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility expands the pool of qualified contract manufacturing organizations (CMOs), thereby enhancing supply chain resilience and reducing the risk of single-source bottlenecks. Moreover, the use of commodity chemicals like propargylamine and acrylamide derivatives ensures a stable and cost-effective raw material supply base, insulating the project from the volatility often seen with exotic custom synthons. The high atom economy of the multicomponent approach means less waste generation per kilogram of product, which directly correlates to lower disposal costs and a reduced environmental footprint, aligning with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure removes a significant capital expenditure barrier, while the use of inexpensive palladium chloride instead of more exotic catalysts lowers the recurring cost of goods. By combining three synthetic steps into one pot, the process drastically reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours associated with intermediate isolations. This consolidation of operations leads to substantial cost savings in the overall manufacturing budget without compromising the quality of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: The starting materials identified in this protocol, such as trifluoroethylimidoyl chlorides and substituted acrylamides, are derived from widely available industrial feedstocks. This broad availability ensures that production schedules are not held hostage by the lead times of niche reagents. Furthermore, the robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens and electron-withdrawing substituents, means that supply chain disruptions due to specific substrate purity issues are minimized. This reliability is critical for maintaining continuous production flows required by just-in-time manufacturing models in the pharmaceutical sector.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of scaling this reaction to the gram level with consistent results, providing a clear pathway for kilogram and ton-scale production. The mild reaction temperature of 30°C reduces the energy load on plant utilities compared to high-temperature reflux processes. Additionally, the avoidance of toxic CO gas simplifies regulatory compliance and permitting processes, accelerating the time-to-market for new drug candidates. The simplified workup procedure also reduces the volume of organic waste streams, facilitating easier adherence to increasingly stringent environmental protection regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details helps stakeholders evaluate the fit of this chemistry within their existing development pipelines and manufacturing capabilities.
Q: What are the safety advantages of this carbonylation method compared to traditional methods?
A: This method utilizes a formic acid and acetic anhydride mixture as a carbon monoxide surrogate, eliminating the need for handling toxic, high-pressure CO gas cylinders, which significantly reduces safety risks and infrastructure costs.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The reaction demonstrates excellent compatibility with various substituents, including alkyl, halogen (Cl, Br, F), alkoxy, nitro, and trifluoromethyl groups on the aromatic rings, allowing for diverse library synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly mentions successful expansion to gram-scale reactions with simple post-treatment procedures, indicating strong potential for kilogram to ton-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multicomponent technology for accelerating drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry safely, and our rigorous QC labs enforce stringent purity specifications to meet the demanding requirements of global regulatory agencies. We are committed to delivering high-purity pharmaceutical intermediates that empower your research teams to focus on innovation rather than supply chain constraints.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient methodology. Contact us today to request specific COA data for related analogues and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the development of next-generation therapeutic agents.
